Communication
ChemComm
Table 3 The hydrocarboxylation reaction of different alkene substrates
on Rh1/POPs catalysta
This research work was supported by the National Key R&D
Program of China (2017YFB0602203).
Entry
Substrate
Product
Yieldb (%)
Conflicts of interest
1
2
80
98
There are no conflicts to declare.
3
72c
82d
Notes and references
1 (a) R. Sang, P. Kucmierczyk, R. Du¨hren, R. Razzaq, K. Dong, J. Liu,
R. Franke, R. Jackstell and M. Beller, Angew. Chem., Int. Ed., 2019, 58,
14365–14373; (b) R. Matthessen, J. Fransaer, K. Binnemans and
D. E. De Vos, Beilstein J. Org. Chem., 2014, 10, 2484–2500; (c) Organic
Acids Market by Type, Application, and Region – Global Forecast to
2022, Report Code FB 4344, Markets and Markets, 2017; (d) M. T.
Sabatini, L. T. Boulton and T. D. Sheppard, Sci. Adv., 2017,
3, e1701028.
2 (a) H. J. Adamo, P. Franke, H. Jenkel, R. Kern, K. Schmidt and
H. J. Woehling, Germany Pat., DD159326A1, 1983; (b) C. Li, L. Yan,
L. Lu, K. Xiong, W. Wang, M. Jiang, J. Liu, X. Song, Z. Zhan, Z. Jiang
and Y. Ding, Green Chem., 2016, 18, 2995–3005; (c) L. Vanoye,
4
a
Reaction conditions: Rh1/POPs (0.15 g, Rh content = 1.0 wt%),
p-TsOHÁH2O (1.0 mmol), acetic acid (100 mmol), substrate (5.7 mmol),
CH3I (2.77 mmol) and ultra-pure water (30 mmol), 1.0 MPa (CO at room
b
temperature), 180 1C, 16 h. Yield was determined by GC analysis.
c
d
1-COOH: 75.2%, 2-COOH: 24.8%. 1-COOH: 49.9%, 2-COOH: 35.5%,
3-COOH: 14.6%.
the spent catalyst exited as PIPs, consisting of a monomer of
quaternary phosphonium salt.
´
A. Aloui, M. Pablos, R. Philippe, A. Percheron, A. Favre-Reguillon
and C. de Bellefon, Org. Lett., 2013, 15, 5978–5981; (d) R. W. Johnson
and E. Fritz, in Fatty acids in industry, ed. M. Dekker, New York,
1989; (e) W. Ren, J. Chu, F. Sun and Y. Shi, Org. Lett., 2019, 21,
5967–5970.
3 (a) W. Reppe, Justus Liebigs Ann. Chem., 1953, 582, 1–37; (b) G. Kiss,
Chem. Rev., 2001, 101, 3435–3456.
4 K. Bittler, N. V. Kutepow, D. Neubauer and H. Reis, Angew. Chem.,
Int. Ed. Engl., 1968, 7, 329–335.
5 A. Y. Putin, E. Y. Bychkova, K. V. Trunilina, A. Y. Zakgeim,
E. A. Katsman, O. N. Shishilov, N. S. Akhmadullina,
I. V. Oshanina, O. N. Temkin and L. G. Bruk, Russ. J. Phys. Chem.
A, 2015, 89, 359–368.
6 (a) H. Alper, J. B. Woell, B. Despeyroux and D. J. H. Smith, J. Chem.
Soc., Chem. Commun., 1983, 1270–1271; (b) M. Karlsson, A. Ionescu
and C. Andersson, J. Mol. Catal. A: Chem., 2006, 259, 231–237;
(c) Z. Huang, Y. Cheng, X. Chen, H.-F. Wang, C.-X. Du and Y. Li,
Chem. Commun., 2018, 54, 3967–3970.
7 Z. He, Z. Hou, Y. Zhang, T. Wang, Y. Dilixiati and W. Eli, Catal.
Today, 2015, 247, 147–154.
