European Journal of Organic Chemistry
10.1002/ejoc.201701314
FULL PAPER
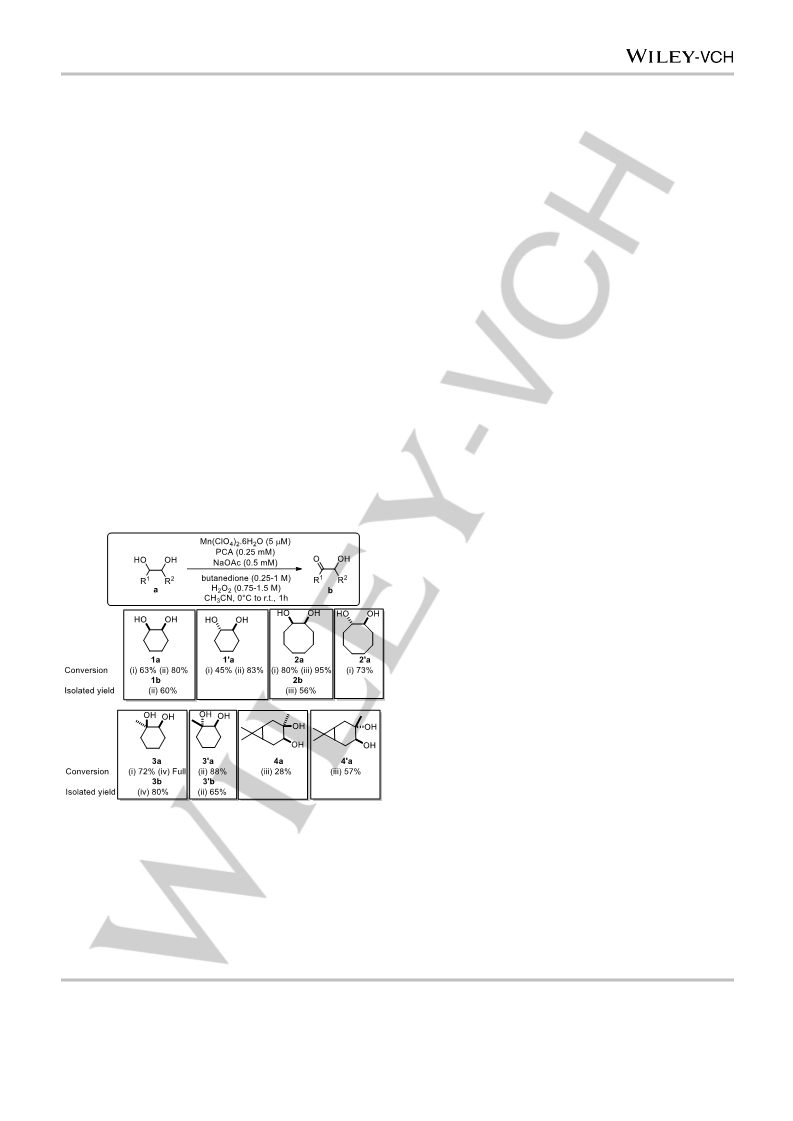
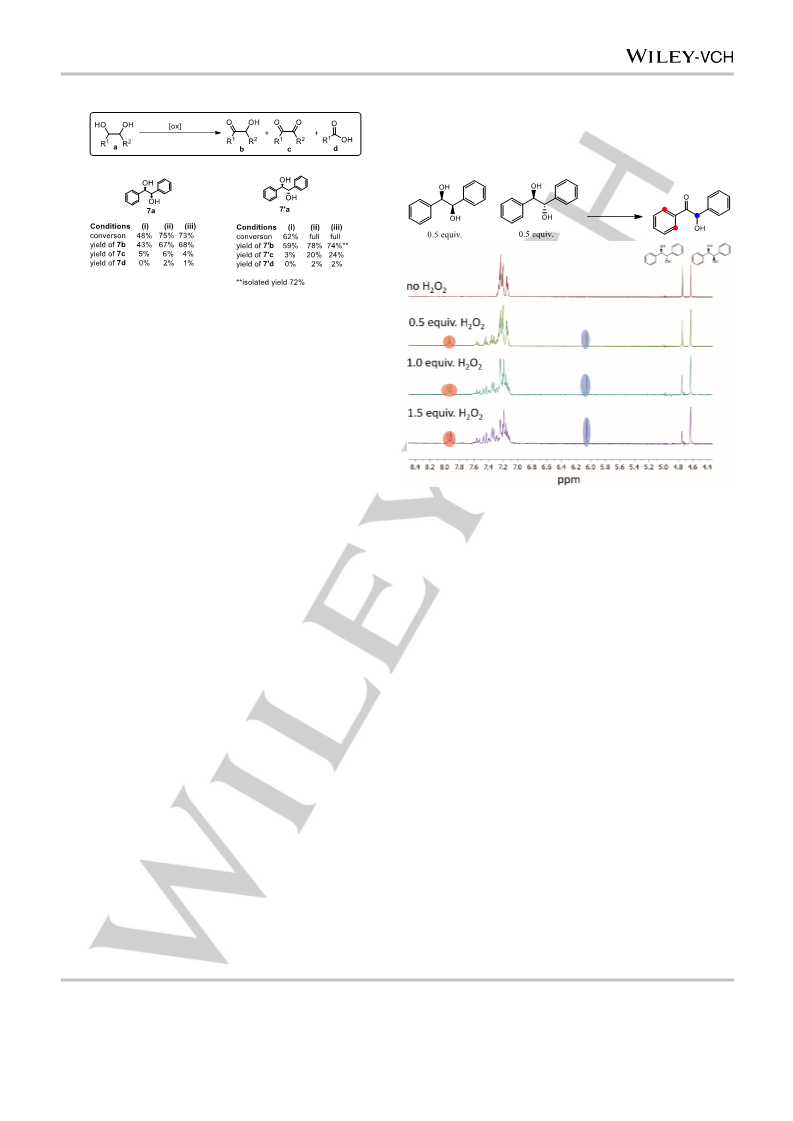
the oxidation of diols to their corresponding α-hydroxyketones.
The method shows selectivity for benzylic and secondary
positions over primary positions in 1,2-diols as expected
considering earlier studies in the oxidation of alcohols. It is
notable that although further oxidation to the diketone and
cleavage products is observed in some cases the primary
product is generally the acyloin. Taken together with the stability
of substrates bearing groups sensitive to rearrangements upon
formation of carbocation and radical intermediates, the data
spectroscopy focused primarily on the intensity of the C=C and C=O
stretching bands between 1550 and 1800 cm (e.g., at 1724 cm−1 for
−1
butanedione), between 600 and 900 cm− relating to the C=C and C=O
1
bending modes (682 cm−1 for butanedione), and the O−O stretching
2 2
mode of H O .
at 870 cm−
1 [27]
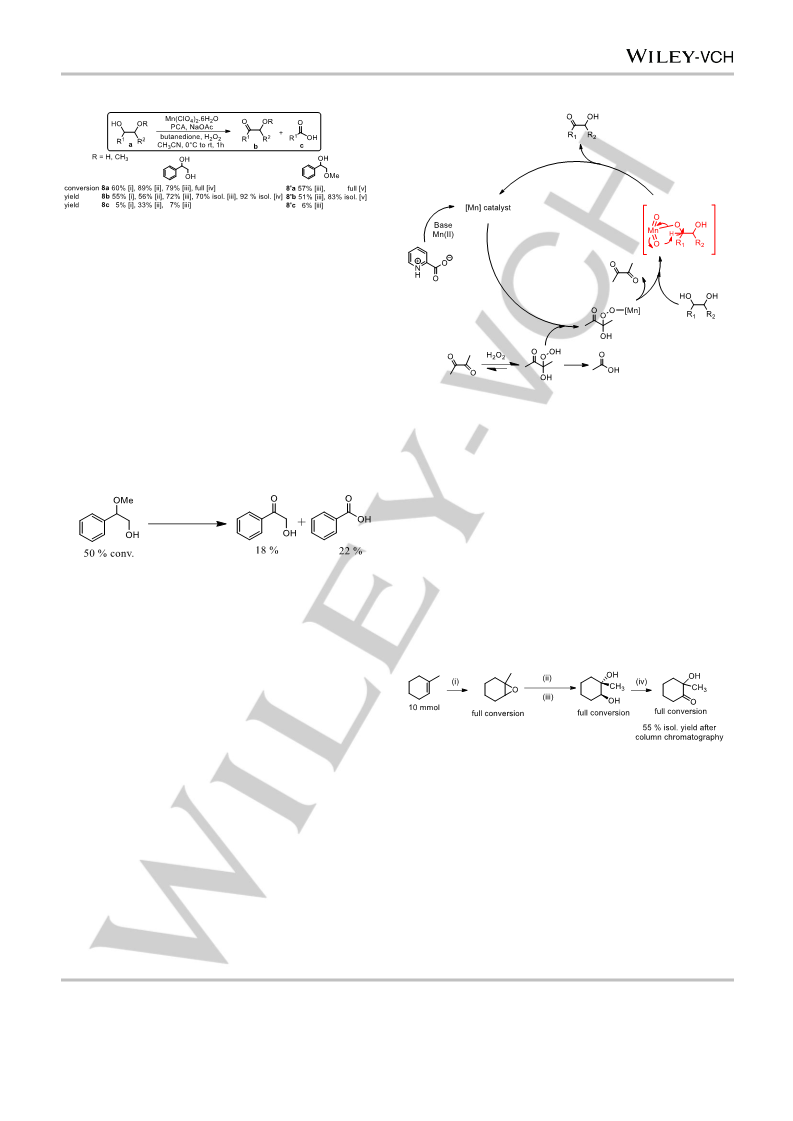
Oxidation of 1-methyl-cyclohex-1-ene to 2-methyl-2-hydroxy-
cyclohexanone in acetone. 1-methyl-cyclohex-1-ene (10 mmol, 960
