2
D. Sivaramakrishna, M.J. Swamy / Chemistry and Physics of Lipids 201 (2016) 1–10
and inhibit transporters with different functions (Wiles et al.,
006). They show varying degrees of potency towards T-type and
N-type calcium ion channel currents (Guo et al., 2008), and show
different activity (inhibition/enhancement) of fatty acid amide
hydrolase (Cascio et al., 2004).
In addition to the above interesting biological properties, NAAs
also exhibit considerable potential for use in various applications.
For example, N-acyl derivatives of carnosine and histidine act as
antioxidants and emulsifying agents (Murase et al., 1993), whereas
N-acylphenylalanine and N-acylmethionine altered the thermal
stability, foaming stability, emulsifying activity and gelation
properties of food ingredients such as egg white and whey protein
b-alanines were extracted with ethyl acetate. The extract was
2
washed successively with 0.1 M HCl and double distilled water. The
product obtained was recrystallized from hexane (for n = 8, 9) or
ethyl acetate (for n = 10–20). The overall yield for different NABAs
ranged around 70–80%. The final products were characterized by
1
13
thin layer chromatography, FTIR, H and C NMR spectroscopy as
well as by high resolution mass spectrometry.
Meltingpointswere determinedusing aSuperfit(Mumbai, India)
melting point apparatus. FTIR spectra were recorded using KBr
1
13
pellets on a Jasco FTIR 5300 Spectrometer. H and C NMR spectra
were recorded on a Bruker Avance NMR spectrometer at 400 and
1
100 MHz, respectively. CDCl
