M.M. Ibrahim, A.M. Fathy, S.A. Al-Harbi et al.
Journal of Organometallic Chemistry 939 (2021) 121764
Table 14
3.8.1. Effect of the catalysis type
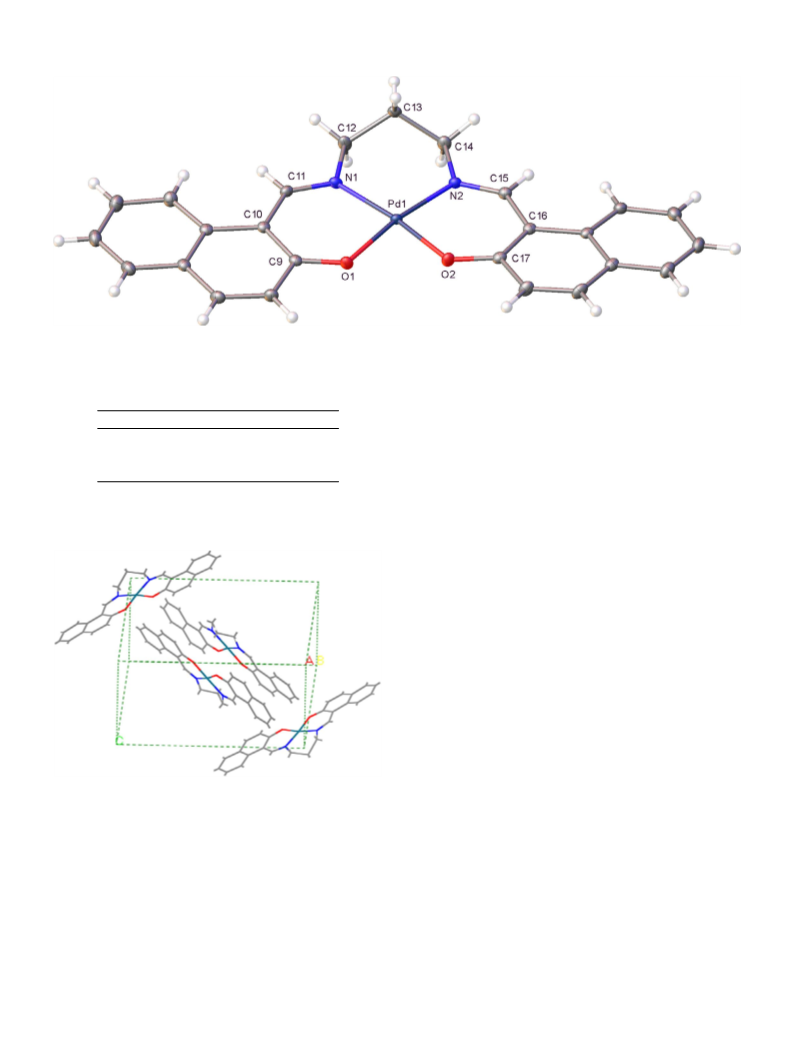
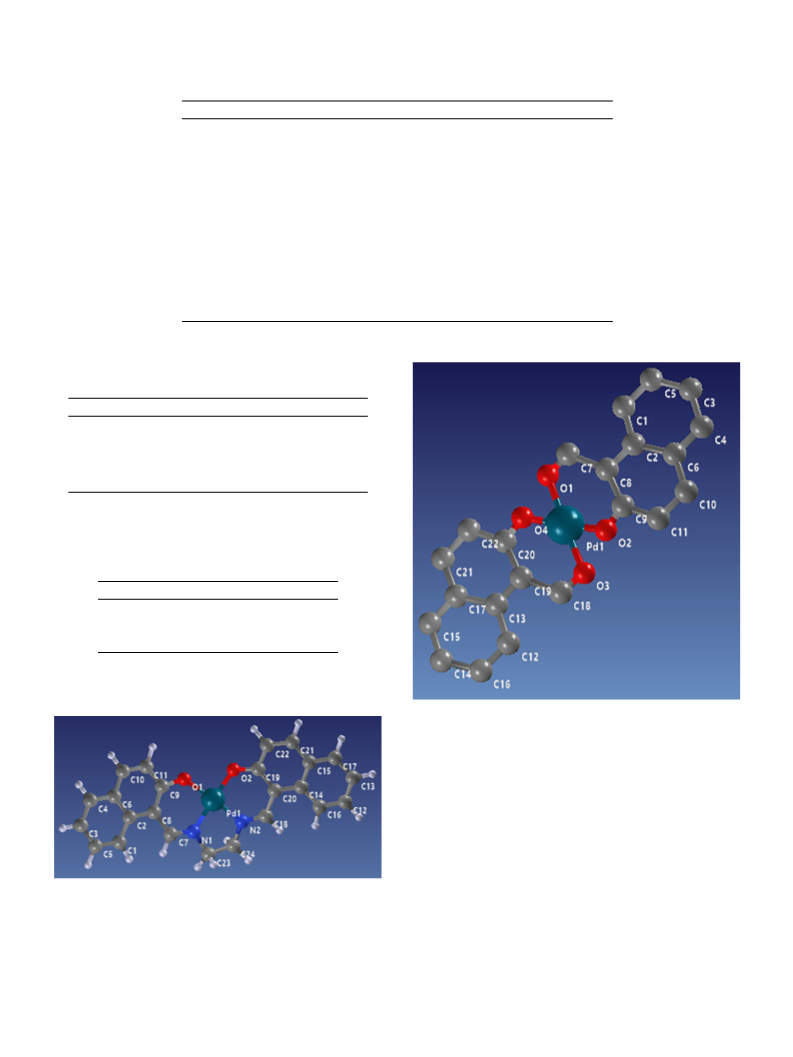
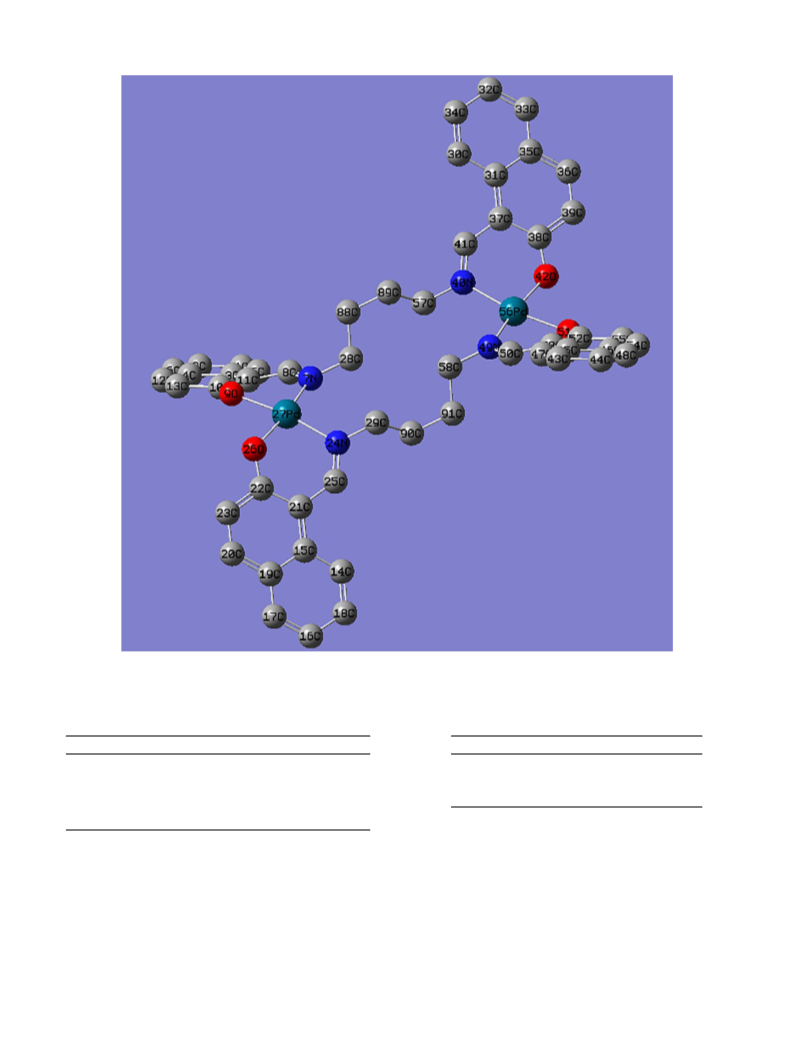
Bond angles (°) of palladium(II) complex 3, (PdL3).
Under prevailing catalytic experimental conditions and with ex-
ception of [PdL2] in DMF, it is difficult to achieve a specific catalysis
type since in other solvents used a certain amount of the catalysts
is always soluble. However, the yield percentage (Table 16) remains
fairly constant indicating that the candidate palladium(II) com-
plexes can catalyze the hydrogenation of cyclohexene by both ho-
mogenous and heterogeneous catalysis. It is assumed that during
the catalytic hydrogenation cycle of alkene by a transition metal,
both H2 and olefin must exist together in the coordination sphere
of the transition metal center to achieve effective hydrogenation.
Regarding the precedence for entry of reaction partners in the co-
ordination sphere of the transition metal center, there are two pos-
sibilities namely the hydride route and the olefin route.
