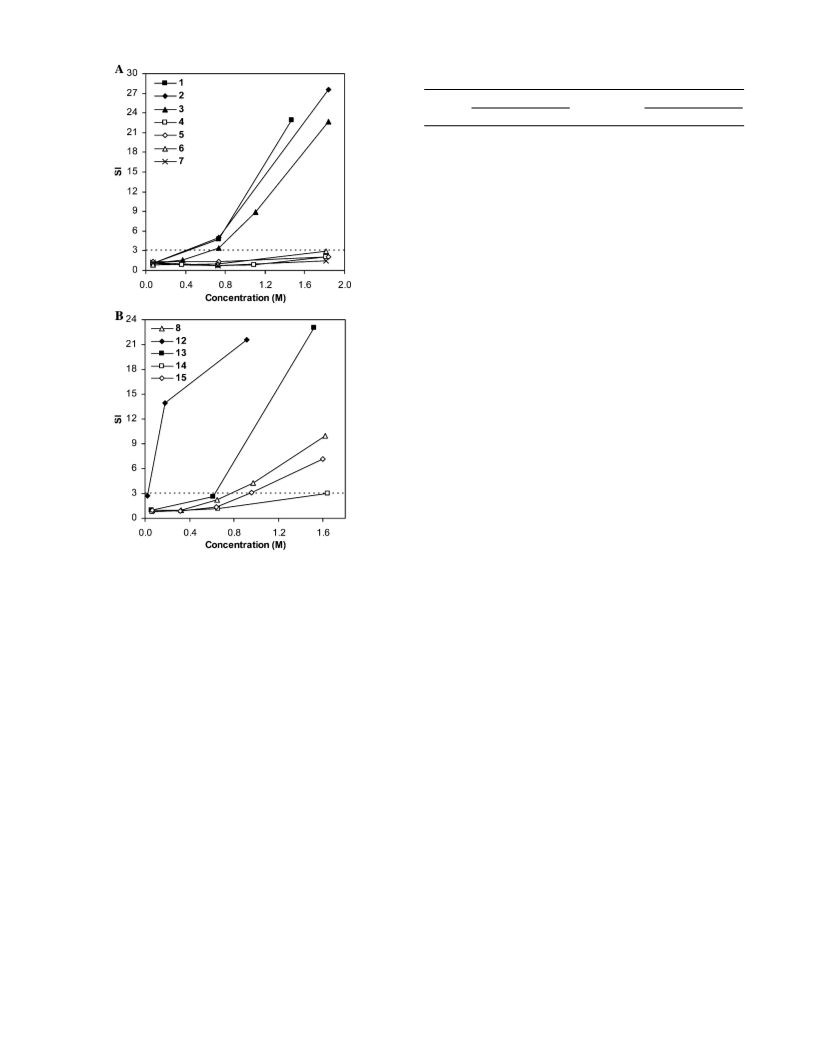
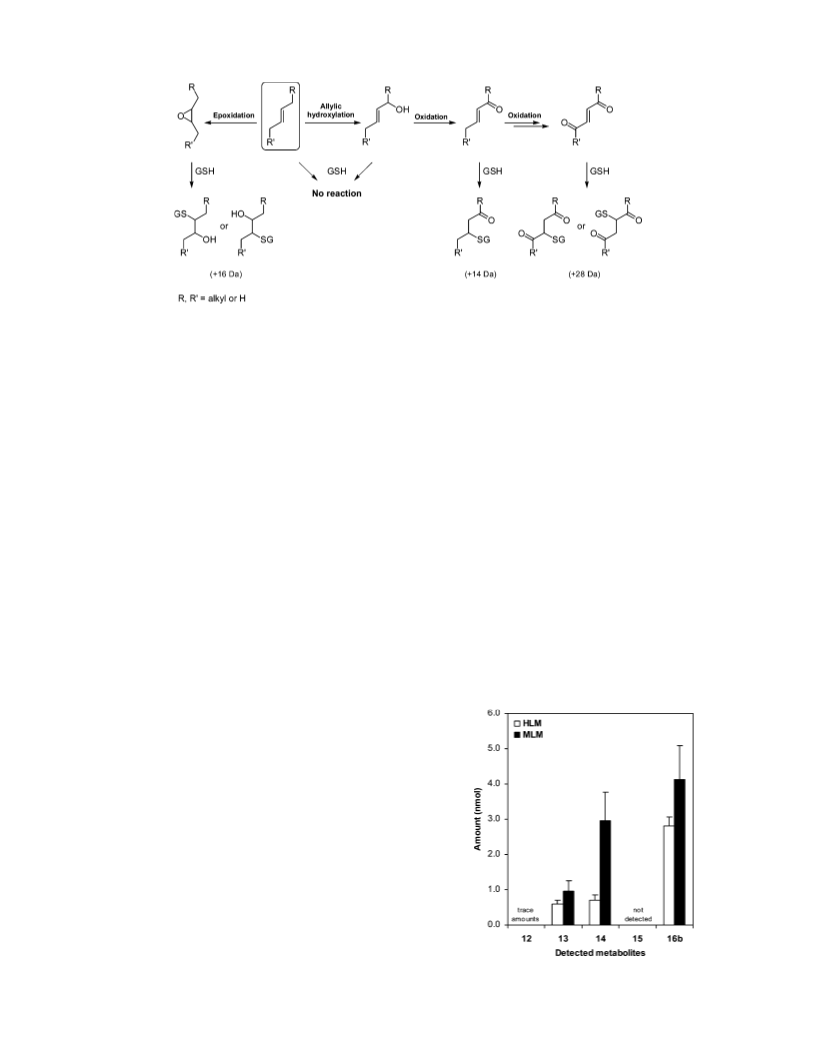
762 Chem. Res. Toxicol., Vol. 19, No. 6, 2006
Bergstro¨m et al.