mg) was added to a 5.0 mL of acetone with Mn(ClO ) ·6H O (5 M), PCA
4 2 2
(0.25 mM). 0.167 mL NaOAc (aqueous, 0.6 M) and 0.435 mL
butanedione (5 mmol) were added to give a final volume of 20 mL and a
final concentration of the substrate of 0.5 M. The mixture was cooled in
2 2
an ice bath and H O (170 µL, 15 mmol) was added dropwise while
stirring. The reaction is allowed to reach r.t. Conversion was verified after
imply
a
selective oxidising species (e.g.
a
high valent
st
manganese complex). The use of a 1 row transition metal
based (non-scarce/toxic) catalysts with
H
O
2 2
under mild
conditions opens a new and versatile route towards an equally
versatile class of intermediates.
1
3
0
0 min by H NMR spectroscopy reaction after which Fe(ClO
4
)
3
(35.4 mg,
.01 mol %) was added and the reaction mixture was stirred overnight at
room temperature. Conversion was verified by 1H NMR spectroscopy
followed by aqueous workup (brine/CHCl ) of the reaction mixture. The
layers were separated and the aqueous layer extracted with CHCl (2 x
0 mL). The combined organic layers were dried over anhydrous MgSO
3
Experimental Section
3
1
4
and the solvent was evaporated in vacuo. The crude product was
submitted to the same reaction conditions as in the first step, except 30
All reagents were of commercial grade and were used as received unless
stated otherwise. Hydrogen peroxide was used as received (Acros
mmol of H
after 30 min was verified by 1H NMR spectroscopy and mixture was
added to 100 mL CHCl and 100 mL saturated aqueous NaCl. The layers
were separated and the aqueous layer extracted with CHCl (2 x 10 mL).
The combined organic layers were dried over anhydrous MgSO and
filtered, and the solvent evaporated in vacuo. The crude product was
2 2
O was added to ensure complete conversion. Conversion
Chemicals) as a 50 wt% solution in water; note that the grade of H
2 2
O
employed can affect the outcome of the reaction; lower conversion is
3
observed especially in the case of
2 2
H O solutions stabilized with
1
13
3
sequestrants. H NMR (400.0 MHz) and C NMR (100.59 MHz) spectra
were recorded on a Varian Avance 400. Chemical shifts are relative to
4
the following: 1H NMR, CDCl
CN (1.94 ppm); 13
3
(7.26 ppm) and CD
3
C
purified by column chromatography (Silica gel, Pentane : Et
give an isolated yield of 55 % (670 mg).