Pd27; (τ4 = 0.1476)
Pd56; (τ4 = 0.1476)
N7 - Pd27 - O9
N7 - Pd27 - N24
N7 - Pd27 - O26
O9 - Pd27 - N24
O9 - Pd27 - O26
N24 - Pd27 - O26
86.4515
103.5371
169.6727
169.6583
84.3238
85.4969
N40 - Pd56 - O42
N40 - Pd56 - N49
N40 - Pd56 - O51
O42 - Pd56 - N49
O42 - Pd56 - O51
N49 - Pd56 - O51
85.497
103.539
169.6616
169.6734
84.3279
86.4455
Table 15
3
˚
Bond lengths (A) of palladium(II) complex 3 (PdL ).
Pd27
Pd56
The hydride route includes activation of H2 on the metal center
as a primary step, with subsequent coordination and hydrogena-
tion of alkene.
N7 - Pd27
O9 - Pd27
N24 - Pd27
O26 - Pd27
2.0755
2.0269
2.0927
2.0315
N40 - Pd56
O42 - Pd56
N49 - Pd56
O51 - Pd56
2.0927
2.0314
2.0755
2.0269
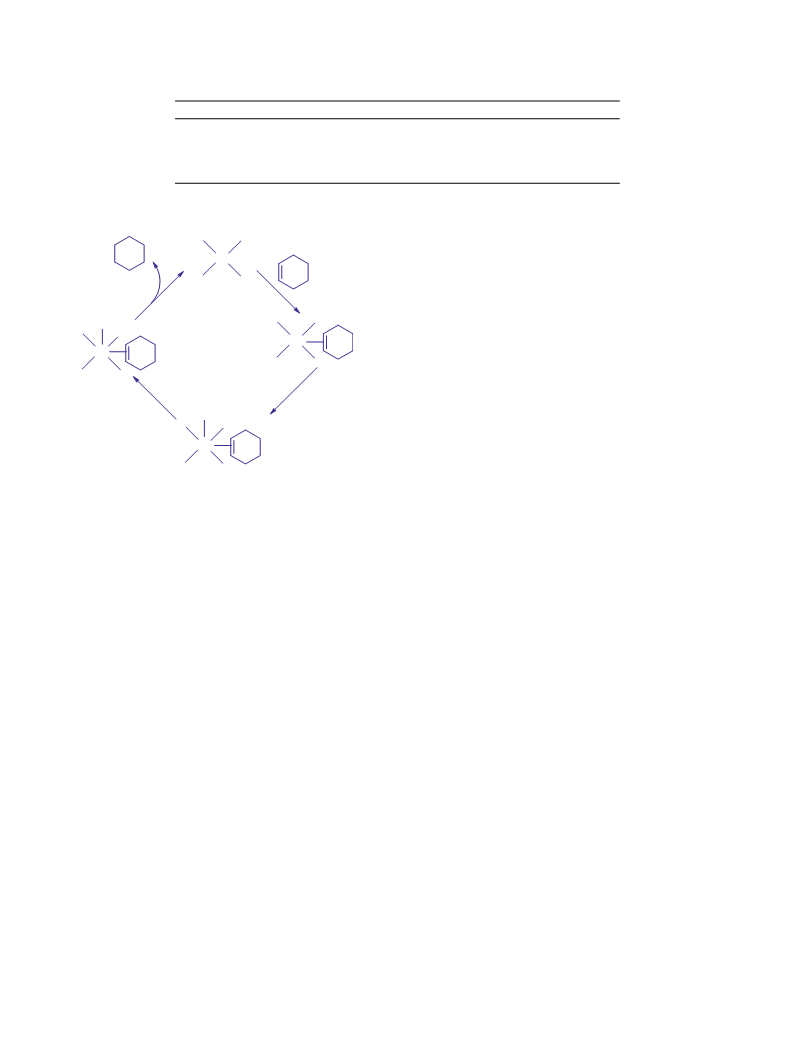
Catalyst + H2 ǭǃǃ ꢀ Catalyst(H)H + alkene → Alkane + Catalyst
In the olefin route, the olefinic substrate molecule first coordi-
natively bonded to the catalyst
Catalyst + olefin ꢀ Catalyst-olefin + H2 → Alkane + Catalyst
For the hydride route, approaching H2 to the square planar pal-
ladium(II) center must proceed in the Z-direction since the equa-
torial xy-plane is occupied by the diimine ligand molecule. In this
ular orbital HOMO of the C=C (π); in this case electrons flow from
π orbital of C=C to σ∗ of H2. On the other hand, molecular or-
bital interaction can occur between the LUMO of C=C (π∗) with
the HOMO of H2 (σ) and here the electrons flow from σ orbital of
H2 to π∗ of C=C. But neither the molecular orbitals σ of H2 and
π∗ of C=C nor π of C=C and σ∗ of H2 have not any net overlap,
hence the reaction is "symmetry forbidden".
