derived from petroleum may be of very low isotope ratio compared
to modern terrestrial materials, introducing additional error.2
Finally, calculation of isotope ratio from a weighted average is
least satisfactory for small molecules because the number of moles
of carbon added in the derivatization can be comparable to the
number of moles of carbon in the parent analyte.6 In recognition
mode. A 50-m × 0.32-mm × 0.52-µm film thickness HP-cross-
linked-FFAP capillary column (Hewlett-Packard, Wilmington, DE)
separated a test analyte, 1-hexadecanol, from the solvent, ethyl
ether. Using an automated rotary valve (Valco Instruments,
Houston, TX), the precursor molecule may be directed either to
a flame ionization detector (FID) for quantitative analysis and
methods development or to the pyrolysis furnace for thermolytic
dissociation. The in-house-constructed pyrolysis furnace consisted
of one continuous piece of deactivated fused silica, 0.25 mm i.d.,
resistively heated by a ceramic furnace; the pyrolysis zone was
25 cm in length. The pyrolysis temperature is normally set to a
single temperature between 500 and 700 °C, (0.5 °C, using a
CN9000A series temperature controller (Omega Engineering,
Stamford, CT). Pyrolysis products eluting from the furnace were
transferred to GC(II) via a heated transfer line and cryofocused
at the column top at -40 °C, followed by separation on a 60-m ×
0.32-mm × 0.25-µm film thickness HP-1 (cross-linked methyl
siloxane) capillary column (Hewlett-Packard). The GC(II) tem-
perature profile consisted of a linear temperature ramp from -40
to 260 °C at 3 °C/ min. After separation, the products were
directed via a second rotary valve either to a Varian Saturn III
QISMS ion-trap mass spectrometer (Walnut Creek, CA) for
structural analysis or to an in-house-built combustion/ water-trap/
open-split interface and then to a Finnigan-MAT model 252 IRMS
7
of these considerations, Tetens et al. introduced a one-carbon
derivatization for high-precision determination of plasma lactate
that substituted a single methyl group, adding a single carbon
