J Amer Oil Chem Soc (2007) 84:681–685
683
4.18(dd, J 5.5, 11.3 Hz, 1H, CHOCO), 4.20(dd, J 5.5, 11.3,
1H, CHOCO). 13C NMR CDCl3, d 14.53(1C, CH3),
23.11(1C, CH2), 25.32(1C, CH2), 29.56-30.11(10C,
10CH2), 32.34(1C, CH2), 34.58(1C, CH2CO), 63.79(1C,
CH2OH), 65.53(1C,CHOH), 70.69(1C, CH2OCO), 174.81
plicity, use of readily available low-priced urea and low
equipment costs. Therefore, glycerol carbonate was pre-
pared by calcined zinc sulfate-catalyzed condensation of
1
glycerol with urea [24] in 96% yield over 2 h. The H-
NMR (DMSO-d6) spectrum of the product was similar to
that reported very recently [25]. Since it was decided that
the experiments to be carried out with an equimolar amount
of fatty acid with glycerol carbonate, it was found that
partial excess of the latter had to be added in order to
obtain optimized yields of monoglycerides. Compared to
dimethyl carbonate, which yields 97% of glycerol car-
bonate in the reaction with glycerol in the ratio of 3/1 in the
presence of potassium carbonate in 3 h [25], the formation
of glycerol carbonate with an identical yield in 2 h starting
with cheaper urea in equimolar amount with glycerol is an
advantage.
+
(1C, CO), MS calculated for C19H38O4 = 330; found: 331.
Glycerol Monostearate 5d:
(32.2 g, 90%), White solid (from normal hexane), MP
77 °C, IR (KBr) tmax 3400(OH), 2928(CH), 2852(CH),
1735(CO), 1463(CH3). 1H NMR (CDCl3) d 0.90(t, J 7.1 Hz,
3H, CH3), 1.29(m, 28H, 14CH2), 1.63(m, 2H, CH2), 2.37(t, J
7.5 Hz, 2H, CH2), 2.60(bs, 2H, OH), 3.61(dd, J 5.9, 11.4 Hz,
1H, CHOH), 3.70(dd, J 5.9, 11.4 Hz, 1H, CHOH), 3.95(m,
1H, CHOH), 4.16(dd, J 6.1, 11.7 Hz, 1H, CHOCO), 4.22(dd,
J 6.1, 11.7 Hz, 1H, CHOCO). 13C NMR (CDCl3) d
14.54(1C, CH3), 23.11(1C, CH2), 25.34(1C, CH2), 29.56–
30.12(12C, 12CH2), 32.35(1C, CH2), 34.59(1C, CH2CO),
63.77(1C, CH2OH), 65.56(1C, CHOH), 70.69(1C, CH2O-
Condensation of lauric acid 2a as the representative fatty
acid with glycerol carbonate was examined in the presence
of triethylamine as catalyst. The progress of reaction was
followed by observing the liberation of carbon dioxide. By
withdrawing reaction samples periodically and taking their
IR spectra, the formation of the product was monitored by
the disappearance of carbonyl peaks for fatty acid and
glycerol carbonate at 1,700 and 1,785 cm–1 respectively,
and the appearance of a carbonyl peak for the ester at
1737 cm–1. The monoglyceride 5a was obtained as a white
solid, which after recrystalization from petroleum ether
(55–70 °C) afforded the pure product in 91% yield (see
Table 1).
+
CO), 174.81(1C, CO), MS calculated for C21H42O4 = 358;
found: 359.
Glycerolmonooleate 5e:
(32.5 g, 91.5%), Colorless liquid, MP 25 °C(from normal
hexane), IR(neat) tmav 3402(OH), 2922(CH),2853(CH),
1
1739(CO), 1458(CH3). H NMR (CDCl3) d0.91(t, J 7.0
Hz, 3H, CH3), 1.28(m,22H, 11CH2), 1.65(m, 2H, CH2),
2.05(m, 4H, 2 CH2-C = C), 2.38(t, J 6.0 Hz, 2H, CH2CO),
3.59(dd, J 5.5, 8.9 Hz,1H, CHOH), 3.68(dd, J 5.5, 8.9
Hz,1H, CHOH), 3.90(m,1H, CHOH), 4.16–4.18 (d, J 5.0
Hz, 2H, CH2OCO). 13C NMR (CDCl3) d14.50(1C, CH3),
23.09(1C, CH2), 25.30(1C, CH2), 27.57(1C, CH2),
27.63(1C, CH2), 29.51–30.17(8C, 8CH2), 32.31(1C, CH2),
34.56(1C, CH2CO), 63.79(1C, CH2OH), 65.44(1C,
CHOH), 70.68(1C, CH2OCO), 130.11(1C, 1C = ),
130.44(1C, 1C=), 174.78(1C, CO), MS calculated for
Based on the result obtained for glycerol monolaurate,
the reactions of other fatty acids such as myristic, palmitic,
stearic, and oleic 2b–e with glycerol carbonate were carried
out in the presence of the same catalyst. The results are
presented in Table 1.