2
situation the 4dZ orbital must be lifted energetically and the
change in the energy state of this orbital is reached through the
addition of a suitable electron donor in the axial position, and the
addition could proceed through the 5pZ orbital, which acts as an
electron acceptor. The alkene coordination with palladium(II) oc-
curs without barriers in the similarity to a reported case where
the stabilizing energy resulting from this coordination was calcu-
lated to be 35.1 kJ/mol [28]. Therefore the initiation step is coordi-
nation of cyclohexene to the palladium(II) center to lift the doubly
Generally, the role of the present palladium(II) Schiff base com-
plexes (catalysts) is to circumvent these symmetry restrictions. The
catalytic role played by the metal center is likely due to the for-
mation of an olefin metal bond in one of two ways. The first
way includes bond formation via the back donation from filled d-
orbital to the empty molecular orbital LUMO (π∗) of the olefin.
The transferring of the electron density to the LUMO orbital, π∗,
of the olefin, will lead to that this orbital becomes now, partially,
a HOMO; obviously has the correct symmetry to interact with
the LUMO orbital σ∗ of H2 molecule. The second route may pass
through the flow of electrons from the olefin bonding orbit π to
an empty orbital of the metal. The loss of the electron density of
π orbital of olefin in the metal - olefin σ bond makes this orbital
(π), in part a LUMO, has the suitable symmetry to accept the elec-
trons flow from the HOMO σ orbit of H2.
2
occupied 4dZ orbital. Consequently, the five coordinated cyclohex-
ene palladium(II) complex is the key intermediate in the catalytic
cycle. The orbital interaction between the palladium(II) center and
the H2 molecule can be explained by the proper geometrical inter-
2
action of the energetically lifted 4dZ orbital with the LUMO of the
H2 molecule, which can be achieved by the "end on" overlapping
2
of both orbitals, σ∗ (LUMO) of H2 and 4dZ of palladium(II) center.
This discussion explains that the running diimine palladium(II)
complexes, whether completely or partially insoluble, are pre-
catalysts that acquire their catalytic effectiveness in the reaction
medium by binding to cyclohexene.
One may express these effects of the transition metal center are
due to the exchange of electrons with incorrect symmetries across
its orbital system to change them into right symmetries.
3.8.2. Effect of the catalyst structure
The values of cyclohexane yield (Table 16) demonstrate that,
the alkyl bridge length between the two azomethine nitrogens has
no effect on the catalytic activity of studied palladium(II) com-
plexes. This can be attributed to the fact that all the examined
palladium(II) complexes have the same square planar geometry.
Since the Schiff base ligand molecule in these complexes is in
the xy plane, the atoms within the bridge are not too far off this
plan. This means that they do not present any substantial steric
hindrance for the coordination of the central metal ion with the
hydrogen molecule or with cyclohexene. This is also in a good
agreement with the above discussion in which it is supposed that
the catalytic role of the current palladium(II) complexes is mainly
As a part of this work, the existing palladium(II) complexes
were employed as catalysts for cyclohexene hydrogenation by H2.
Initial ratings for the palladium(II) complexes in the cyclohexene
hydrogenation were carried out at hydrogen pressure of 490 mm
Hg, 22°C, catalyst and cyclohexene concentrations are 0.0026 and
0.04 M respectively. Under these conditions, the current complexes
showed catalytic activities to afford 100% cyclohexane with conver-
sions ranging from 80.34% to 99% within 150 h. In order to confirm
the catalytic potential of the examined palladium(II) complexes in
fully observed catalytic hydrogenation reactions, blank experiments
were performed without using the catalysts under the prevailing
experimental conditions. The absence of cyclohexane formation in
these blank trials confirmed the catalytic tendency of palladium(II)
complexes in question. The catalytic activity of hydrogenation cat-
alysts candidates is represented in two terms using the relations:
Yield percentage = [product / (reactants + product)] × 100
The catalytic activity was studied in relation to the effect of cat-
alyst structure, catalysis type, nature and amount of the solvents
and co-solvents.
2
achieved through the interaction of the energetically lifted 4dZ or-
bital (HOMO) of the metal with the LUMO of the H2 molecule.
In the same context, quantitative comparison of the hydro-
genation of cyclohexene using three naphthaldiimine palladium(II)
complexes is represented in Fig. 8. It can be seen that there is a
slight difference in the catalytic activity of the three represented
complexes over a period of time 150 hours. Fig. 8 shows also, that
the current catalytic hydrogenation reactions are kinetically very
12

 Ibrahim, Mohamed M.
Ibrahim, Mohamed M.
 Fathy, Ahmed M.
Fathy, Ahmed M.
 Al-Harbi, Sami A.
Al-Harbi, Sami A.
 Sallam, Shehab A.
Sallam, Shehab A.
 Al-Juaid, Saleh
Al-Juaid, Saleh
 Ramadan, Abd El-Motaleb M.
Ramadan, Abd El-Motaleb M.