3
was used as the solvent for all H NMR
13
(
Ma et al., 1993). N-Acyl amino acids and N-acyl amino acid esters
as well as C NMR spectral studies with shorter chain NABAs (8–10),
whereas CDCl containing a few drops of CD OD was used as the
form gels in organic solvents and selectively form gels in oil-water
mixtures (Duarte et al., 2012). In view of their biodegradable and
non-toxic nature they can potentially be used in drug delivery
applications (Vintiloiu et al., 2008).
3
3
solvent for longer chain NABAs. High-resolution ESI mass spectra for
NABAs with 8–20 C-atoms in the acyl chain were recorded in the
positive ion mode on a Bruker MaXis Mass Spectrometer.
In view of the foregoing, it is important to investigate NAAs in a
systematic manner in order to understand their physicochemical
properties, phase behavior and interaction with other amphiphiles
in order to utilize them in various applications. In this direction we
have recently reported the thermotropic phase behavior, 3-
dimensional structure and supramolecular assembly of N-acyl-
2
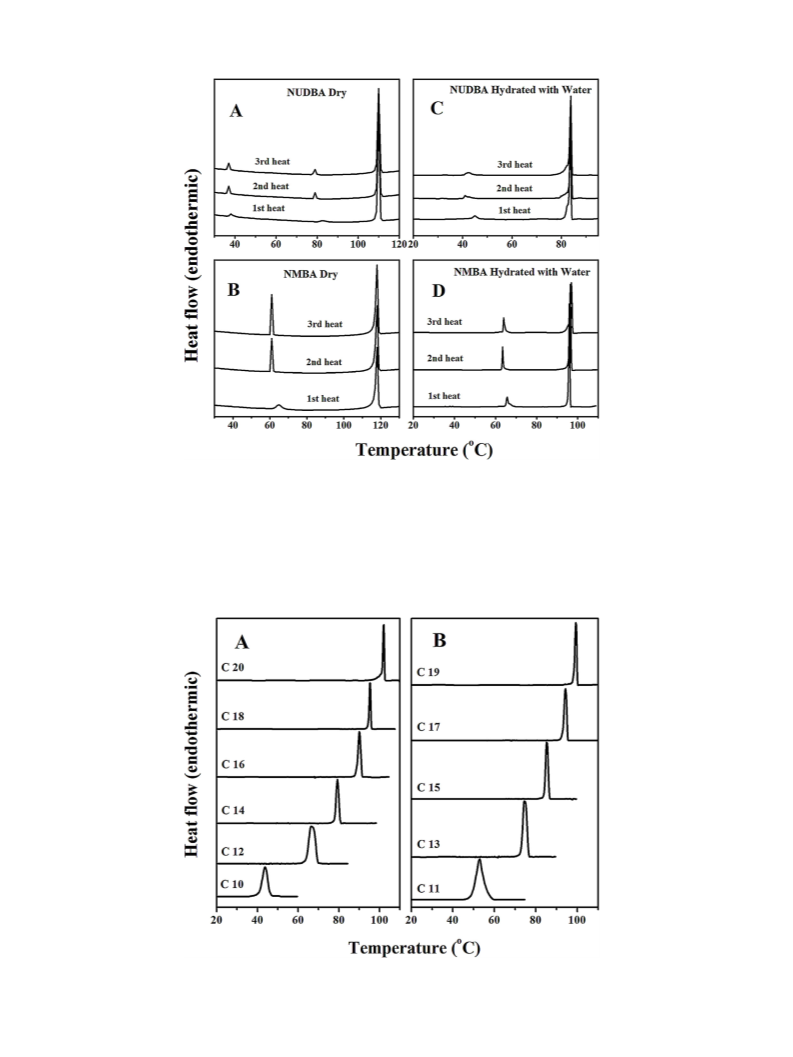
.3. DSC of dry N-acyl-b-alanines
DSC experiments with dry NABAs were carried out on a Perkin-
Elmer Pyris Diamond differential scanning calorimeter. The dry
compounds were accurately weighed using microbalance
Perkin-Elmer) in aluminum sample pans, covered with lids and
sealed by crimping. Each sample was subjected to three heating
a
glycines (NAGs) (Reddy et al., 2014) and N-acyl
Sivaramakrishna et al., 2015) which are constituents of mamma-