The hydrophilicity of the support is a vital factor in a
hydrocarboxylation reaction because the generation of the
desired carboxylic acids needs the participation of water. Water
contact angle (WCA) measurements were carried out on POPs
and PIPs, a quaternization product of POPs by CH3I in acetic
acid (Fig. S6a and b, ESI†), to determine the hydrophobicity or
hydrophilicity. The WCA for POPs and PIPs is 119.891 and
14.351, respectively, indicating that POPs are a relatively hydro-
phobic material but PIPs are a significantly hydrophilic one
(Fig. S6c and d, ESI†).16 The water adsorption results indicate
that the PIPs possess a water adsorption ratio of 1446%, far
more than 540% of POPs (Table S2, ESI†). Thus, the in situ
transformation from POPs to PIPs is favourable for adsorbing
water to participate in the reaction.
8 (a) I. W. Davies, L. Matty, D. L. Hughes and P. J. Reider, J. Am. Chem.
Soc., 2001, 123, 10139–10140; (b) S. Jayasree, A. Seayad and
R. V. Chaudhari, Chem. Commun., 1999, 1067–1068; (c) A. Aghmiz,
The general applicability of the Rh1/POPs catalyst for other
olefins in hydrocarboxylation was investigated with many sub-
strates. The results are listed in Table 3. Besides cyclohexene,
cyclopentene was also tested, obtaining a relatively better yield
of the corresponding carboxylic acid (Table 3, entry 1). In
´
´
´
M. Gimenez-Pedros, A. M. Masdeu-Bulto and F. P. Schmidtchen,
Catal. Lett., 2005, 103, 191–193.
9 T. G. Ostapowicz, M. Schmitz, M. Krystof, J. Klankermayer and
W. Leitner, Angew. Chem., Int. Ed., 2013, 52, 12119–12123.
addition, linear terminal alkenes, such as ethylene, 1-hexene 10 T. Oku, M. Okada, M. Puripat, M. Hatanaka, K. Morokuma and
J.-C. Choi, J. CO2 Util., 2018, 25, 1–5.
11 (a) Q. Sun, M. Jiang, Z. Shen, Y. Jin, S. Pan, L. Wang, X. Meng,
and 1-octene were converted into target products successfully
(Table 3, entries 2–4). The yields are 98%, 72% and 82%,
W. Chen, Y. Ding, J. Li and F.-S. Xiao, Chem. Commun., 2014, 50,
respectively. Thus, the Rh1/POPs can be widely applied in
various olefin substrates.
11844–11847; (b) R. Tao, X. Ma, X. Wei, Y. Jin, L. Qiu and W. Zhang,
J. Mater. Chem. A, 2020, 8, 17360–17391.
12 Z. Ren, Y. Lyu, X. Song, Y. Liu, Z. Jiang, R. Lin and Y. Ding,
Adv. Mater., 2019, 31, 1904976.
In summary, atomic dispersed Rh on porous organic poly-
mers was firstly applied in heterogeneous hydrocarboxylation 13 (a) M. Jiang, L. Yan, Y. Ding, Q. Sun, J. Liu, H. Zhu, R. Lin, F. Xiao,
Z. Jiang and J. Liu, J. Mol. Catal. A: Chem., 2015, 404–405, 211–217;
(b) Z. Ren, Y. Liu, Y. Lyu, X. Song, C. Zheng, S. Feng, Z. Jiang and
Y. Ding, J. Catal., 2019, 369, 249–256; (c) W.-Y. Huang, G.-Q. Wang,
of olefins to produce carboxylic acids with one more carbon.
