Page 13 of 15
Ple Na es we dJ oo u nr no at l ao df jCu hs et mm i as tr rgy ins
Journal Name
ARTICLE
1
2
3
4
5
6
7
8
9
1
1
1
1
1
1
1
1
1
1
2
2
2
2
2
2
2
2
2
2
3
3
3
3
3
3
3
3
3
3
4
4
4
4
4
4
4
4
4
4
5
5
5
5
5
5
5
5
5
5
6
h).
8
5. Conclusions
Conflicts of interest
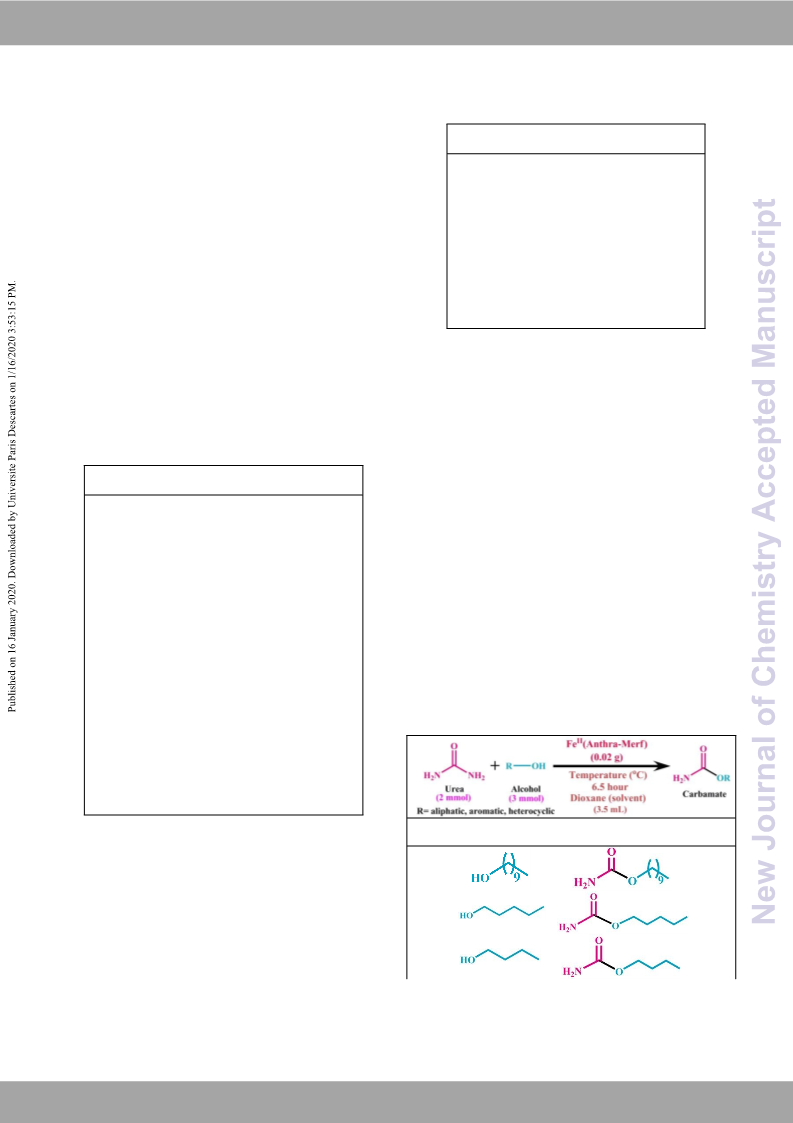
At the end of these present discussions, it can be explored that we
have developed an efficient polymer-based iron catalyst There are no conflicts to declare.
II
Fe (Anthra-Merf) which can effectively promotes upto the
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
satisfactory yield of primary carbamates and N-substituted ureas. Notes and references
The proposed catalytic pathway is inexpensive, convenient as well
II
C. J. nez-Gonzalez, C. S. Ponder, Q. B. Broxterman and
as industrially valuable. The synthesized Fe (Anthra-Merf) catalyst is
thermally stable up to 358 C. Additionally iron being a less
contaminated element, so our developed Fe (Anthra-Merf) catalyst
[
1]
°
J. B. Manley, Org. Process Res. Dev., 2011, 15, 912.
R. Ballini, Eco-Friendly Synthesis of Fine Chemicals,
Royal Society of Chemistry, Oxford, UK 2009.
