34
J.S. Santos et al. / Applied Catalysis A: General 443–444 (2012) 33–39
conditions. Caetano et al. [17] studied the esterification of palmitic
acid with methanol using H3PW, H3PMo and H4SiW immobilized
by sol–gel technique on silica, at 60 ◦C. Xu et al. [18] prepared a
mesoporous composite catalyst of H3PW/Ta2O5, and applied it as a
solid acid catalyst for the esterification reaction of lauric acid with
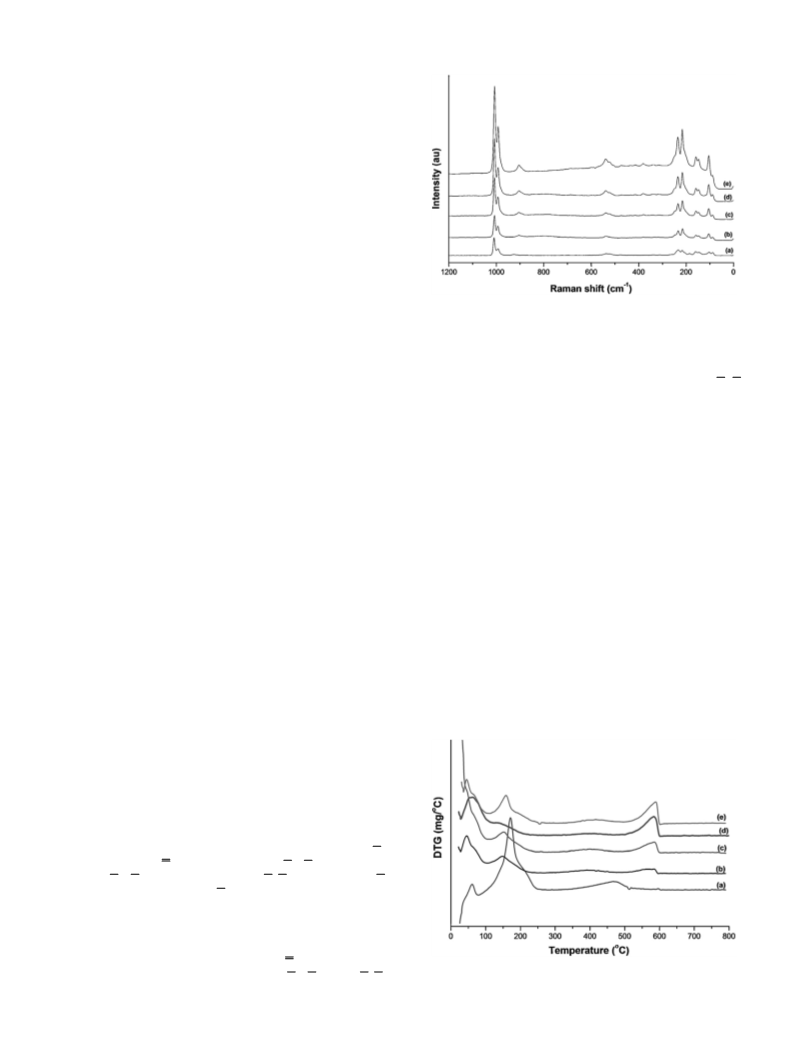
2.2. Characterization of the catalysts
Infrared spectra of the mixed salts and the pyridine-adsorbed
POMs were obtained with a Thermo Scientific Nicolet spectrometer
model 6700 FTIR with 128 scans and a spectral resolution of 4 cm−1
Each sample was pressed in dried 1-wt% KBr (Merck) pellets.
.
FT-Raman spectra were recorded on a Bruker FRA 106/S
module attached to Bruker Equinox 55 spectrometer
Novel mixed salts of cesium and ammonium derivatives of
a
H3PW ((NH4)xCs2.5−xH0.5PW12O40
,
abbreviated thereinafter as
(128 scans and 4 cm−1 resolution). The laser excitation
(Nd:YAG = neodymium:yttrium aluminum garnet) and laser
power were 1064 nm and 250 mW, respectively, and the signal
was detected by a liquid N2-cooled Ge detector. Samples without
any thermal treatment were packed into sample cups for analysis.
For liquid samples (reaction products), a 1-cm quartz cuvette with
a mirror surface toward the scattering direction was used.
(NH4)xCs2.5−xH0.5PW) have recently been synthesized and char-
acterized [19]. During the synthesis, the primary nanoparticles
aggregated, forming structures with mesopores and micropores.
The pores were created by partial unidirectional growth of the
nanocrystals during the formation of larger, round microcrystal-
lites, leading to materials with higher surface area and lower
solubility than the parent acid. These properties are important for
surface-type catalysis and liquid phase-separation purposes.
The goal of this work is to study the thermal stability and acidity
of these mixed salts of ammonium and cesium derivatives of H3PW
and to apply them to the esterification reaction with ethanol. The
mixed salts were characterized by FT-Raman to confirm their struc-
tures and their stability was evaluated by calcination followed by
FTIR and thermal analysis. The nature of the acid sites was accessed
by adsorption of gaseous pyridine detected by FTIR. The amount
and strength of the sites were calculated by desorption of pyridine,
measured by thermal analysis. The catalytic activities of the mixed
salts were tested on the model esterification reaction of oleic acid
with ethanol.