acrolein (0.98 g, 11.0 mmol) was added dropwise. After it was
stirred at -78 °C for 60 min, the mixture was allowed to reach
room temperature and water (100 mL) was added. The aqueous
phase was extracted with n-pentane (100 mL), and the combined
organic phases were washed with water (100 mL) and brine (100
mL), dried over MgSO4, and concentrated under reduced pressure
at 0 °C. The crude product was purified by column chromatography
on silica gel (100% n-pentane) affording a 97:3 cis/trans mixture
6H, J ) 7.0, 3 × H10, 3 × H11, minor isomer), 0.89 (d, 6H, J )
6.6, 3 × H10, 3 × H11, major isomer), 1.09-1.90 (m, 10H, 2 ×
H4, 2 × H5, H6, 2 × H7, 2 × H8, H9), 2.58 (s, 2H, 2 × H2,
minor isomer), 2.63 (s, 2H, 2 × H2, major isomer). 13C NMR (100
MHz): δ 20.0 (C10, C11, major isomer), 20.1 (C10, C11, minor
isomer), 27.2 (C5, C7, major isomer), 28.9 (C5, C7, minor isomer),
32.3 (C9, minor isomer), 32.6 (C9, major isomer), 33.2 (C4, C8,
major isomer), 33.7 (C4, C8, minor isomer), 43.0 (C6, major
isomer), 43.1 (C6, minor isomer), 54.0 (C2, major isomer), 55.1
(C2, minor isomer), 58.6 (C3, major isomer), 60.0 (C3, minor
isomer). EI-MS (70 eV), m/z (%): (major isomer) 154 [M+] (4),
139 (62), 125 (16), 122 (17), 111 (51), 98 (40), 84 (100); (minor
isomer) 154 [M+] (4), 139 (50), 125 (13), 122 (13), 111 (45), 98
(35), 84 (100). IR (mixture of isomers) (cm-1): 3031, 2939, 2862,
1466, 1442, 917. Anal. calcd for C10H18O (mixture of isomers):
C, 77.87; H, 11.76. Found: C, 77.78; H, 11.74.
1
of 7 as a colorless oil (0.66 g, 48%). H NMR (400 MHz) (major
isomer): δ 0.90 (d, 6H, J ) 6.7, 3 × H1, 3 × H9), 1.02 (t, 3H, J
) 7.3, 3 × H8), 1.63 (nonet, 1H, J ) 6.7, H2), 2.07-2.15 (m, 4H,
2 × H3, 2 × H7), 4.81 (s, 1H, H10), 4.95 (s, 1H, H10), 5.48 (dt,
1H, J ) 11.6, 7.1, H4), 5.83 (d, 1H, J ) 11.6, H5). 13C NMR (100
MHz) (major isomer): δ 12.9 (C8), 22.5 (C1, C9), 29.1 (C2), 30.1
(C7), 37.8 (C3), 112.1 (C10), 130.6 (C5), 131.3 (C4), 147.4 (C6).
EI-MS (70 eV), m/z (%): 138 [M+] (34), 123 (35), 109 (100), 95
(29), 81 (57), 67 (75). IR (mixture of isomers) (cm-1): 3082, 2957,
2871, 1633, 1463, 895. Anal. calcd for C11H18 (mixture of
isomers): C, 86.88; H, 13.12. Found: C, 86.70; H, 13.06.
