Aliphatic Aldehyde Reductase Activity in Vietnamese Coriander Leaves
647
Comparison of volatile constituents of Persicaria odorata
(Lour.) Sojak (Polygonum odoratum Lour.) and Persicaria
hydropiper L. Spach (Polygonum hydropiper L.). J. Agric. Food
Chem., 54, 3067–3071 (2006).
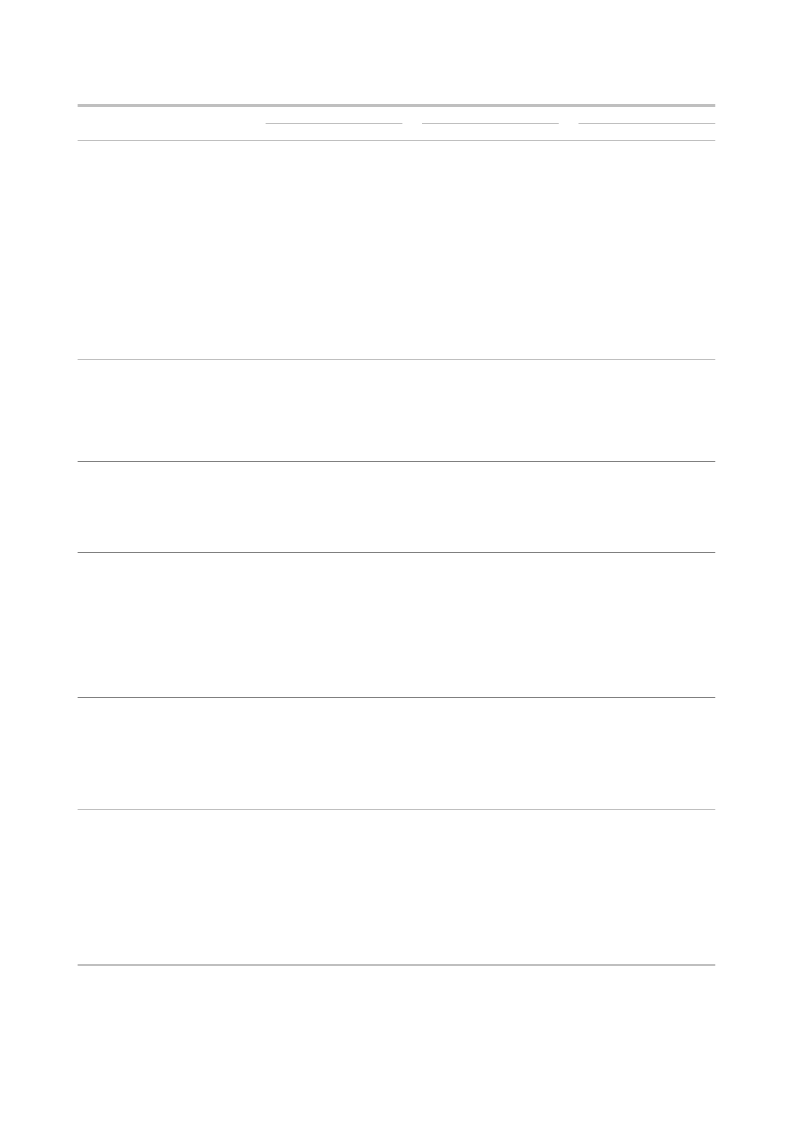
suggestion is supported by the strong activity of this
enzyme in either the homogenized mixture (Fig. 1A and
B) or in the crude enzyme solution (Fig. 2A), in which
the content of heptanol formed with the addition of
NADH as the coenzyme was much more than twice that
of NADPH (Fig. 3A and B).
5) Benitez, D. E., Cadwallader, K. R., and Suriyaphan, O.,
‘‘Aroma-Active Components of Vietnamese Coriander (Poly-
gonum odoratum),’’ IFT Annual Meeting, Louisiana, New
Orlands (2001).
6) Schreier, P., Chromatographic studies of biogenesis of plant
volatiles. In ‘‘Chromatographic Methods,’’ eds. Bertsch, W.,
Effect of inhibitors
The crude enzyme from Vietnamese coriander leaves
and NADH or NADPH were incubated for 2 min prior to
adding the substrate to test their inhibitory effect with
1,10-phenanthroline, a potent inhibitor of alcohol de-
hydrogenase, and p-hydroxymercuribenzoate (Table 2).
Assays were conducted at 25 ꢀC.
