Journal of the American Chemical Society
Article
Our simulations suggest that, like the symmetrical tertiary
amides, the presence of a second alkyl chain causes a kink in
the conformation, which leads to more gauche conformations
than in linear amides (Table S14 in the Supporting
Information). In agreement with the observed trends in
rectification ratios, all of the asymmetrically branched amides
had r+ < 7.0. This result indicated that the amides are less
ordered in their orientation with respect to the surface of the
gold in comparison to linear amides and thus that these SAMs
of tertiary amides are probably in more disordered
conformations. This conclusion was supported by our MD
simulations, particularly for SAMs made from amide 11(8,4)
(ftrans = 0.78, fdown = 0.58) and SAMs made from amide 12(10,2)
(ftrans = 0.91, fdown = 0.35), which indicate substantial
conformational disorder.
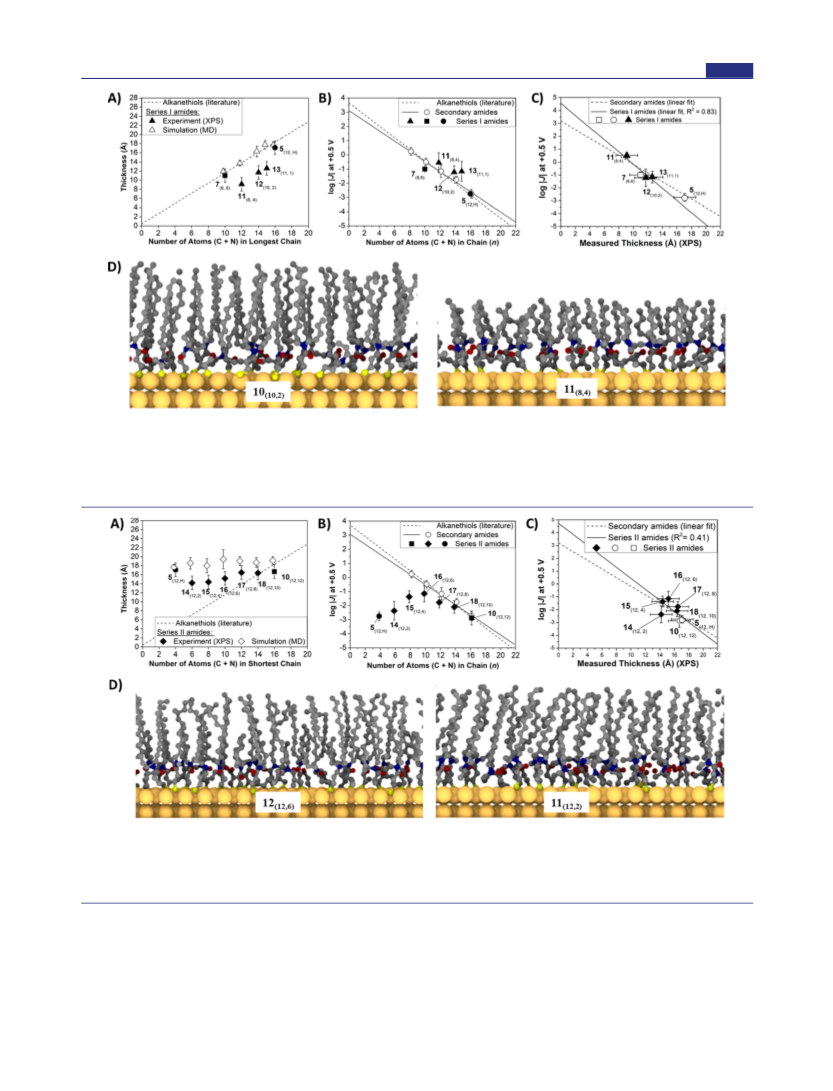
Unlike the secondary amides and symmetrical tertiary
amides, the current density measurements for series I amides
did not correlate linearly with chain length (Figure 7A, R2 =
0.65). A positive correlation was observed, however, between
the current density and the thickness (determined by ARXPS)
of the SAMs (Figure 7C, R2 = 0.83). While this result implies
that the rate of tunneling through this set of SAMseven
when they have disordered conformationsstill correlates
with the thickness of the monolayer, the value of β (i.e., the
slope) is larger for series I amides than for secondary amides.
This difference in β indicates that the relationship between
thickness and J(V) may be different for ordered than for
disordered SAMs.
Series II Amides (Figure 2; X(n,m), n = 12). Series II
amides, which include the asymmetrical tertiary amides 14−
18, the secondary amide 5(12,H) and the symmetrical tertiary
amide 10(12,12), was designed to test how the asymmetry in
branching (which we believe correlates with conformational
disorder) influences tunneling. Measurements of packing
density by XPS indicated that, like series I amides, the density
of asymmetrical tertiary amides (i.e., 14−18) was greater than
that of symmetrical tertiary amides but less than that of
secondary amides. This result suggests that asymmetrical
branching forces the longer chain to adopt a bent
conformation, in order to maximize their intermolecular
contacts and reduce the volume they occupy on the surface.
