
Journal of Organic Chemistry p. 2559 - 2562 (1987)
Update date:2022-08-11
Topics:
 Anelli, Pier Lucio
Anelli, Pier Lucio
 Biffi, Carlo
Biffi, Carlo
 Montanari, Fernando
Montanari, Fernando
 Quici, Silvio
Quici, Silvio
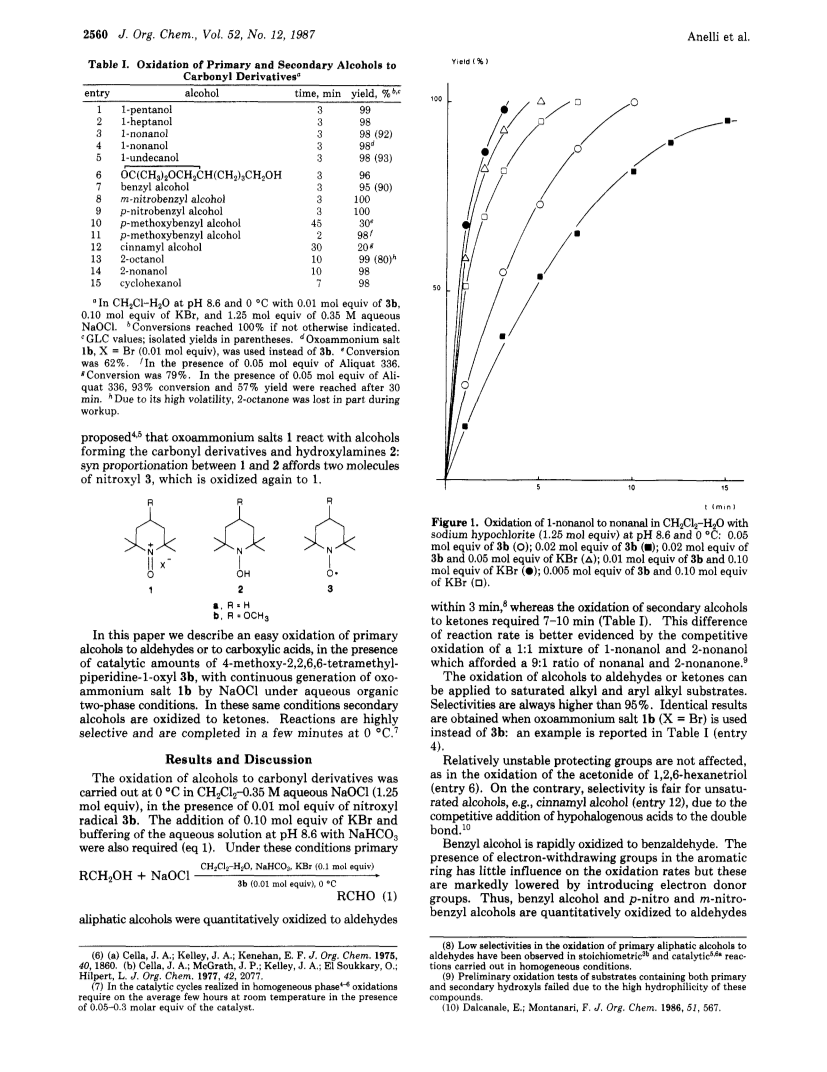
Primary alcohols are quantitatively oxidized to aldehydes in a few minutes at 0 deg C in CH2Cl2-0.35 M aqueous NaOCl in the presence of catalytic amounts of 4-methoxy-2,2,6,6-tetramethylpiperidine-1-oxyl (3b).Cocatalysis by Br- and buffering of pH at 8.6 with NaHCO3 are also required.Secondary alcohols are converted to ketones.Further oxidation of aldehydes to carboxylic acids is slow, but the reaction is completed in a few minutes under the same conditions by addition of catalytic amounts of phase-transfer catalyst.All reactions are highly selective.Onlya slight excess of NaOCl is required.The method can be applied to saturated alkyl and aryl alkyl substrates.
View More


Changsha Yonta Industry Co., Ltd.
Contact:+ 86-731-8535 2228
Address:Rm.1717, North Bldg., No.368, East 2nd Ring Road(2nd Section)
Hangzhou Dawn Ray Pharmaceutical Co.,Ltd
website:http://www.dawnraypharma.com/
Contact:+86-571-85335020
Address:1018 Guangyin Building,No.42 E.Fengqi Road,Hangzhou,310012 China
Lishui Nanming Chemical Co., Ltd
Contact:+86-0578-2134101,2697830
Address:No.19 Tongji Road Shuige Industrial zone
Anhui Redstar Pharmaceutical Corp., Ltd
Contact:+86-563-5120837
Address:Jingxian Industrial Development Zone, Anhui , China
Zhejiang PRIMAR Import & Export Trade Co., Ltd.
Contact:86-570-3630818
Address:No.1Puzhuyuan,Quhua,Zhejiang Province,China324004
Doi:10.1016/0022-328X(84)80570-7
(1984)Doi:10.1039/c39860000277
(1986)Doi:10.1021/jp962554n
(1996)Doi:10.1039/jr9430000227
(1943)Doi:10.1007/BF00354675
(1996)Doi:10.1021/ja00313a031
(1984)