Jennings, W. G., and Kaiser, R. E., Huthig, Heidelberg, pp. 72–
¨
74 (1984).
7) Smallfield, B. M., Perry, N. B., Beauregard, D. A., Foster,
L. M., and Dodds, K. G., Effect of postharvest treatments on
yield and composition of coriander herb oil. J. Agric. Food
Chem., 42, 354–359 (1994).
8) Sekiwa, Y., Mizuno, Y., Yamamoto, Y., Kubota, K., Kobayashi,
A., and Koshino, H., Isolation of some glucosides as aroma
precusors from ginger. Biosci. Biotechnol. Biochem., 63, 384–
389 (1999).
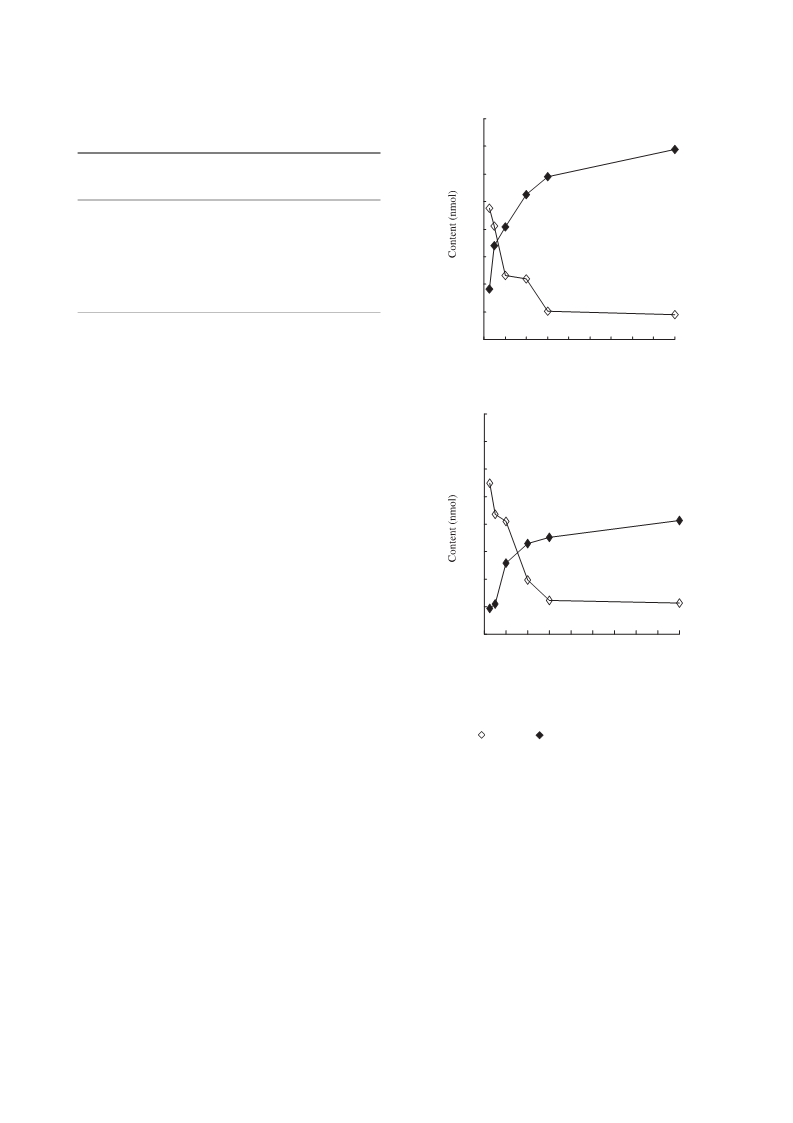
The results show that 1,10-phenanthroline with NADH
or NADPH at 5 mM caused 16% or 25% inhibition,
respectively. On the other hand, the enzyme activity with
the addition of either NADH or NADPH was inhibited
completely by p-hydroxymercuribenzoate. Our findings
agree well with those of Negm, F.B.,21) who have
reported p-hydroxymercuribenzoate was a strong inhib-
itor of aldehyde reductase from Euonymus leaves. These
results also distinguish the Vietnamese coriander enzyme
from aliphatic alcohol dehydrogenase and help to
confirm the presence of aliphatic aldehyde reductase
in this plant. This is the first report of aliphatic
aldehyde reductase activity being detected in Vietnamese
coriander leaves.