(1R,4R)-4-Isopropenyl-1-methyl-2-methylenecyclohexane (11)
(23). Sodium bis(trimethylsilyl)amide (2 M in THF, 9.00 mL, 18.0
mmol) was added dropwise to a stirred suspension of methyltri-
phenylphosphonium bromide (6.43 g, 18.0 mmol) in dry THF (70
mL) under N2 at room temperature. After 10 min, (2R,5R)-5-
isopropenyl-2-methylcyclohexanone (2.28 g, 15.0 mmol) was added
dropwise and the reaction mixture was heated to reflux. After 1 h,
the reaction mixture was allowed to reach room temperature and
n-pentane (70 mL) and water (70 mL) were added. The aqueous
phase was extracted with n-pentane (70 mL), and the combined
organic phases were washed with water (70 mL) and brine (70 mL),
dried over MgSO4, and concentrated under reduced pressure at 0
°C. The crude product was purified by column chromatography on
silica gel (100% n-pentane) affording 11 as a colorless oil (2.00 g,
1-Isopropyl-4-methylenecyclohexene, â-Terpinene (5) (20).
4-Isopropylcyclohex-3-enone was synthesized according to the
literature (21). Sodium bis(trimethylsilyl)amide (2 M in THF, 8.15
mL, 16.3 mmol) was added dropwise to a stirred suspension of
methyltriphenylphosphonium bromide (5.82 g, 16.3 mmol) in dry
THF (80 mL) under N2 at room temperature. After 30 min,
4-isopropylcyclohex-3-enone (1.50 g, 10.9 mmol) was added
dropwise and the mixture was heated to reflux for 1 h. After the
reaction mixture was allowed to reach room temperature, n-pentane
(50 mL) was added and the mixture was filtered. The filtrate was
washed with aqueous HCl (1 M, 3 × 50 mL), water (50 mL), and
brine (50 mL), dried over MgSO4, and concentrated under reduced
pressure at 0 °C. The crude product was purified by column
chromatography on silica gel (100% n-pentane) affording 5 as a
1
90%). H NMR (400 MHz): δ 0.98-1.11 (m, 1H, H6), 1.05 (d,
3H, J ) 6.6, 3 × H7), 1.31-1.42 (m, 1H, H5), 1.71 (s, 3H, 3 ×
H10), 1.79-1.89 (m, 2H, H5, H6), 1.92-2.22 (m, 3H, H1, H3,
H4), 2.31-2.41 (m, 1H, H3), 4.60 (s, 1H, H11), 4.69 (s, 3H, 2 ×
H9, H11). 13C NMR (100 MHz): δ 18.3 (C7), 20.9 (C10), 31.9
(C5), 36.5 (C6), 37.2 (C1), 41.9 (C3), 47.4 (C4), 104.7 (C11), 108.4
(C9), 150.1 (C8), 153.5 (C2). EI-MS (70 eV), m/z (%): 150 [M+]
(20), 135 (84), 121 (31), 107 (92), 93 (100), 79 (71).
1
colorless oil (1.26 g, 85%). H NMR (400 MHz): δ 0.99 (d, 6H,
J ) 7.0, 3 × H9, 3 × H10), 2.07-2.13 (m, 2H, 2 × H6), 2.18
(octet, 1H, J ) 7.0, H8), 2.27-2.32 (m, 2H, 2 × H5), 2.74-2.79
(m, 2H, 2 × H3), 4.72 (s, 2H, 2 × H7), 5.33-5.37 (m, 1H, H2).
13C NMR (100 MHz): δ 21.5 (C9, C10), 28.5 (C6), 32.2 (C5),
33.6 (C3), 35.1 (C8), 107.2 (C7), 117.5 (C2), 143.7 (C1), 146.8
(C4). EI-MS (70 eV), m/z (%): 136 [M+] (48), 121 (21), 93 (100),
91 (54), 77 (41).
(4R,7R)-7-Isopropenyl-4-methyl-1-oxa-spiro[2.5]octane (15).