Thermal analysis consisted of thermogravimetric (TGA), deriva-
tive thermogravimetric (DTG) and differential thermal analysis
(DTA). They were obtained in a simultaneous TG-DSC (TA Instru-
ments, model SDT 2960), with a heating rate of 10 ◦C min−1, from
room temperature (∼25 ◦C) up to 800 ◦C under a synthetic air
(80.0 0.5% N2 and 20.0 0.5% O2) flow of 110 mL min−1
.
2.3. Thermal activation of the catalysts
After preparation, the catalysts underwent different heating
treatments for the thermal stability studies: 200, 300, 450 and
650 ◦C for 4 h in a muffle furnace (EDG model EDG3PS) under static
air conditions at a heating rate of 10 ◦C min−1. Before the catalytic
tests, the materials were activated at 200 ◦C for 4 h in the same
furnace.
2. Experimental
2.4. Gas phase adsorption of pyridine
2.1. Preparation of the ammonium and cesium mixed salts of
H3PW
Fresh pyridine (Vetec) was purified by distillation over CaH2
(Merck) using a 12 in. Vigreux column and stored under 3A
molecular sieves. The gas-phase pyridine (Py) adsorption was con-
ducted simultaneously for all POMs. Platinum crucibles loaded
with the samples (∼20 mg) were placed in a shallow porcelain
plate and inserted into a glass tube inside a tubular furnace (Ther-
molyne, model F21135). The catalysts were dehydrated in dried N2
(100 mL min−1) at 200 ◦C for 1 h. The system was then cooled to
100 ◦C and gaseous pyridine diluted with N2 was allowed to pass
through the samples for 1 h. Finally, the temperature was held at
150 ◦C under N2 for 2 h to remove any physically adsorbed pyridine.
The samples with adsorbed pyridine were analyzed by TG/DTG and
FTIR.
The mixed salts were synthesized by the precipitation
method, which consisted of the addition of (NH4)2CO3 (Vetec)
to H3PW (Sigma) followed by Cs2CO3 (Aldrich), leading to the
salts (NH4)xCs2.5−xH0.5PW (x = 2, 1.5, 1, 0.5): (NH4)2Cs0.5H0.5PW,
(NH4)1.5Cs1H0.5PW, (NH4)1Cs1.5H0.5PW and (NH4)0.5Cs2H0.5PW.
The samples were prepared by adding pre-determined amounts of a
0.20-mol L−1 aqueous solution of the carbonates (freshly prepared)
to a 0.08-mol L−1 fresh solution of H3PW (at a rate of 1 mL min−1).
The stoichiometry was in agreement with Eqs. (1)–(4):
(NH4)2CO3 + 0.25Cs2CO3 + H3PW12O40
→ (NH4)2Cs0.5H0.5PW12O40 + 1.25CO2 + 1.25H2O
(1)
(2)
(3)
(4)
Acidity was obtained by TG/DTG data, as explained below. The
total number of acid sites (nPy) was determined by quantitative
analysis of TG/DTG curves of the catalysts before (Sample) and after
pyridine adsorption (Sample-Py), according to Eq. (5)
0.75(NH4)2CO3 + 0.5Cs2CO3 + H3PW12O40
→ (NH4)1.5Cs1.0H0.5PW12O40 + 1.25CO2 + 1.25H2O
Sample-Py
Sample
ꢀ
ꢁꢂ
ꢃ
ꢀ
ꢁꢂ
ꢃ
(m700/(mtotal − m200)) − (m700/(mtotal − m200))
nPy (mmol) =
MMPy
0.5(NH4)2CO3 + 0.75Cs2CO3 + H3PW12O40
×1000
(5)
→ (NH4)1.0Cs1.5H0.5PW12O40 + 1.25CO2 + 1.25H2O
First, it is determined the difference between the total mass
(mtotal) of a sample (containing pyridine) and the mass loss (m200
)
0.25(NH4)2CO3 + Cs2CO3 + H3PW12O40
between room temperature (25 ◦C) and 200 ◦C (i.e., the range of
temperature where there are losses of water and any remainder
physically adsorbed pyridine). This difference is related to the anhy-
drous sample with pyridine chemically adsorbed. Then, the mass
loss (m700) between 200 and 700 ◦C is normalized to a gram, divid-
ing this mass (m700) by that difference (mtotal − m200). Accordingly,
the same method is applied to the sample without pyridine (i.e.,
→ (NH4)0.5Cs2.0H0.5PW12O40 + 1.25CO2 + 1.25H2O
The solution was aged overnight at 25 ◦C, evaporated to dryness
at 40–50 ◦C and ground into fine particles in a mortar for further
calcination. Other details of this preparation and complete charac-
terization can be found elsewhere [19].

 Santos, Joicy S.
Santos, Joicy S.
 Dias, José A.
Dias, José A.
 Dias, Sílvia C.L.
Dias, Sílvia C.L.
 De MacEdo, Julio L.
De MacEdo, Julio L.
 Garcia, Fillipe A.C.
Garcia, Fillipe A.C.
 Almeida, Liana S.
Almeida, Liana S.
 De Carvalho, Eduardo N.C.B.
De Carvalho, Eduardo N.C.B.