Please do not adjust margins
Page 3 of 5
Green Chemistry
DOI: 10.1039/C5GC01404B
Journal Name
COMMUNICATION
CHO
OH
Conclusions
2
40
50
50
50
60
50
50
50
60
60
50
60
60
98
95
92
95
95
94
94
96
91
94
98
92
90
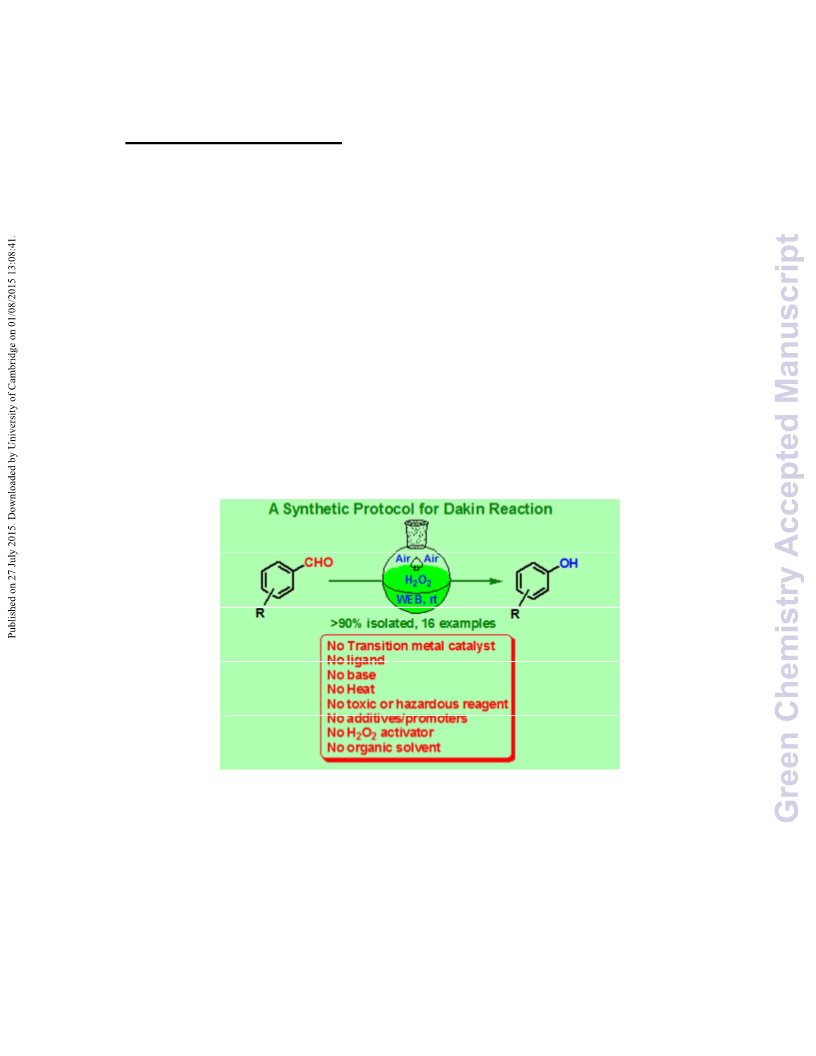
In conclusion, we have developed an aerobic and mild catalytic
system for the Dakin oxidation. In this manuscript we
investigated the applicability of the hydrogen peroxide using
natural feedstock WEB for the Dakin oxidation of a wide range
of organic compounds. Our method is flexible and is tolerant
of various electron-donating and electron-withdrawing groups
at the ortho-, meta-, and para-positions on the aromatic ring.
It is remarkable that the catalytic system does not require
activation or any toxic ligand and additive/promoter. The
developed catalyst system represents, so far, the most
efficient greener protocol toward the Dakin oxidation in air at
room temperature and it provide efficient and safe oxidants to
the synthetic community. In terms of both economical and
environmental considerations, we believe that the oxidation
protocol holds a potential value in laboratory and industry in
HO
HO
CHO
CHO
CHO
OH
OH
OH
OH
OH
3
OH
OH
OH
Br
OH
Br
4
Br
Br
OH
OH
5
6
OEt
CHO
OEt
OH
OH
OH
O
2
N
O N
2
near future. All these advantages make
2 2
H O –WEB a
CHO
CHO
OH
OH
competitive catalyst and thus can be a clean and convenient
alternative for other industrially important reactions.
