Gold Nanoparticles for Aerobic Oxidation
FULL PAPER
Conclusion
After 1 h the slurry was filtered and the total gold adsorption was
checked by atomic absorption spectroscopy of the filtrate.
Preparation of Au/TiO
2
catalysts: The AuꢀTiO
2
catalysts were prepared
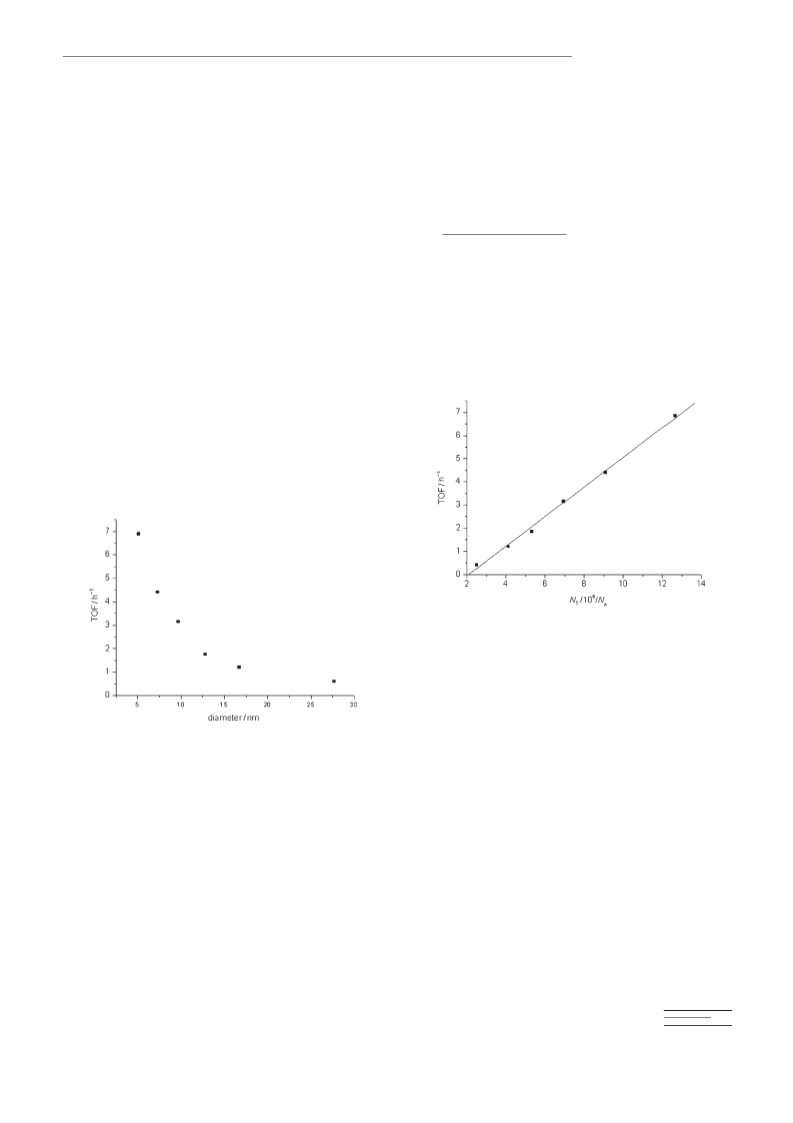
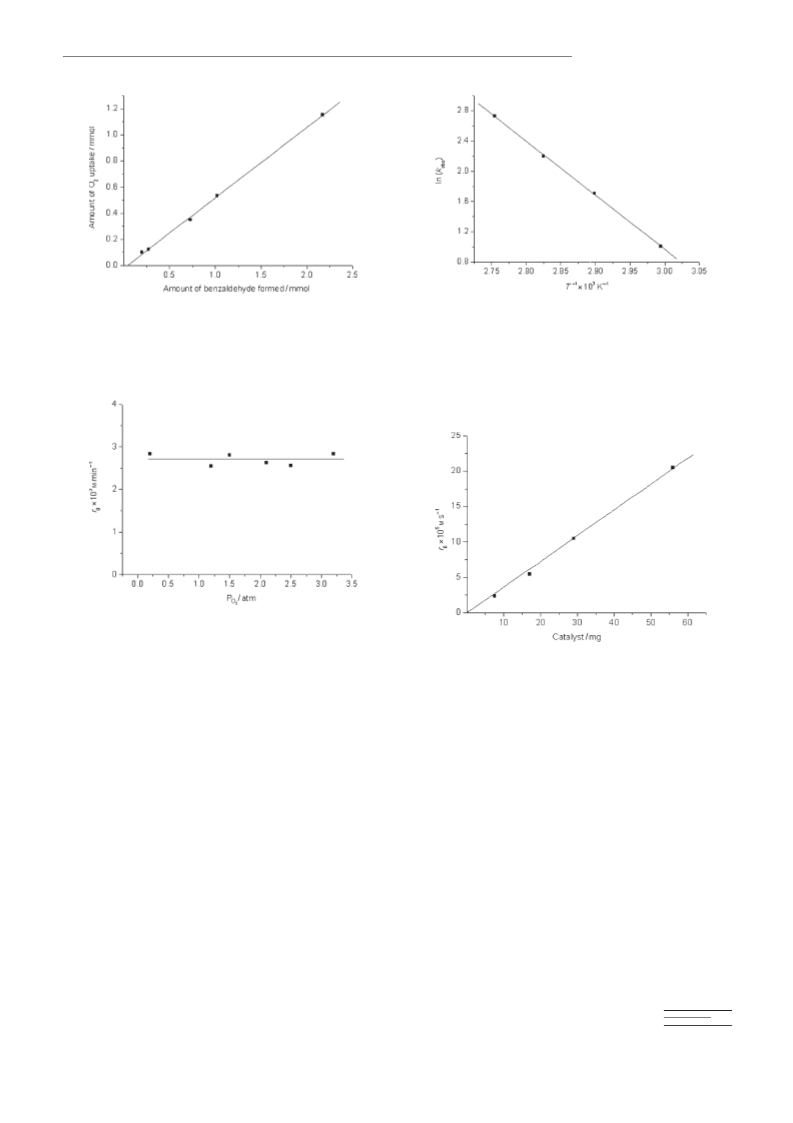
From the results presented in this manuscript it is possible
to say that the activity of gold catalyst for the aerobic oxida-
tion of alcohols involves the presence of a high density of
positive gold atoms that could act as Lewis-acid sites. These
sites coordinate with alcohols to form gold alcoholates and
they also accept hydrides. In this regard the role of the sup-
port should be on the one hand, to provide stability for posi-
tive gold species by interfacial gold-support interactions, and
on the other hand to facilitate oxygen activation to promote
the reoxidation of metal hydrides. Apparently these features
and particularly the second one, have been achieved to a re-
markable extent by cerium nanoparticles. Thus, cerium
nanoparticles interact strongly with supported gold, as evi-
denced by the temperature of hydrogen reduction and also
by the deposition–precipitation method by using P-25 titanium oxide
from Degussa as support. Particles of different sizes were obtained by
changing the pH used in the deposition of gold nanoparticles over the
TiO
2
support and the calcination temperature.
AuꢀTiO
(5.2 nm mean diameter): A solution of HAuCl ·3H O (60 mg)
in of deionised water (30 mL) was brought to pH 6.3 by addition of a so-
lution of NaOH (0.2m). Once the pH value was stable, P-25 Degussa
2
4 2
TiO
The Au/TiO
water. The catalyst was dried at 808C during 6 h and calcined using the
2
(1.0 g) was added. The slurry was vigorously stirred for 1 h at 708C.