C. A. M. Afonso and J. G. Crespo, Green Separation
Processes: Fundamentals and Applications, Wiley-VCH,
Weinheim, Germany, 2005.
M. Eissen, J. O. Metzger, E. Schmidt and U.
Schneidewind, Angew. Chem. Int. Ed., 2002, 41, 414.
E. V. Kondratenko, G. Mul, J. Baltrusaitis, G. O.
Larrazábal and J. P. Ramírez, Energy Environ. Sci, 2013,
, 3112.
P. Anastas and N. Eghbali, Chem. Soc. Rev., 2010, 39,
01.
M. Gupta, S. Paul and R. Gupta, Curr. Sci., 2010, 99,
341.
P. Basu, P. Bhanja, N. Salam, T. K. Dey, A. Bhaumik, D.
Das and S. M. Islam, J. Mol. Catal., 2017, 439, 31.
P. Basu, T. K. Dey, A. Ghosh and S. M. Islam, J. Inorg.
Organomet. Polym Mater., 2018, 28, 1158.
S. Badreshia and J. G. Marks Jr, Am. J. of Contact
Dermat., 2002, 13, 77.
T. Goto, Y. Ito, S. Yamada, H. Matsumoto, H. Ok and H.
Nagase, Anal. Chim. Acta, 2006, 555, 225.
J. Ma, N. Lu, W. Qin, R. Xu, Y. Wang and X. Chen,
Ecotoxicol. Environ. Saf., 2006, 63, 268.
II
[2]
is environmentally benign and safe. Concerning with this current
catalytic system, the proposed pathway is highly advantageous due
to mild reaction temperature, less time consuming and high
product yield having immense importance compared to the former
reported system. We have used different alcohols for carbamate