Identification of 5a–e as the pure a-monoglycerides
were carried out on the basis of their spectroscopic infor-
mation. With the exception of glycerol monomyristate
(5b), which showed the M+ peak at 302, glycerol mon-
olaurate (5a), glycerol monopalmitate (5c), glycerol mon-
ostearate (5d) and glycerol monooleate (5e) showed the
molecular masses of 275, 331, 359 and 357 respectively,
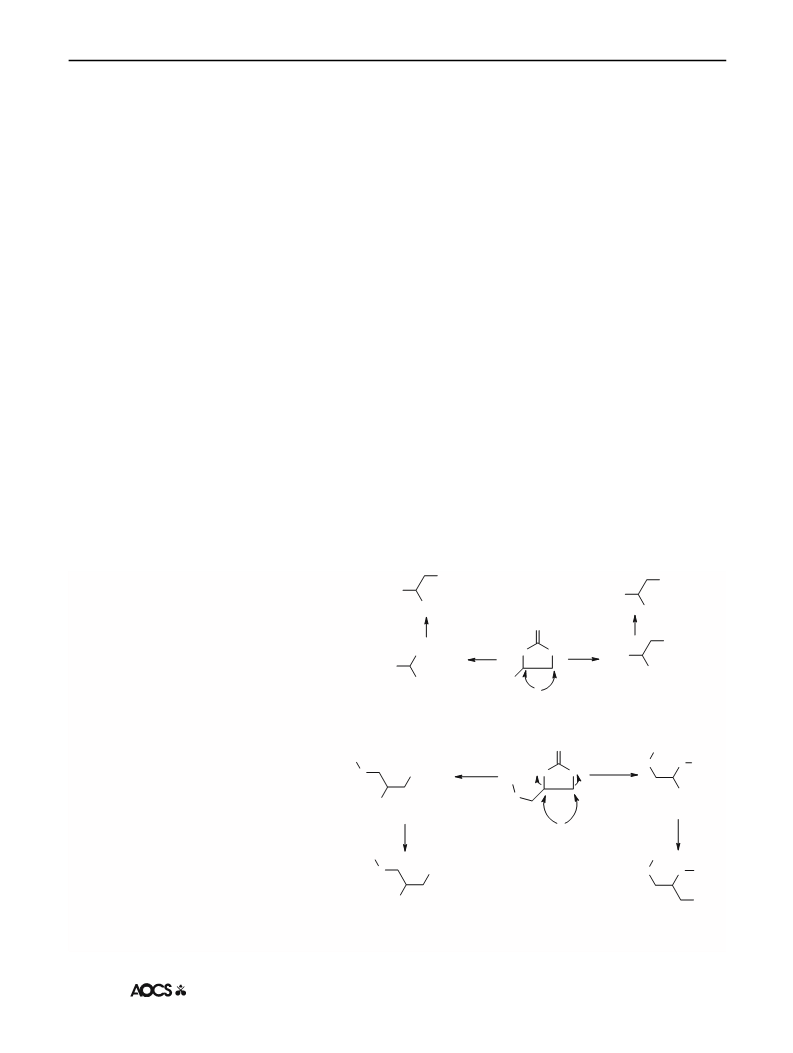
consistent with M + 1 ion peaks. It is known that the
molecular ion abundance of alcohols decreases and is often
not observable. Offsetting the parent ion peak, alcohols do
undergo ion-molecule reactions to give a pressure-depen-
dent (M + H) ion peak, which is useful for molecular
weight determination [26]. It is worth mentioning that the
mass of a-glycerol monolaurate previously prepared by
Brunel and co-workers via catalytic condensation of lauric
acid with glycidol has been reported as 257 [13].
+
C21H40O4 = 356; found: 357.
Results and Discussion
Various processes have been reported for the production of
glycerol carbonate, such as the reaction of glycerol with
phosgene [18], cyclization of 1,2-dihydroxypropyl carba-
mate [19], catalytic reaction of glycerol and CO2 [20],
transesterification of glycerol with ethylene carbonate [21]
or dimethyl carbonate [22] and finally use of urea for the
conversion of glycerol into its carbonate derivative [23].
Only the last two processes are technically and economi-
cally feasible for industrial and laboratory preparation. The
urea process is particularly attractive because of its sim-
It should be remembered that b-glycerol monofatty acid
esters can be easily identified on the basis of their H and
1
13C NMR due to the identical C1 and C3 protons and
123

 Ghandi, Mehdi
Ghandi, Mehdi
 Mostashari, Abdoljalil
Mostashari, Abdoljalil
 Karegar, Mojgan
Karegar, Mojgan
 Barzegar, Mina
Barzegar, Mina