2
catalyst was then filtered and exhaustively washed with
ꢀ
1
following temperature program: room temperature, 38Cmin , 2008C,
h. The total Au content of the final catalyst Au/TiO was 2.9 wt% as
determined by chemical analysis.
AuꢀTiO (7.3 nm mean diameter): This AuꢀTiO
2
2
2
2
catalyst was prepared
(3.2 nm),
following the same procedure used in the synthesis of AuꢀTiO
2
but at pH value of 5.0 instead of 6.3. The dried solid was calcined at
O can be adsorbed through oxygen vacancies on the sur-
4008C during 2 h. The gold content of the final catalyst was 3.0 wt%.
2
[50]
face.
The combination of these properties explains the
AuꢀTiO
and calcined at 4008C for 2 h. The gold content of the final catalyst was
.8 wt%.
For crystal analysis and indexation, Au/npCeO
by bright- and dark-field electron microscopy in a Jeol 2200 HRTEM
high-resolution transmission electron microscopy) operated at an accel-
erating voltage of 200 kV. The TEM images and the particle size distribu-
tion corresponding to Au/TiO samples were obtained in a Philips-CM10
operated a 100 kV. Chemical analyses of gold in the catalysts were car-
ried out after dissolving the solids by attack with a 2:1 mixture of HNO
2
(16.7 nm mean diameter): The catalyst was prepared at pH 4.5