synthesis and it was noticed that alcohols with electron
withdrawing group have more influence in carbamate synthesis as
indicated from the high yield of desired products. In this
connection, aliphatic alcohols are also better candidates for their
corresponding carbamate synthesis. During the synthesis of N-
substituted ureas by taking aniline as a substrate, surprisingly it
produced an isolated product with minute yield compared to the
substituted aromatic and aliphatic amines. The important part of
our catalytic system is that up to six reaction cycles it can be
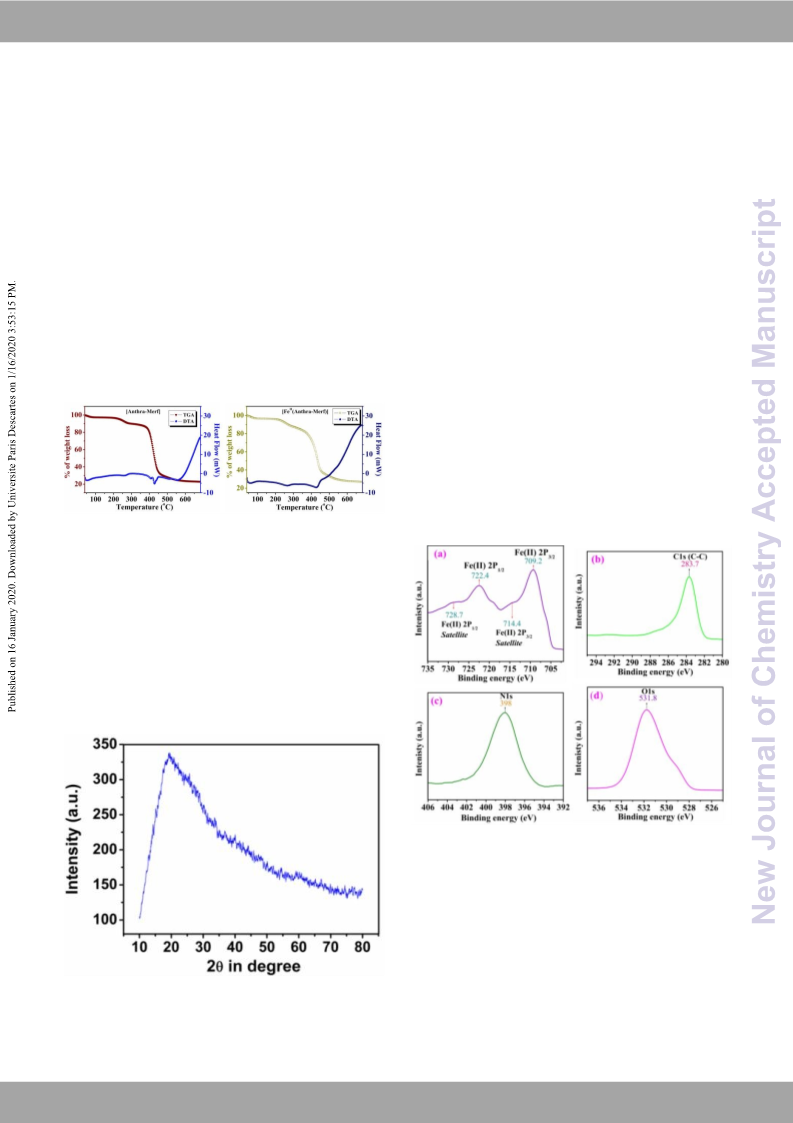
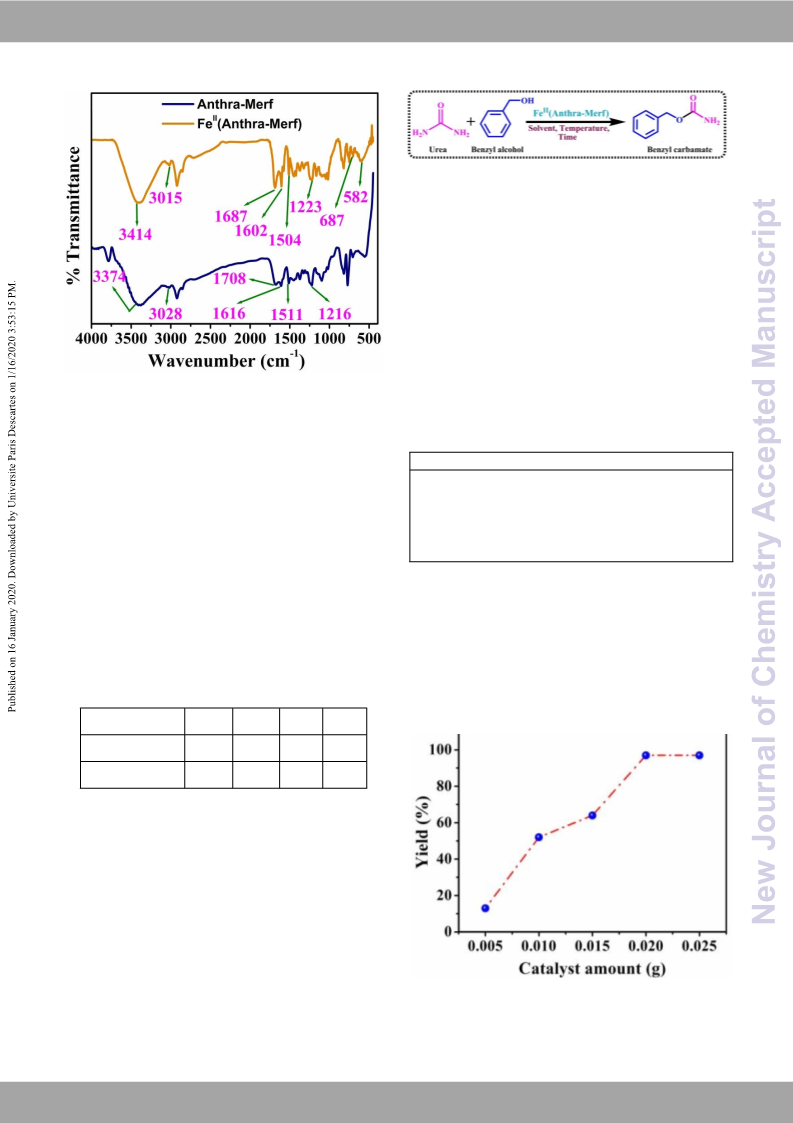
recycled. The characterizations like XRD, FE-SEM, and FTIR
[
[
3]
4]
[5]
6]
[7]
6
[
3
1
[
[
8]
9]
II
instrumental analysis were executed for recycled Fe (Anthra-Merf)
catalyst and no special change in structure or configuration was
examined compared to that of fresh catalyst. From the above
[
[
[
10]
11]
12]
II
discussion and to the best of our delight the developed Fe (Anthra-
Merf) catalyst may explore a new degree of advancement to the
economic synthesis of organic primary carbamates and N-
substituted ureas in industrial scale as well as in the catalysis field.
