J Surfact Deterg
hydrochloric acid. The precipitated solid was filtered off
and dried. Recrystallizing from diethyl ether gave
N-decanoyl-L-serine as white crystals [18].
The mass spectrograms, 1H NMR and the yields of
serine series are listed below.
(KRUSS, Germany, Model K100C) at 25 °C. All surface
tension values shown were the average of four measure-
ments. The CMC and cCMC were determined from the
breakpoint of the plot of the surface tension of their
aqueous solutions versus concentration.
Ser-6C
MS (negative): m/z 202.1 [M - H]-, 237.9 [M ? Cl]-,
405.1 [2 M - H]-, 441.0 [2 M ? Cl]-
Foamability
1H NMR (400 M, DMSO-d6) d0.86 (3H, t, CH3), 1.26
(4H, m, 2 9 CH2), 1.48 (2H, quint, CH2), 2.13 (2H, t,
CH2), 3.62 (2H, m, CH2), 4.26 (1H, sext, CH), 7.89 (1H, d,
NH), 12.48 (1H, s, COOH), yield 24.79 %.
Ser-8C
MS (negative): m/z 230.1 [M - H]-, 266.0 [M ? Cl]-,
344.0 [M ? CF3CO2]-, 483.3 [2 M - 2H ? Na]-
1H NMR (400 M, DMSO-d6) d0.86 (3H, t, CH3), 1.26
(8H, m, 4 9 CH2), 1.48 (2H, quint, CH2), 2.13 (2H, t,
CH2), 3.62 (2H, m, CH2), 4.26 (1H, sext, CH), 7.89 (1H, d,
NH), 12.48 (1H, s, COOH), yield 49.85 %.
Ser-10C
MS (negative): m/z 258.2 [M - H]-, 294.0 [M ? Cl]-,
372.1 [M ? CF3CO2]-
1H NMR (400 M, DMSO-d6) d0.86 (3H, t, CH3), 1.27
(12H, m, 6 9 CH2), 1.48 (2H, quint, CH2), 2.16 (2H, t,
CH2), 3.62 (2H, m, CH2), 4.26 (1H, sext, CH), 7.93 (1H, d,
NH), 12.48 (1H, s, COOH), yield 53.04 %.
Ser-12C
First, 30 mL of the sodium of lipo-amino acids aqueous
solution was added into a 100-mL stoppered cylinder. Then
the stoppered cylinder was violently shaken 40 times. The
total volume and the volume of the aqueous solution left
were recorded at 0 min (time 0 min is set after the shak-
ing), 1, 2, 5, 10 and 30 min. The differences between the
two values was the volume of the foam.
Result and Discussion
Synthesis of N-Acyl Amino Acid
The hydrophobic chain is introduced by acyl chloride.
However, the instability of acyl chloride in an aqueous
solution is deadly during the preparation process of long-
chain acyl-amino acid. For acyl-glycine, the pH must be
exactly controlled at 8.5–9.5 with sodium hydroxide, and
this pH range is helpful for the nucleophilicity of the amino
group and makes it possible to reduce the hydrolysis
reaction of acyl chloride as much as possible. For the
preparation of long-chain acyl-serine, in order to avoid the
hydrolysis reaction as much as possible, the pH was con-
trolled with sodium carbonate, the dropping speed of acyl
chloride should be necessarily slow.
MS (negative): m/z 286.0 [M - H]-, 322.1 [M ? Cl]-,
400.0 [M ? CF3CO2]-
1H NMR (400 M, DMSO-d6) d0.86 (3H, t, CH3), 1.27
(16H, m, 8 9 CH2), 1.48 (2H, quint, CH2), 2.16 (2H, t,
CH2), 3.64 (2H, m, CH2), 4.26 (1H, sext, CH), 7.88 (1H, d,
NH), 12.48 (1H, s, COOH), yield 57.84 %.
Ser-14C
MS (negative): m/z 314.0 [M - H]-, 350.0 [M ? Cl]-,
428.0 [M ? CF3CO2]-
Proton NMR Spectroscopy
1H NMR (400 M, DMSO-d6) d0.86 (3H, t, CH3), 1.27
(20H, m, 10 9 CH2), 1.48 (2H, quint, CH2), 2.16 (2H, t,
CH2), 3.64 (2H, m, CH2), 4.26 (1H, sext, CH), 7.88 (1H, d,
NH), 12.48 (1H, s, COOH), yield 61.22 %.