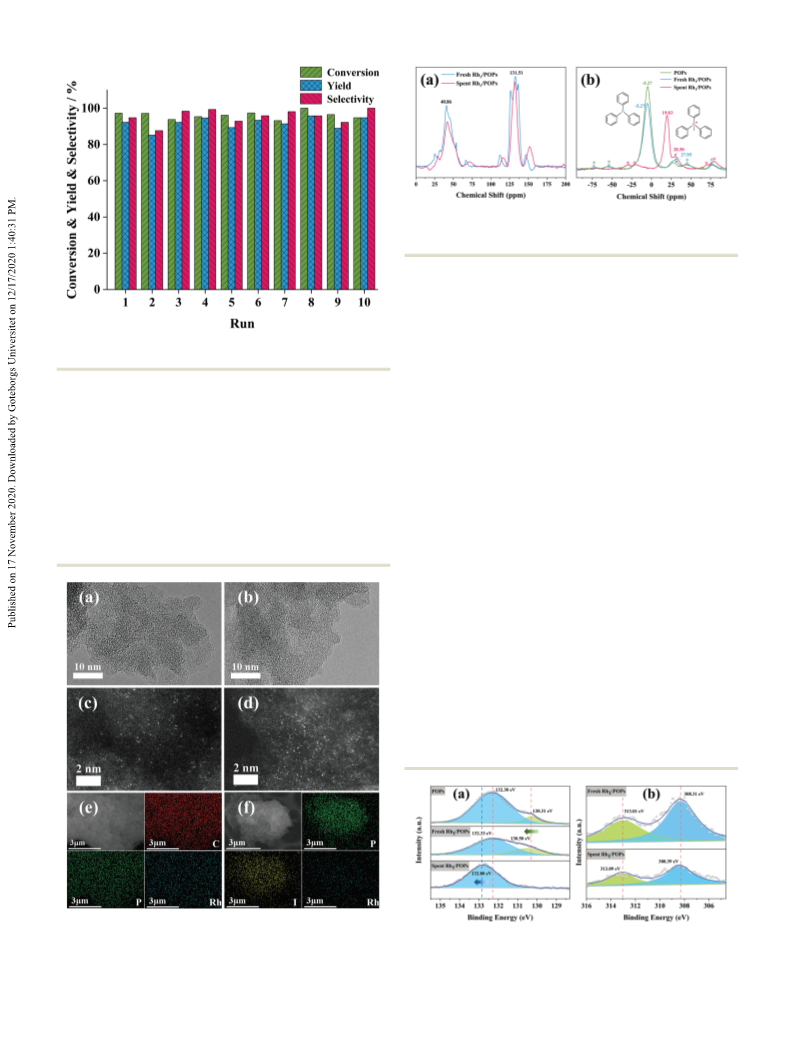
The cyclohexene substrate was selected to optimize the reaction
conditions, and also to investigate the stability of the single-Rh-
site catalyst. An in situ transformation from POPs to PIPs
resulted in an excellent performance, attaining 97% conversion
W.-H. Li, T.-T. Li, G.-J. Ji, S.-C. Ren, M. Jiang, L. Yan, H.-T. Tang,
Y.-M. Pan and Y.-J. Ding, Chemistry, 2020, 6, 2300–2313.
14 X. Lan, W. Zhang, L. Yan, Y. Ding, X. Han, L. Lin and X. Bao, J. Phys.
Chem. C, 2009, 113, 6589–6595.
for cyclohexene and 92% yield of cyclohexanecarboxylic acid for 15 (a) S. Hoste, D. F. Van De Vondel and G. P. Van Der Kelen, J. Electron
Spectrosc. Relat. Phenom., 1979, 17, 191–195; (b) L. S. Dake, D. R. Baer
and D. M. Friedrich, J. Vac. Sci. Technol., A, 1989, 7, 1634–1638.
16 W. A. Herrmann and C. W. Kohlpaintner, Angew. Chem., Int. Ed.
ten times recycling without measurable loss of activity. Further-
more, the universal protocol can be applied in heterogeneous
hydrocarboxylation of various olefins.
Engl., 1993, 32, 1524–1544.
Chem. Commun.
This journal is © The Royal Society of Chemistry 2020

 Yuan, Qiao
Yuan, Qiao
 Song, Xiangen
Song, Xiangen
 Feng, Siquan
Feng, Siquan
 Jiang, Miao
Jiang, Miao
 Yan, Li
Yan, Li
 Li, Jingwei
Li, Jingwei
 Ding, Yunjie
Ding, Yunjie