Acknowledgments
S. Ray, S. R. Pathak and D. Chaturvedi, Drug Future,
[13]
[14]
[15]
2
005, 30, 161.
R. H. Heyn, I. Jacobs and R. H. Carr, Adv. Inorg. Chem.,
014, 66, 83.
O. Kreye, H. Mutlu and M. A. R. Meier, Green Chem.,
S.M.I. acknowledges the Department of Science and Technology
(
DST-SERB, Project No. EMR/2016/004956), New Delhi, Govt. of
2
India, and Board of Research in Nuclear Sciences (BRNS), Project
reference No: 37(2)/14/03/2018-BRNS/37003, Govt. of India, for
funding. PB is thankful to the NFOBC for UGC, New Delhi for her
fellowship. AG is grateful to the SERB-NPDF, DST, New Delhi, India
A. K. Ghosh and M. Brindisi, J. Med. Chem., 2015, 58,
(
File No. PDF/2017/000478) for his National Post Doctoral
[17]
2
895.
Fellowship. SMI is also thankful to Council of Scientific and
Industrial and Research, CSIR (project reference no
02(0284)2016/EMR-II dated 06/12/2016) New Delhi, Govt. of India
and Department of Science and Technology, West Bengal (DST-WB,
Sanction No. 91(sanc.)/ST/P/S&T/4G-8/2014 Dated: 19.09.18) for
providing financial support. A.K. thanks the Researchers Supporting
Project (Grant no.: RSP-2019/127), King Saud University, Riyadh,
Saudi Arabia for the financial support. SMI acknowledges the
University of Kalyani, India for providing personal research grant. SB
acknowledges University Grants Commission for D. S. Kothari Post
Doctoral Fellowship (Award letter no. F.4-2/2006 (BSR)/CH/16-
K. Miller, B. Neilan and D. M. Sze, Recent Pat
Anticancer Drug Discov., 2008, 3, 14.
A. I. Llobet, M. Lvarez and F. Albericio, Chem. Rev.,
009, 109, 2455.
R. A. Watile and B. M. Bhanage, RSC Adv., 2014, 4,
3022.
M. Zhang, X. Zhao and S. Zheng, Chem. Commun.,
014, 50, 4455.
S. L. Peterson, S. M. Stucka and C. J. Dinsmore, Org.
Lett., 2010, 12, 1340.
A. Ion, C. V. Doorslaer, V. Parvulescu, P. Jacobs and D.
D. Vos, Green Chem., 2008, 10, 111.
D. Chaturvedi and S. Ray, Curr. Org. Chem., 2007, 11,
87.
I. D. Inaloo and S. Majnooni, Eur. J. Org. Chem., 2019,
7, 6359.
[18]
[19]
[20]
[21]
[22]
[23]
[24]
[25]
2
2
2
1
7/0026). We sincerely acknowledge the DST and UGC, New Delhi,
Govt. of India, for providing a grant under FIST, PURSE and SAP
program to the Department of Chemistry, University of Kalyani. PB
and TKD are thankful near Dr. Anupam Singha Roy, EBRI, UK for his
impressive support in developing this research work.
9
3
*
Author to whom correspondence should be addressed.
[26] A. R. Sardarian and I. D. Inaloo, RSC Adv., 2015, 5,
This journal is © The Royal Society of Chemistry 20xx
J. Name., 2013, 00, 1-3 | 13
Please do not adjust margins

 Basu, Priyanka
Basu, Priyanka
 Dey, Tusar Kanto
Dey, Tusar Kanto
 Ghosh, Aniruddha
Ghosh, Aniruddha
 Biswas, Surajit
Biswas, Surajit
 Khan, Aslam
Khan, Aslam
 Islam, Sk. Manirul
Islam, Sk. Manirul