high and general activity of Au/npCeO even in organic sol-
2
2
vents, as we have shown here, and is in agreement with the
reaction mechanism proposed.
2
samples were examined
(
2
Experimental Section
3
/
HF on a Varian-10 Plus Atomic Absorption Spectrometer or directly of
the solids using a Philips Minipal 25 fm Analytic X-Ray apparatus and a
calibration plot.
Preparation of Au/npCeO
Synthesis of nanoparticulated ceria: The preparation of nanoparticulated
2
[
51]
ceria was carried out following a reported procedure.
aqueous solution of Ce(NO (375 mL, 0.8m) was treated, under stirring
and at ambient temperature, with an aqueous solution of ammonia
1.12 L, 0.8m). The colloidal dispersion of CeO nanoparticles was heated
In short, an
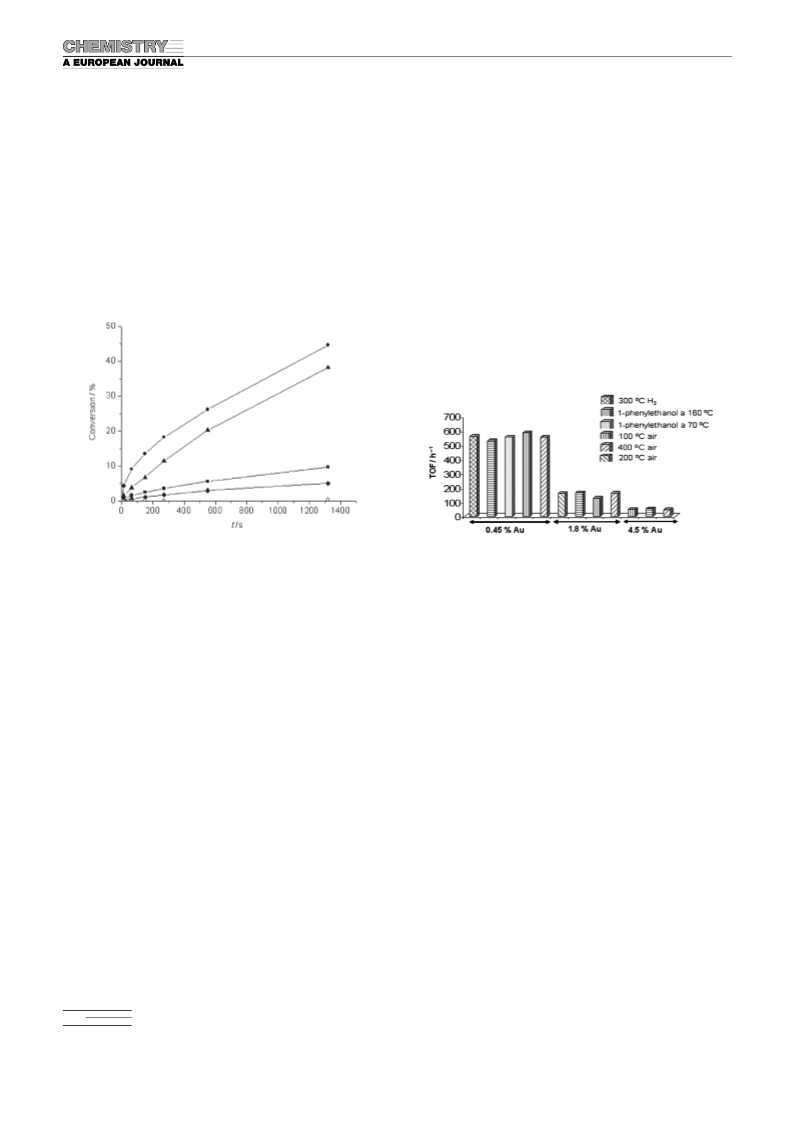
Typical procedure for the aerobic oxidations of alcohols: A suspension of
A
C
H
T
R
E
U
N
G
3 4
)
0
2
.44 wt% Au/npCeO (0.2 mol%) in toluene (5 mL) was treated with
benzyl alcohol (1 mmol). The resulting mixture was then heated at 908C
for 1 h and benzaldehyde was produced in >99% GC yield. After the re-
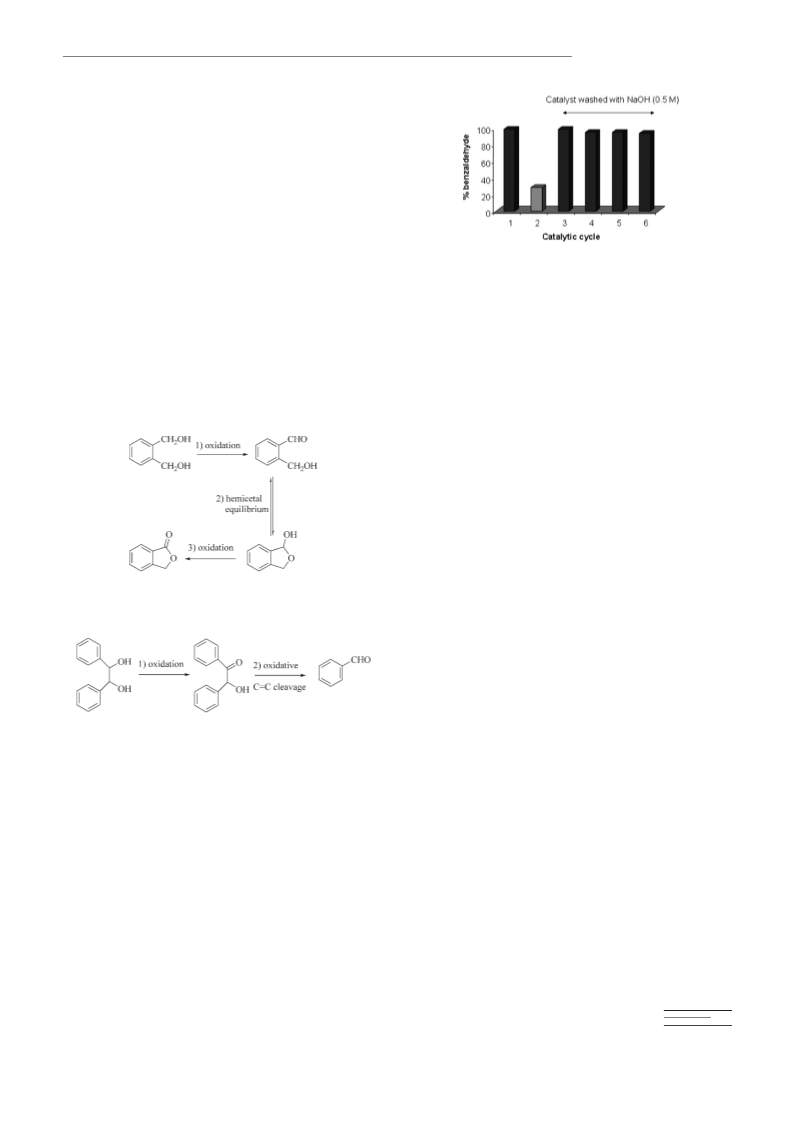
action, the catalyst was separated by filtration and washed with acetone.