lian brain. In the present study we have focused our attention on N-
acyl -alanines (NABAs), which are homologues of NAGs and
L-alanines (NAAlas)
(
(
ꢀ
st
and two cooling scans at a scan rate of 2 /min. After the 1 heating
scan, small changes were observed in the minor transition peak
intensity and peak positions, whereas second and third heating
thermograms were almost identical. Therefore, the second heating
scans were considered for further analysis of all samples.
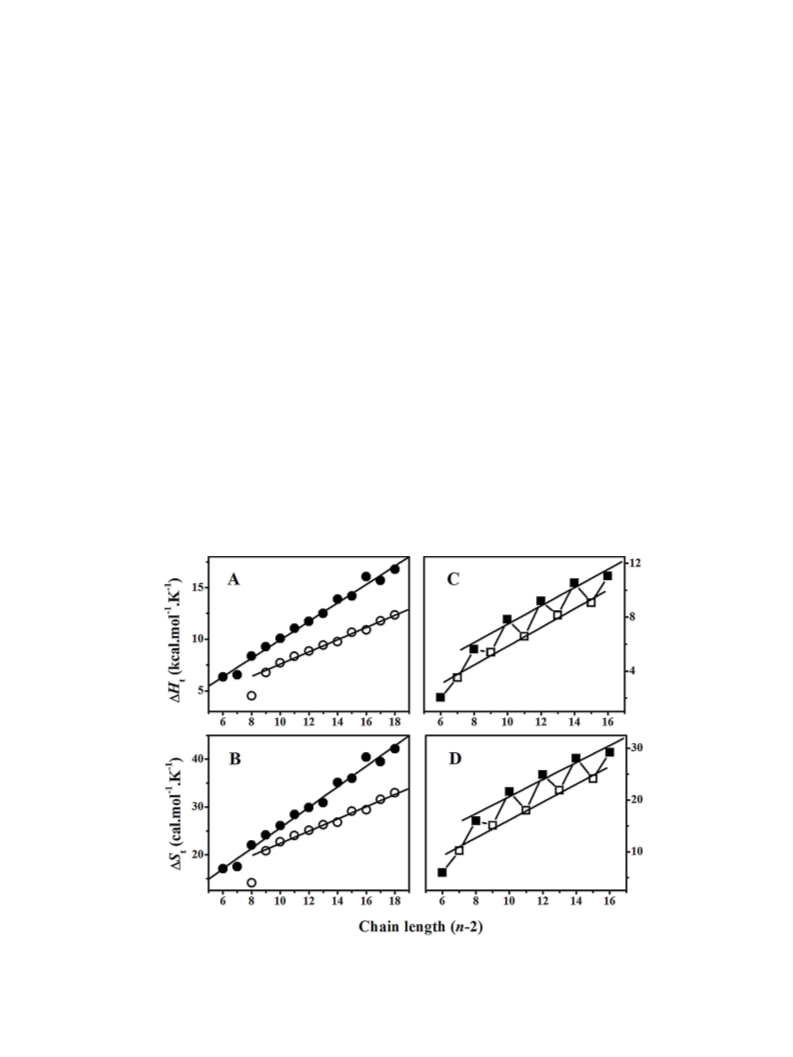
Transition enthalpies (
area under the transition curve using the software provided by the
instrument manufacturer. Transition entropies ( ) were calcu-
b
achiral isomers of NAAlas. We have synthesized a homologous
series of NABAs bearing saturated acyl chains (n = 8–20), investi-
gated their phase transitions in the dry state and upon complete
hydration (water and phosphate buffer) by differential scanning
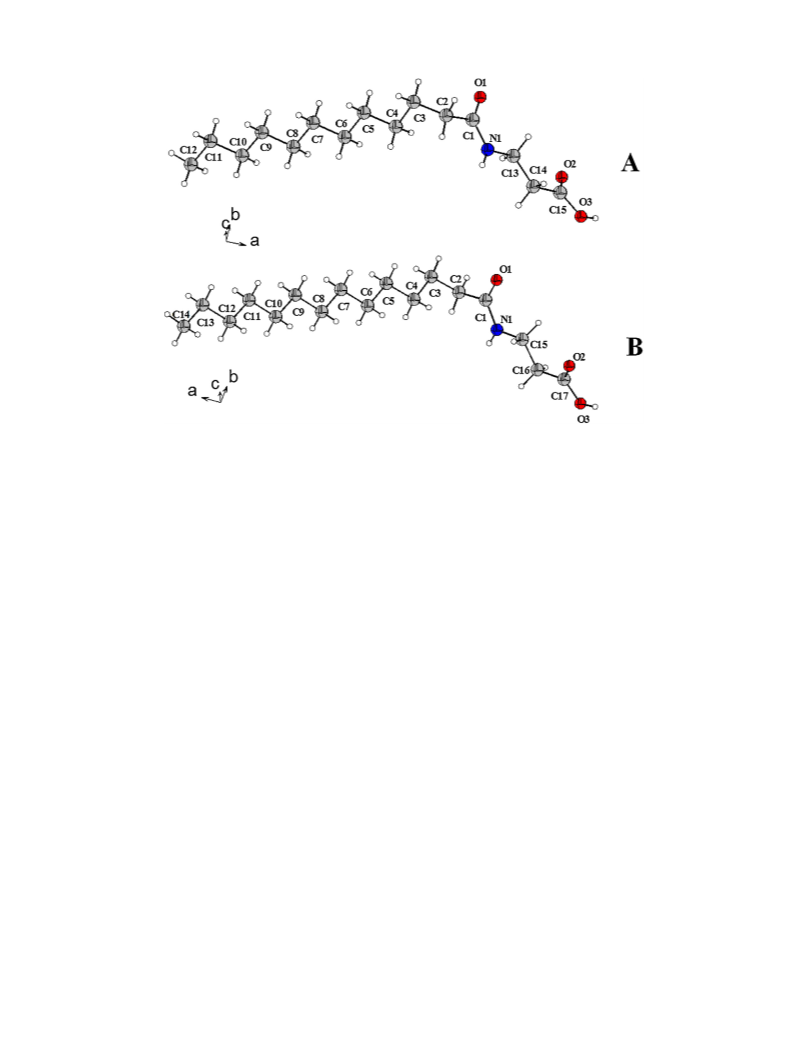
calorimetry. Crystal structures of two representatives, namely N-
t
DH ) were obtained by integrating the peak
lauroyl b-alanine (NLBA) and N-myristoyl b-alanine (NMBA) have
DS
t
been determined by single-crystal X-ray diffraction and analyzed
the molecular packing and intermolecular interactions have been
analyzed. The results obtained are discussed in this report.
lated from the transition enthalpies assuming a first order
transition according to equation 1 (Marsh, 1990).
D
S
t
=
D
H
t
/T
t
(1)
2. Experimental section
where T
t
refers to the transition temperature.