2
O = 8:2) to
NMR, CDCl (77 ppm) and CD CN (118 ppm). Raman spectra were
3
3
recorded at 785 nm with a RamanFlex (Perkin Elmer) spectrometer
equipped with a fibre optic probe (Inphotonics).
Caution! The drying or concentration of solutions that potentially contain
H
2
O
2
should be avoided. Prior to drying or concentrating, the presence of
should be tested for using peroxide test strips followed by
H
2
O
2
Acknowledgements
neutralization on solid NaHSO
3
or another suitable reducing agent. When
working with H , suitable protective safeguards should be in place at
all times due to the risk of explosion.
2
O
2
The COST association action CM1305 ECOSTBio is
acknowledged for discussion and the European Research
Council (ERC 279549, WRB) is acknowledged for financial
support.
Caution! Although used as a food additive, butanedione has been
implicated in certain lung diseases upon prolonged exposure to its
vapours. It should be handled in a properly ventilated fume hood, and
exposure to vapours should be avoided.
Keywords: Oxidation • manganese • hydroxyl ketone • catalysis
•
hydrogen peroxide
Typical procedure for catalytic oxidations. The substrate (1 mmol)
was added to
4 2 2
a solution containing Mn(ClO ) ·6H O and PCA in
acetonitrile. NaOAc (aqueous, 0.6 M), butanedione (0.5 mmol) and
acetonitrile (amount depending on that of the substrate) were added to
give a final volume of 2 mL and a final concentration of the substrate of
[1]
K. T. Finley, Chem. Rev. 1964, 64, 573–589.
[
[
2]
3]
J. C. Tarr, J. S. Johnson, J. Org. Chem. 2010, 75, 3317–3325.
M. Feurer, G. Frey, H.-T. Luu, D. Kratzert, J. Streuff, Chem.
Commun. 2014, 50, 5370–5372.
0
.5 M (unless stated otherwise). The solution was stirred in an ice/water
bath before addition of H (50 wt%). Reaction progress was monitored
in situ by Raman spectroscopy.
spectroscopy by dilution of a part of the reaction mixture in CD
2 2
O
[
27]
Conversion was verified by 1H NMR
CN.
[
4]
M. Y. Jin, S. M. Kim, H. Han, D. H. Ryu, J. W. Yang, Org. Lett. 2011,
13, 880–883.
3
Spectra were assigned by comparison with authentic samples. Product
isolation typically involved addition of brine (10 mL) and extraction with
dichloromethane (3 x 10 mL). The combined organic layers were washed
[
[
5]
6]
B. A. C. P. Zhou, in Asymmetric Oxid. React., 2001, p. 128.
B.-C. C. F. A. Davis, in Stereoselective Synth., 1996, pp. 4497–
with brine, dried over Na
dichloromethane removed in vacuo. In the case of hydrobenzoin(s), the
diketone product is selectively removed after evaporation of CH Cl , by
washing the resulting powder with small amounts of Et O to remove the
2 4
SO (anhydrous), and filtered, and the
4518.
[
[
[
7]
8]
9]
G. J. Chuang, W. Wang, E. Lee, T. Ritter, J. Am. Chem. Soc. 2011,
133, 1760–1762.
2
2
2
yellow byproduct. 1,2-Dichlorobenzene, which has a negligible effect on
the reaction, was employed as internal standard for Raman and 1H NMR
spectroscopy where necessary. Reaction monitoring with Raman
O. A. Hamed, A. El-Qisairi, H. Qaseer, E. M. Hamed, P. M. Henry, D.
P. Becker, Tetrahedron Lett. 2012, 53, 2699–2701.
A. K. El-Qisairi, H. A. Qaseer, J. Organomet. Chem. 2002, 659, 50–
This article is protected by copyright. All rights reserved.

 Mecozzi, Francesco
Mecozzi, Francesco
 Dong, Jia Jia
Dong, Jia Jia
 Saisaha, Pattama
Saisaha, Pattama
 Browne, Wesley R.
Browne, Wesley R.