The recovered Au/npCeO was washed with an aqueous solution of
2
NaOH (0.5m) and water (100 mL) and dried under vacuum before recy-
(
2
in a PET vessel at 1008C for 24 h. The resulting yellow precipitate was
filtered and dried under vacuum overnight. The cerium oxide synthesised
has, owing to the small size of the nanoparticles, a very high surface area
2
ꢀ1
(
180 m g ).
Au was deposited on the nanoparticulated ceria by using the following
procedure: A solution of HAuCl ·3H O (800 mg) in deionised water
160 mL) was brought to pH 10 by addition of a solution of NaOH 0.2m.
Once the pH value was stable the solution was added to a gel containing
colloidal CeO (4.01 g) in H O (50 mL). After adjusting the pH of the
slurry at a value of 10 by addition of a 0.2m solution of NaOH 0.2m, the
slurry was continuously stirred vigorously for 18 h at RT. The Au/npCeO
solid was then filtered and exhaustively washed with several litres of dis-
tilled water until no traces of chlorides were detected by the AgNO test.
The catalyst was dried under vacuum at room temperature for 1 h. Then
cling.
4
2
(
Acknowledgement
2
2
Financial support by the Spanish DGI (CTQ06–6857) is gratefully ac-
knowledged. A.A. thanks to the Spanish Ministry of Education for a
postgraduate scholarship.
2
3
1
1
-phenylethanol (30 g) was treated with the supported catalyst (3.5 g) at
608C and the mixture was allowed to react for 20 min. The catalyst was
[
[
[
[
filtered, washed with acetone and water, and dried under vacuum at RT.
The total Au content of the final catalyst Au/npCeO was 4.5 wt% as de-
termined by chemical analysis. For the preparation of 1.8 and 0.45 wt%
Au/npCeO the method used was the same as above, but changing only
the amount of HAuCl ·3H O aqueous. This catalyst Au/npCeO is com-
4] C. Gonzµlez-Arellano, A. Abad, A. Corma, H. García, M. Iglesia, F.
Sµnchez, Angew. Chem. 2007, 119, 1558; Angew. Chem. Int. Ed.
2
2
4
2
2
mercially available from Instituto de Tecnología Química (ITQ) (http://
www.upv.es/itq).
2
007, 46, 1536.
Preparation of 1 wt% Au/C catalyst: A colloidal solution of gold nano-
particles stabilized by polyvinyl alcohol were deposited on activated
carbon (KB-B-100, provided by Aldrich), following the procedure report-
[
52]
ed by Porta et al.
HAuCl (2 L, 100 mgmL ) was treated with an aqueous solution of poly-
vinyl alcohol (PVA) (10 mL, 27 mgmL ). To this, a fresh solution of
NaBH (38 mL, 0.1m) was added. The AuNPs generated were immobi-
lised simply by adding the active carbon (2 g) into the metal dispersion.
Under vigorous stirring an aqueous solution of
ꢀ
1
4
ꢀ
1
4
Chem. Eur. J. 2008, 14, 212 – 222
ꢀ 2008 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
221

 Abad, Alberto
Abad, Alberto
 Corma, Avelino
Corma, Avelino
 Garcia, Hermenegildo
Garcia, Hermenegildo