2.1. Materials
2
.4. DSC of hydrated N-acyl-
b-alanines
b
-Alanine ethyl ester hydrochloride and long chain fatty acids
(CH COOH, n = 6–18) were purchased from Sigma-Aldrich
(
(
(
CH
3
2 n
)
DSC studies with hydrated samples were performed using a VP-
DSC equipment from Microcal LLC (Northampton, MA, USA).
Milwaukee, WI, USA). Oxalyl chloride was purchased from Merck
Germany). Other chemicals and solvents (analytical grade) were
Accurately weighed compounds (ꢃ4–5 mg each) were dissolved in
purchased from local chemical suppliers.
about 300 ml chloroform containing a drop of methanol and the
2
solvent was evaporated by blowing a gentle stream of dry N gas.
2
.2. Synthesis of N-acyl -alanines
b
The lipid films thus obtained were vacuum desiccated for 5–6 h
and then hydrated with double distilled water or 20 mM phosphate
buffer, pH 7.6, containing 1 M NaCl (PBS) by subjecting the sample
through 4–5 cycles of freeze-thawing. To get a homogenous
mixture, the samples were sonicated for 1–2 min. Heating scans
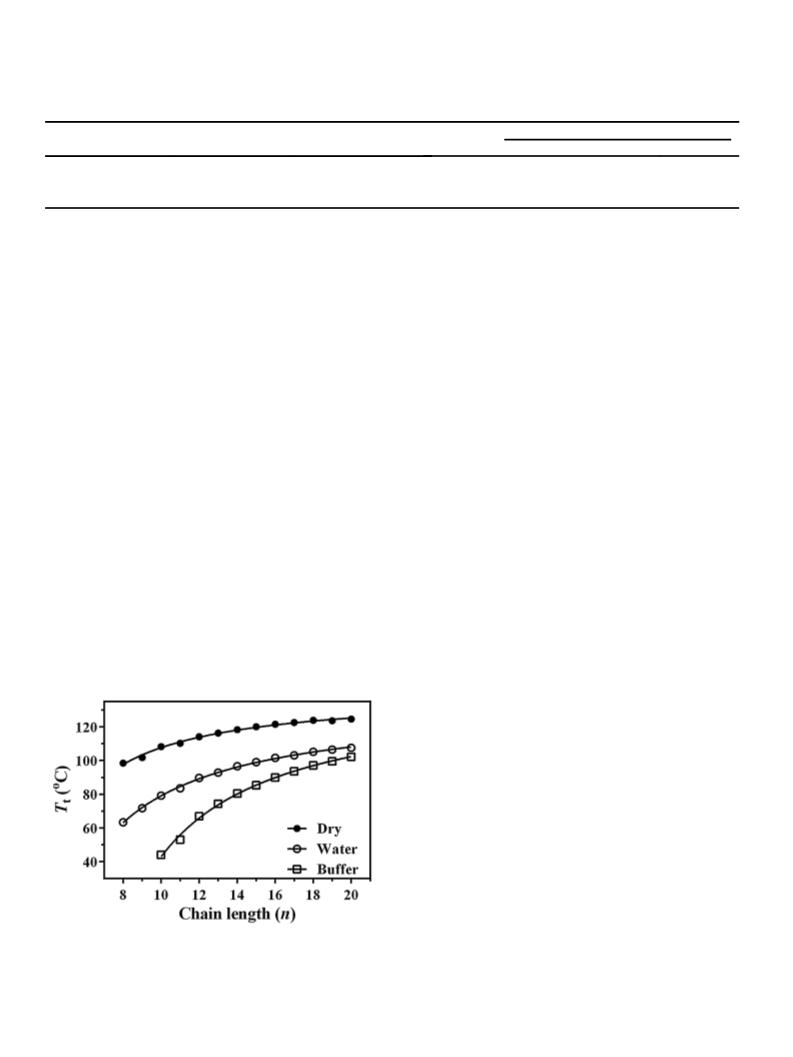
were recorded from 10 to 120 C at a scan rate of 60 /h. Three
heating and two cooling scans were performed for each sample.
The transition enthalpy was found to decrease in the second and
third heating scans. Therefore, the first heating scans were
considered for further analysis. Transition temperatures, enthal-
pies and width at half height were determined using the Origin
software provided by the calorimeter. Transition entropies were
calculated from the transition enthalpies assuming a first order
transition according to Eq. (1).