Ser-16C
MS (negative): m/z 342.3 [M - H]-, 378.3 [M ? Cl]-,
456.2 [M ? CF3CO2]-
1H NMR (400 M, DMSO-d6) d0.86 (3H, t, CH3), 1.27
(24H, m, 12 9 CH2), 1.48 (2H, quint, CH2), 2.16 (2H, t,
CH2), 3.64 (2H, m, CH2), 4.26 (1H, sext, CH), 7.88 (1H, d,
NH), 12.48 (1H, s, COOH), yield 83.87 %.
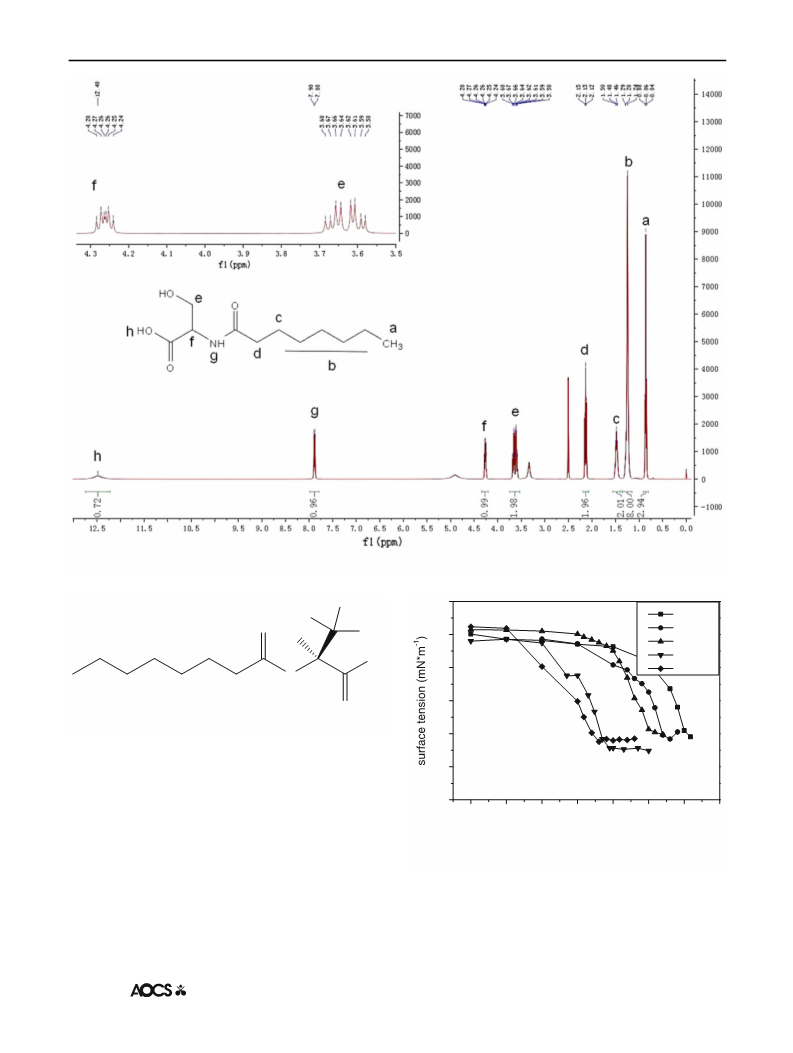
The 1H-NMR spectra of N-acyl amino acids were all
recorded. Taking N-caprylyl glycine as an example of the
glycine series, the chemical shifts of the constituent protons
are shown in Fig. 1. A triplet is observed at a d value of
0.8, which is characteristic of methyl protons adjacent to
methylene protons in long-chain alkyl group. The methy-
lene [(–CH2)4–] proton resonance is observed at d of 1–2 as
a multiplet. The two methylene groups adjacent to the
amide group give the signals at d of 1.62 (quint) and 2.24
(t). The signals at d of 3.89 are the resonance of protons in
the methylene in the structure of glycine. The rest are
solvent signals. There are no signals revealing the protons
of the acylamino group and carboxy group in CD3OD.
Due to the structural similarities between the glycine
and serine series, some similar signals can be seen in the
1H-NMR spectra from Figs. 1 and 2. Taking N-caprylyl
serine as an example (shown in Fig. 2), the protons in the
Determination of CMC and cCMC
The surface tension of different concentrations of N-acyl
amino acid sodium solutions was measured by the
Wilhelmy plate technique with a processor tensiometer
123

 Qiao, Weihong
Qiao, Weihong
 Qiao, Yangyang
Qiao, Yangyang