per molecule, for a trimethylsilyl group that added three carbon
atoms per molecule, presented previously by Khalfallah et al.8
A separate factor making derivatization undesirable is that the
physical organic mechanism of the derivatization reaction may
induce isotopic fractionation in the analyte.6 Thus, for highest
accuracy, no carbon should be added to analyte, and any other
treatments should be quantitative. As yet, no general strategies
for conversion of analytes to volatile analogues without addition
of extraneous carbon have been presented.
We recently introduced an online strategy for the determina-
tion of intramolecular carbon isotope ratios using fatty acid methyl
9
esters (FAMEs) as a test case, termed position-specific isotope
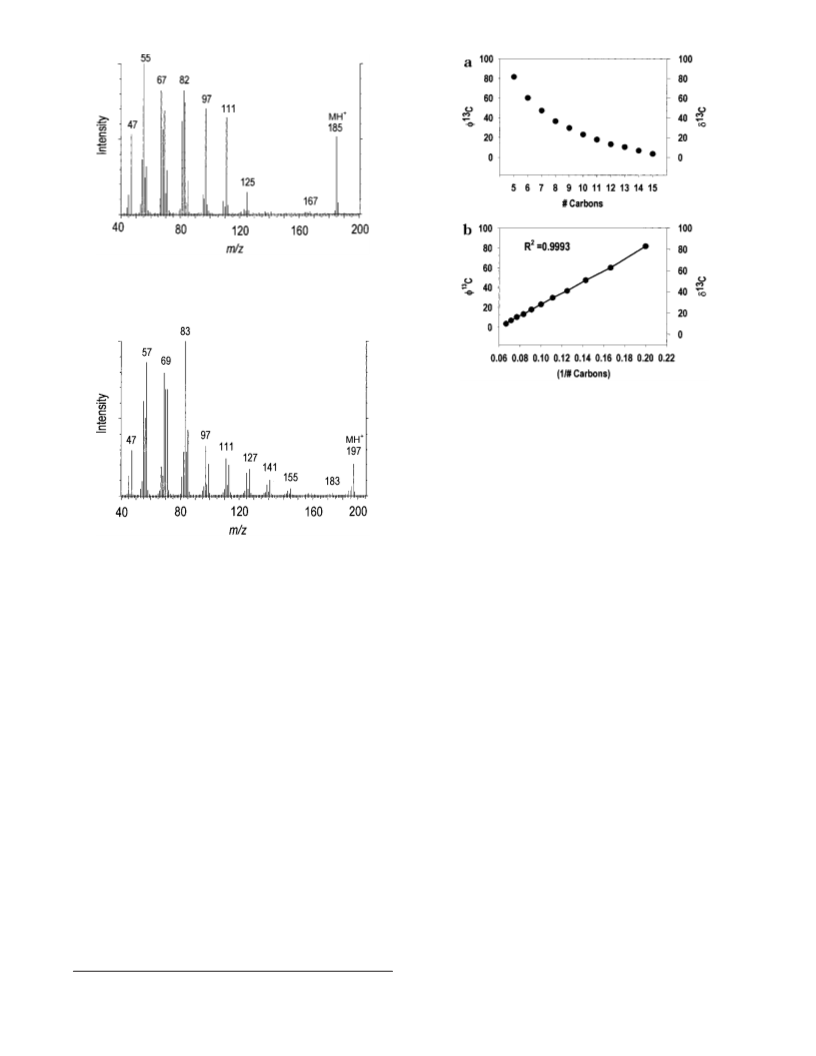
analysis (PSIA). Purified FAMEs are fragmented pyrolytically,
and GCC-IRMS is applied to separate and determine isotope ratio
for individual fragments. The derivative methyl group adds only
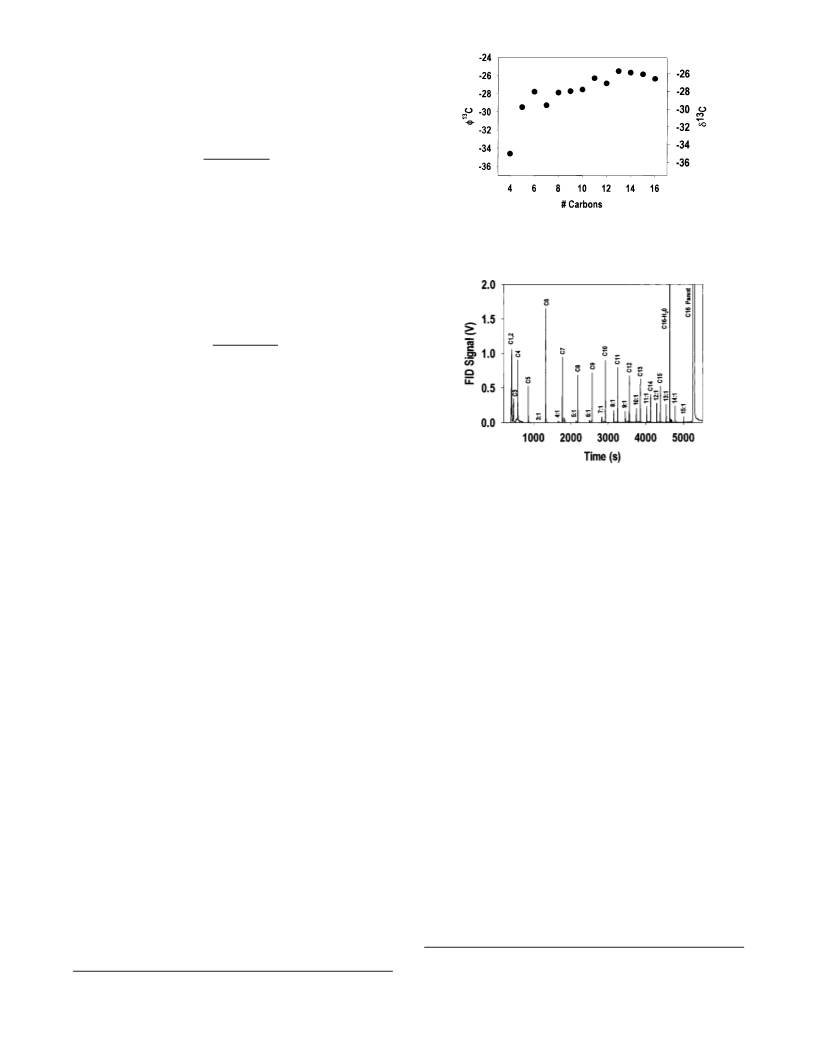
a single carbon per fatty acid, or about 5% total carbon. Previous
measurements have indicated that the methyl group may be
depleted in 13C to a level of about δ C ) -50‰ which results in
an overall depletion in isotope ratio by -1‰ or more, based on
mass balance considerations and assuming the fatty acid is
biosynthesized from carbon fixed by a modern plant. More
dramatic is the estimated effect on pyrolytic fragments of the
(
Bremen, Germany) for isotopic analysis. The combustion furnace
consisted of deactivated fused silica filled with oxidized Cu metal
held at 850 °C.11
13
Molecular mass spectra were obtained in positive ion electron
impact (EI) mode and in chemical ionization (CI) mode using
either methane or ethanol as a CI reagent. Conditions common
to all analyses were as follows: electron multiplier voltage, 1.6
kV; emission current, 10 mA, manifold (ion trap) temperature )
3
analyte as small as C , where addition of a single derivatization
carbon represents as much as one-quarter of the total carbon,
leading to a depletion of overall isotope ratio by 5‰.