The thickness of the monolayers (as measured by ARXPS)
across the series remained approximately the same (Figure
8A), comparable to the case for the linear amide 5(12,H). A
small decrease in thickness was observed as the length of chain
b decreased, but chain awith its greater number of carbon
atomsappeared to dominate the thickness of the monolayer.
The simulations also show a relatively constant thickness of the
monolayer for this series of amides (Figure 8A).
|J(V)| against monolayer thickness in Figure 8C. Unlike the
case with series I amides, we observed no linear relationship
between log |J(V)| and monolayer thickness for series II
amides, indicating that in this series the rate of charge
tunneling does not correlate directly with thickness. Moreover,
the discrepancy between J(V) and thickness of 14−18, relative
to 10(12,12) or 5(12,H), cannot be accounted for by changes in
packing density within the SAM (as 5 has a higher packing
density than any of 14−18 and 10 has a lower packing density
than any of 14−18). We thus conclude, again by elimination,
that the conformation of these SAMs must be influencing the
rate of tunneling and that conformation does influence
tunneling rates in molecular junctions.
Unlike symmetrical tertiary amides, the unsymmetrical
tertiary amides in series II have tunneling rates that are higher
than those of secondary amides (or alkanethiols) of equivalent
thickness (or chain length). We believe that this result is a
consequence of the asymmetry in the lengths of chains a and b,
which causes more extensive disorder in asymmetrical tertiary
amides than in symmetric tertiary amides. This difference in
disorder could result in a poorly defined interface between the
top EGaIn electrode and the SAM and may cause electrical
contacts to be made farther down the chain than the ARXPS-
determined thickness would suggest.
CONCLUSION
■
By analyzing molecules that are similar electronically but that
differ in their conformation, we studied the effects of
conformation, thickness, and packing density of molecules in
a SAM on the rate of charge transport (CT) through them. We
observe differences in tunneling characteristics that cannot be
accounted for by changes in thickness or packing density and
conclude by elimination that charge tunneling through SAMs is
influenced by the conformations of the molecules and is not
solely determined by the distance between the two electrodes.
By comparing symmetric tertiary amides to secondary
amides, we conclude that the rate of CT is higher through
alkane chains with trans conformations than through those
with gauche conformations. By analyzing Series I amides, which
have the same number and type of atoms but are distributed
differently among chains a and b, we conclude that the spatial
arrangement of atoms matters. That is, tunneling is not solely
dependent on the density of atoms of a particular type within
the monolayer. These results suggest that CT may be a
through-bond process.
We conclude from our analysis of series II amides that the
conformation (or conformational disorder) influences the rates
of CT. This series revealed that a change in thickness does not
always correlate with an equivalent change in J(V), even for
simple alkanethiols. We believe that the results of this series of
amides indicate that conformational disorder can create a
poorly defined electrode−molecule interface, which can
artificially increase J(V).
Finally, we observed a clear relationship between conforma-
tional order and the dipole-induced rectification in these
systems. Specifically, SAMs that were more conformationally
ordered yielded molecular junctions with larger rectification
ratios in comparison to more conformationally disordered
SAMs. This result strongly suggests that conformation, and not
solely molecular structure, should be considered in the design
and analysis of molecular junctions.
The rectification ratios at 1.0 V for amides (r+ < 7.5; Table
S12 in the Supporting Information) are all lower than those for
the well-ordered SAMs in the series, 5(12,H) (r+ = 10.42) and
10(12,12) (r+ = 11.22), which suggests that amides 14−18 form
disordered SAMs. This interpretation was supported by the
MD simulations (Table S14 in the Supporting Information),
which show a significant number of gauche conformations
(ftrans ranges from 0.82 to 0.93) and varied chain orientations
(fdown ranges from 0.15 to 0.6).
The tunneling rates of series II amides (Figure 8B) are all
higher than that of a corresponding secondary amide of length
n = 16 (i.e, 5(12,H)). From the results of series I amides,
however, we did not expect that the rates of tunneling would
necessarily correlate with chain length, and we thus plotted log
3490
J. Am. Chem. Soc. 2021, 143, 3481−3493

 Belding, Lee
Belding, Lee
 Root, Samuel E.
Root, Samuel E.
 Li, Yuan
Li, Yuan
 Park, Junwoo
Park, Junwoo
 Baghbanzadeh, Mostafa
Baghbanzadeh, Mostafa
 Rojas, Edwin
Rojas, Edwin
 Pieters, Priscilla F.
Pieters, Priscilla F.
 Yoon, Hyo Jae
Yoon, Hyo Jae
 Whitesides, George M.
Whitesides, George M.