OH
OH
7
8
9
OMe
OMe
Acknowledgment
OH
OH
OH
P. Borah is highly thankful to UGC, New Delhi for the financial
support of this study.
OMe
CHO
OMe
OH
Notes and references
OH
1
2
3
(a) H. D. Dakin, Am. Chem. J., 1909, 62, 477–498; (b) H. D.
Dakin, H. T. Clarke and E. R. Taylor, Catechol. Org. Synth.
Coll., 1941, 1, 149–152; (c) H. D. Dakin, Org. Synth., 1933,
OH
CHO
OH
OH
149–154.
OH
OH
OH
OH
(a) M. B. Hocking, Can. J. Chem., 1973, 51, 2384–2392; (b) M.
Hocking and J. Ong, Can. J. Chem., 1977, 55, 102–110; (c) M.
B. Hocking, K. Bhandari, B. Shell and T. A. Smyth, J. Org.
Chem., 1982, 47, 4208–4215.
1
0
1
2
3
4
Br
Br
Cl
CHO
OH
(a) R. Anand, R. Maheswari, K. U. Gore and V. R. Chumbhale,
1
1
1
1
Chem. Commun., 2002,
Yamashita, JP Patent 53009734, 1978; (c) H. R. Bjorsvik, L.
Liguori and F. Minisci, Org. Process Res. Dev., 2000, , 534–
3, 321–326; (b) K. Nakajima and A.
Cl
4
CHO
CHO
OH
OH
OH
OH
543; (d) G. S. Clark, Perfum. Flav., 1990, 15, 45–54; (e) Y. M.
C. Sunny and W. M. Yu, US Patent 2002192606, 2002; (f) E.
Okutsu and H. Tsuzuki, JP Patent 2002116527, 2002; (g) E.
Klarmann, L. W. Gatyas and V. A. Shternov, J. Am. Chem.
Soc., 1932, 54, 298–305; (h) E. Klarmann, L. W. Gatyas and V.
A. Shternov, J. Am. Chem. Soc., 1931, 53, 3397–3407; (i) E.
Klarmann, L. W. Gatyas and V. A. Shternov, J. Am. Chem.
Soc., 1932, 54, 1204–1211; (j) J. Lerchová, M. Obali, L.
Kotulak and J. J. Pospíšil, Polym. Sci.: Polymer Symposia
Volume 40, Issue 1, pages 297–306, 1973.
OH
OH
OH
CHO
4
(a) S. Chen, M. S. Hossain and F. W. Foss Jr., Org. Lett., 2012,
MeO
OMe MeO
OMe
OH
1
2
4
, 2806–2809; (b) S. Chen and F. W. Foss Jr., Org. Lett.,
012, 14, 5150–5153; (c) R. S. Varma and K. P. Naicker, Org.
, 189–191; (d) R. Bernini, A. Coratti, G.
Provenzano, G. Fabrizi and D. Tofani, Tetrahedron, 2005, 61
821–1825; (e) T. V. Hansen and L. Skattebøl, Tetrahedron
OH
OH
OH
CHO
Lett., 1999, 1
OH
,
1
5
6
50
50
95
94
1
Lett., 2005, 46, 3357–3358; (f) E. T. da Silva, C. A. Camara, O.
A. C. Antunes, E. J. Barreiro and C. A. M. Fraga, Synth.
Commun., 2008, 38, 784–788; (g) G. W. Kabalka, N. K. Reddy
and C. Narayana, Tetrahedron Lett., 1992, 33, 865–866; (h) S.
Yamazaki, Chem. Lett., 1995, 127–128; (i) A. Roy, K. R. Reddy,
P. K. Mohanta, H. Ila and H. Junjappa, Synth. Commun., 1999,
29, 3781–3791.
CHO
OH
OH
OH
1
OMe
OMe
a
Reaction conditions: arylaldehydes (1 mmol), 30% H O (2 equiv.
2
2
with respect of each substrate) in WEB (3 mL) at room temperature.
5
(a) J. L. Zambrano and R. Dorta, Synlett, 2003, 1545–1546; (b)
G. J. Brink, ten, I. W. C. E. Arends and R. A. Sheldon, Chem.
Rev., 2004, 104, 4105–4124.
b
Yields refer to isolated yields.
This journal is © The Royal Society of Chemistry 20xx
J. Name., 2013, 00, 1-3 | 3
Please do not adjust margins

 Saikia, Bishwajit
Saikia, Bishwajit
 Borah, Parinita
Borah, Parinita
 Barua, Nabin Chandra
Barua, Nabin Chandra