n-Butyllithium (1.46 M in hexanes, 10.1 mL, 14.7 mmol) was added
dropwise to a stirred solution of trimethylsulfonium iodide (2.99
g, 14.7 mmol) in dry THF (80 mL) under N2 at -40 °C. After 5
min, (2R,5R)-5-isopropenyl-2-methylcyclohexanone (2.20 g, 14.7
mmol) was added dropwise and the mixture was stirred at -40 °C
for 1.5 h. The reaction mixture was allowed to reach room
temperature and concentrated under reduced pressure. Water (40
mL) was added, and the mixture was extracted with n-pentane (2
× 40 mL). The organic phases were washed with water (40 mL)
and brine (40 mL), dried over MgSO4, and concentrated under
reduced pressure. The crude product was purified by column
chromatography on silica gel (5% diethyl ether in hexanes) affording
4-Isopropyl-1-methylenecyclohexane (7) (22). Sodium bis-
(trimethylsilyl)amide (2 M in THF, 10.7 mL, 21.4 mmol) was added
dropwise to a stirred suspension of methyltriphenylphosphonium
bromide (7.65 g, 21.4 mmol) in dry THF (130 mL) under N2 at
room temperature. After 30 min, 4-isopropylcyclohexanone (2.00
g, 14.3 mmol) was added dropwise and the reaction mixture was
heated to reflux for 1.5 h. After the reaction mixture was allowed
to reach room temperature, n-pentane (100 mL) was added and
the solid was filtered off. The filtrate was washed with water (3 ×
50 mL) and brine (50 mL), dried over MgSO4, and concentrated
under reduced pressure at 0 °C. The crude product was purified by
column chromatography on silica gel (100% n-pentane) affording
7 as a colorless oil (1.44 g, 73%). 1H NMR (400 MHz): δ 0.86 (d,
6H, J ) 7.3, 3 × H9, 3 × H10), 1.06 (dq, 2H, J ) 3.8, 12.4, H3,
H5), 1.16-1.26 (m, 1H, H4), 1.46 (octet, 1H, J ) 7.3, H8), 1.73-
1.81 (m, 2H, H3, H5), 1.94-2.05 (m, 2H, H2, H6), 2.31 (d, 2H, J
) 13.6, H2, H6), 4.58 (br s, 2H, 2 × H7). 13C NMR (100 MHz):
δ 20.0 (C9, C10), 31.3 (C3, C5), 32.6 (C8), 35.1 (C2, C6), 43.8
(C4), 106.4 (C7), 150.5 (C1). EI-MS (70 eV), m/z (%): 138 [M+]
(51), 123 (45), 109 (25), 95 (100), 81 (53), 67 (59).
1
15 as a colorless oil (0.96 g, 39%). H NMR (400 MHz): δ 0.72
(d, 3H, J ) 7.0, 3 × H9), 1.26-1.46 (m, 3H, H5, H6, H8), 1.68-
1.91 (m, 4H, H4, H5, H6, H8), 1.70 (s, 3H, 3 × H12), 2.29 (tt,
1H, J ) 12.0, 3.4, H7), 2.46 (d, 1H, J ) 4.8, H2), 2.84 (d, 1H, J
) 4.8, H2), 4.67-4.71 (m, 2H, 2 × H11). 13C NMR (100 MHz):
δ 14.2 (C9), 21.1 (C12), 31.6, 32.9 (C5, C6), 33.7 (C4), 40.0 (C8),
42.8 (C7), 51.0 (C2), 60.5 (C3), 108.7 (C11), 149.7 (C10). EI-MS
(70 eV), m/z (%): 166 [M+] (8), 151 (65), 135 (42), 123 (74), 112
(89), 93 (100), 79 (65), 67 (65). IR (cm-1): 3043, 2924, 2856,
1644, 1453, 1370, 889. Anal. calcd for C11H18O: C, 79.46; H, 10.91.
Found: C, 78.91; H, 10.84.
6-Isopropyl-1-oxa-spiro[2.5]octane (8). m-Chloroperbenzoic
acid (77%, 2.84 g 12.7 mmol) was added in portions to a stirred
solution of 7 (1.59 g, 11.5 mmol) in dichloromethane (25 mL) at
0 °C. The mixture was treated with aqueous sodium sulfite (10%,
10 mL) after 15 min. The organic phase was washed with saturated
aqueous potassium carbonate (2 × 10 mL) and brine (10 mL), dried
over MgSO4, and concentrated under reduced pressure. The crude
product was purified by column chromatography on silica gel (5%
ethyl acetate in hexanes) affording a 35:65 cis/trans mixture of 8
(5R)-1-(Hydroxymethyl)-5-isopropenyl-2-methylcyclohex-2-
ene-1-ol (16a,b). Compound 12 was synthesized as previously
described (14). Sulfuric acid (2 M, 20 mL) was added to a stirred
solution of 12 (70 wt %, 4.70 g, 20.1 mmol) in acetonitrile (40
mL). The reaction mixture was after 10 min neutralized with
aqueous sodium hydroxide (6 M), diluted with water (100 mL),
and extracted with ethyl acetate (3 × 100 mL). The combined
organic phases were washed with water (100 mL) and brine (100
mL), dried over MgSO4, and concentrated under reduced pressure.
1
as a colorless oil (1.44 g, 81%). H NMR (400 MHz): δ 0.89 (d,

 Bergstroem, Moa Andresen
Bergstroem, Moa Andresen
 Luthman, Kristina
Luthman, Kristina
 Nilsson, J. Lars G.
Nilsson, J. Lars G.
 Karlberg, Ann-Therese
Karlberg, Ann-Therese