NABAs were prepared by a procedure that is essentially similar
to that reported for the synthesis of NAAlas (Sivaramakrishna et al.,
015). First, fatty acyl chlorides were prepared from the
corresponding fatty acids (1 mol eq.) by treating with oxalyl
chloride as described earlier (Akoka et al., 1988). -Alanine ethyl
2
ꢀ
ꢀ
b
ester hydrochloride (1 mol eq.) and sodium bicarbonate (2 mol eq.)
were dissolved in about 5 ml of distilled water, stirred for 5–10 min
and 10 ml of chloroform was added. After stirring for 10–15 min at
room temperature, the acid chloride, dissolved in chloroform, was
added dropwise and the reaction mixture was kept under constant
stirring for 3 h. The N-acyl
extracted with chloroform and washed successively with the
double distilled water, saturated brine solution, 0.1 M HCl and
b-alanine ethyl ester obtained was
double distilled water. The solid N-acyl-
obtained after evaporating the solvent were taken in MeOH:H
3:1, v/v) and hydrolyzed with LiOH by overnight stirring. Then the
reaction mixtures were acidified with 2 M HCl and the N-acyl
b
-alanine ethyl esters
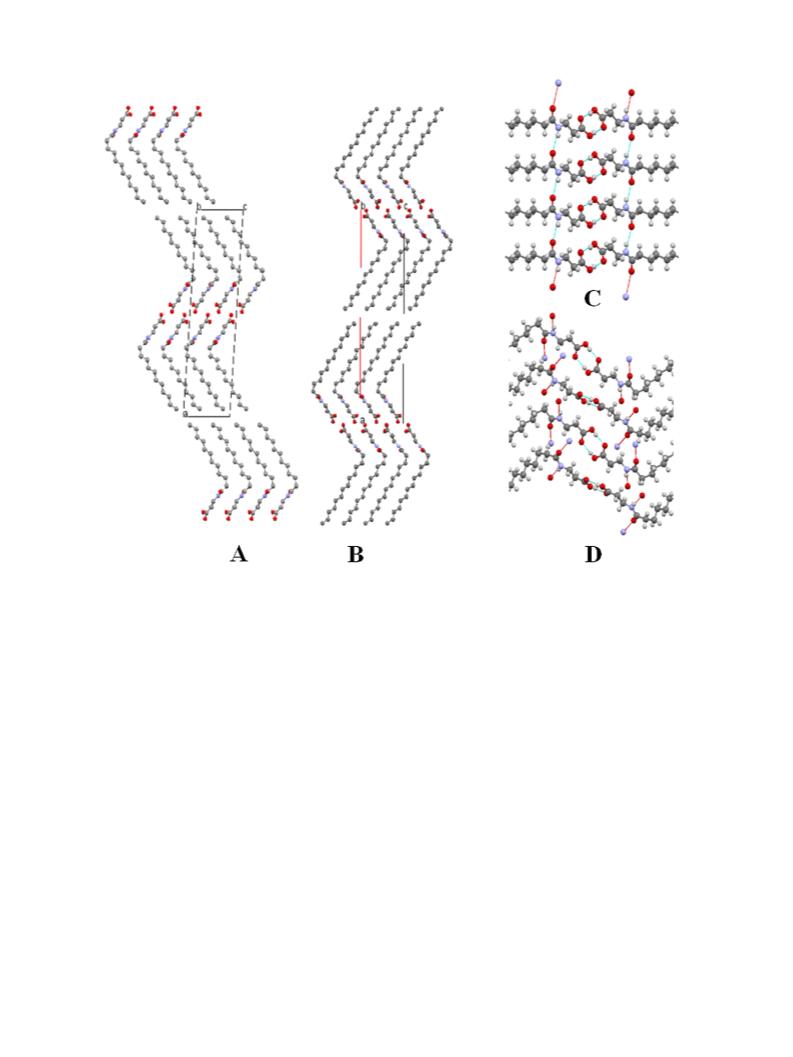
2.5. Crystallization and X-ray diffraction
2
O
(
Thin plate-type, colorless single crystals of NLBA and NMBA
were grown at room temperature from ethyl acetate containing a

 Sivaramakrishna
Sivaramakrishna
 Swamy, Musti J.
Swamy, Musti J.