It is concluded from these results that the aliphatic
aldehyde reductase activity would have influenced the
formation of alcohols related to corresponding alde-
hydes in Vietnamese coriander. Aliphatic aldehyde
dehydrogenase, which is responsible for the acidic form,
was also found in this herb with weaker activity than
that of reductase. This implies that the expression and
strength of the aliphatic aldehyde reductase activity are
strongly correlated with the flavor quality of this herb.
9) Guth, H., and Grosch, W., Identification of potent odourants in
static headspace samples of green and black tea powders on the
basis of aroma extract dilution analysis (AEDA). Flavour
Fragrance J., 8, 173–178 (1993).
10) Iijima, Y., Aizawa, Y., and Kubota, K., Geraniol dehydrogenase
activity related to aroma formation in ginger (Zingiber officinale
Roscoe). J. Agric. Food Chem., 49, 5902–5906 (2001).
11) Van der Plas, L. H. W., and Van der Pal, R. H. M., Induction of
alcohol dehydrogenase in explants of potato tuber (Solanum
tuberosum L.). Plant Cell Rep., 2, 40–42 (1983).
12) Brzezinski, R., Talbot, B. G., Brown, D., Klimuszko, D.,
Blakeley, S. D., and Thirion, J. P., Characterization of alcohol
dehydrogenase in young soybean seedlings. Biochem. Genet.,
24, 9–10 (1986).
13) Iijima, Y., Guodong, W., Eyal, F., and Eran, P., Analysis of the
enzymatic formation of citral in the glands of sweet basil.
Biochem. Biophys., 448, 141–149 (2006).
14) Tulsiani, D. R. P., and Touster, O., Resolution and partial
characterization of two aldehyde reductase of mammalian liver.
J. Biol. Chem., 252, 2545–2550 (1977).
15) Wermuth, B., Munch, J. D. B., and Pierre, J. V. W., Purification
and properties of NADPH-dependent aldehyde reductase from
human liver. J. Biol. Chem., 252, 3821–3828 (1977).
16) Davidson, W. S., and Flynn, G., A functional arginine residue in
NADPH-dependent aldehyde reductase from pig kidney. J. Biol.
Chem., 254, 3724–3729 (1979).
Acknowledgments
17) Felsted, R., Richter, D. R., and Bachur, N. R., Rat liver aldehyde
reductase. Biochem. Pharmacol., 26, 1117–1124 (1977).
18) Colrat, S., Latche, A., Guis, M., Pech, J. C., Bouzayen, M.,
Fallot, J., and Roustan, J. P., Purification and characterization of
an NADPH-dependent aldehyde reductase from mung bean that
detoxifies eutypine, a toxin from Eutypa lata. Plant Physiol.,
119, 621–626 (1999).
This work was financially supported in part by a
Sasagawa Scientific Research grant from The Japan
Science Society.
References
1) Dung, N. X., Hac, L. V., and Leclercq, P. A., Volatile
constituents of the aerial parts of Vietnamese Polygonum
odoratum Lour. J. Essent. Oil Res., 7, 339–340 (1995).
19) Oberschall, A., Deak, M., Torok, K., Sass, L., Vass, I., Kovacs,
I., Feher, A., Dudits, D., and Horvath, G. V., A novel aldose/
aldehyde reductase protects transgenic plants against lipid
peroxidation under chemical and drought stresses. Plant J.,
24, 437–446 (2000).
2) Hunter, M. V., Brophy, J. J., Ralph, B. J., and Bienvenu, F. E.,
Composition of Polygonum odoratum Lour. from Southern
Australia. J. Essent. Oil Res., 9, 603–604 (1997).
20) Brown, H. M., and Purves, W. K., Isolation and characterization
of Indole-3-acetaldehyde reductase from Cucumis sativus. J.
Biol. Chem., 251, 907–913 (1976).
3) Cadwallader, K. R., Benitez, D., Pojjanapimol, S., Suriyaphan,
O., and Singh, T., ‘‘Characteristic Aroma Components of the
Cilantro Mimics,’’ A.C.S symposium series Vol. 908, American
Chemical Society, Washington, DC, pp. 117–128 (2005).
4) Christian, S., Ludmila, L., Yvan, N., Eric, P., and Didier, R.,
21) Negm, F. B., Purification and properties of an NADPH-aldose
reductase (aldehyde reductase) from Euonymus japonica leaves.
Plant Physiol., 80, 972–977 (1986).

 Quynh, Cung Thi To
Quynh, Cung Thi To
 Iijima, Yoko
Iijima, Yoko
 Morimitsu, Yasujiro
Morimitsu, Yasujiro
 Kubota, Kikue
Kubota, Kikue