1
00 °C, axial modulation ) 2.4 V, and acquisition time ) 1 s.
Spectral interpretation for structural assignment was augmented
using the Wiley mass spectra database (Palisades, Newfield, NY).
Searches were conducted using the purity search parameter.
Natural abundance 1-hexadecanol, palmitic acid (hexadecanoic
Pioneering early MS work conducted on synthetic peptides
4
employed reduction by LiAlD to generate the more volatile
polyamine analogues for electron impact ionization.10 Although
this approach has been superseded by more convenient deriva-
tization reagents, it offers clear advantages for the isotopic analysis.
We report here application of this strategy to convert fatty acid
methyl esters to their corresponding alcohols prior to IRMS
analysis, to provide suitable chromatography while avoiding the
addition of extraneous carbon. We also report the feasibility of
online pyrolysis of fatty alcohols for PSIA analysis.
acid, 16:0), LiAlH
4
, and BF
3
in methanol (14%) were purchased
13
from Sigma Chemical Co. (St. Louis, MO), and [1- C]-16:0 was
purchased from Cambridge Isotope Laboratories (Cambridge,
MA). Ethyl ether was obtained from Fisher Scientific (Pittsburgh,
PA). All were used without further purification.
Palmitic acid was converted to methyl palmitate (Me16:0) by
reaction with BF
previously.12 The quantitative reduction of Me16:0 to hexadecanol
was carried out via the following procedure. LiAlH (1.5 molar
3
in methanol according to methods described
EXPERIMENTAL SECTION
4
The online PSIA system consists of an in-house-built GC(I)-
equiv) in the form of dry powder and about 1 mL of peroxide-free
ethyl ether were added to about 2.5 mg of neat analyte in a coned-
bottom test tube. The reaction mixture was refluxed at 50 °C for
pyrolysis-GC(II) system coupled to a combustion/ high-precision
IRMS and an ion trap MS for both high-precision 13C/ 12
C
measurements and structure identification, as described previ-
ously.9 GC(I) is used for separations prior to pyrolysis, and GC-
4
5 min and then cooled on ice. Two drops of distilled, deionized
water were added to exhaust excess reagent by conversion of
LiAlH to solid Li and Al hydroxides. Water must be added
carefully, as the reaction with LiAlH is rapid, exothermic, and
evolves H gas. The mixture was centrifuged, and the organic
(
II) separates pyrolytic fragments for analysis. Injections were
4
made in GC(I) using a split/ splitless injector, operated in split
4
(
(
(
7) Tetens, V.; Kristensen, N. B.; Calder, A. G. Anal. Chem. 1 9 9 5 , 67, 858-
62.
8) Khalfallah, Y.; Normand, S.; Tissot, S.; Pachiaudi, C.; Beylot, M.; Riou, J. P.
Biol. Mass Spectrom. 1 9 9 3 , 22, 707-711.
9) Corso, T. N.; Brenna, J. T. Proc. Natl. Acad. Sci. U.S.A. 1 9 9 7 , 94, 1049-
2
8
layer was removed and filtered through a 0.2-µm Teflon filter (MSI,
(11) Goodman, K. J. Anal. Chem. 1 9 9 8 , 70, 833-837.
(12) Sheaff, R. C.; Su, H.-M.; Keswick, L. A.; Brenna, J. T. J. Lipid Res. 1 9 9 5 ,
36, 998-1008.
1
053.
(
10) Beimann, K.; Gapp, F.; Seible, J. J. Am. Chem. Soc. 1 9 5 9 , 81, 22-74.
Analytical Chemistry, Vol. 70, No. 18, September 15, 1998 3753

 Corso
Corso
 Lewis
Lewis
 Brenna
Brenna