Tetrahedron Letters
Using N-substituted-2-amino-4,6-dimethoxypyrimidines
in the synthesis of aliphatic guanidines
⇑
Julian W. Shaw, Laure Barbance, David H. Grayson, Isabel Rozas
School of Chemistry, Trinity Biomedical Sciences Institute, Trinity College Dublin, 152-160 Pearse St., Dublin 2, Ireland
a r t i c l e i n f o
a b s t r a c t
Article history:
The use of 2-chloro-4,6-dimethoxypyrimdine as a tool for the syntheses of substituted guanidines is pre-
sented. This method, that we had previously shown to be very useful for aromatic amines, introduces an
atom economical, cost effective and environmentally safe method for the installation of the guanidine
functionality in aliphatic primary and secondary amines.
Received 15 June 2015
Revised 29 June 2015
Accepted 1 July 2015
Available online 7 July 2015
Ó 2015 Elsevier Ltd. All rights reserved.
Keywords:
Guanidine
N-Substituted-2-amino-4,6-
dimethoxypyrimidines
Aliphatic amines
The need for atom economical, environmentally friendly and
cost effective synthetic methods is a significant driving force in
organic chemistry.1–3 The use of protecting groups is somewhat
in defiance of these principles and in an ideal synthesis their incor-
poration would be avoided.4 However, there are a plethora of
instances when their use is essential.5 The introduction of the
guanidine functional group is one such example where protecting
group chemistry is advised due to the remarkably high polarity of
this functional group. Numerous preparative methods have been
developed to synthesize protected guanidines; however, many of
these methods require toxic reagents, high molecular weight pro-
tecting groups or are relatively low yielding.6 With the prevalence
of guanidine containing molecules throughout the chemical
sciences,7–9 we aimed to design a synthesis that would help with
their facile introduction.
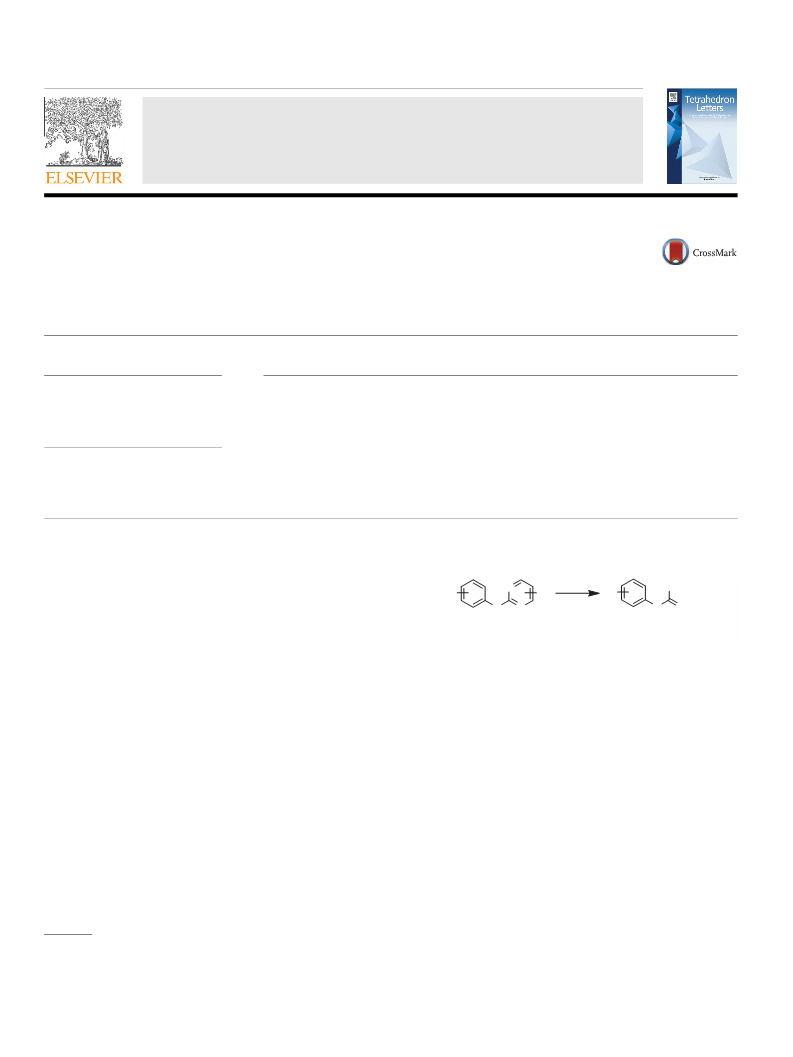
Previously, we have approached the synthesis of aromatic
guanidines10 by the cleavage of appropriately substituted
2-aminopyrimidines, and we found that only one example had
been reported in the literature for a similar type of reaction.11
Our previous study (Scheme 1), that was limited to simple phenyl
systems, made us realize the potential for differently substituted
2-aminopyrimidines to act as synthons for the guanidine
functional group.
NH2
NH
N
R
R
R'
N
H
N
H
N
Scheme 1.
With this knowledge in hand, we planned to explore the suit-
ability of a number of primary and secondary amines attached to
non-aromatic systems as substrates. Additionally, we considered
the reaction with the NH group of the aromatic imidazole system.
Thus, non-aromatic amines 1a–i and imidazole 1j were reacted
with 2-chloro-4,6-dimethoxypyrimidine 2 under basic conditions
to prepare the corresponding N-substituted 2-amino-4,6-
dimethoxypyrimidines 3. All primary, secondary and sterically hin-
dered aliphatic amines as well as imidazole (Table 1, entries 1–10)
provided the N-substituted 2-amino-4,6-dimethoxypyrimidines
3a-j in moderate to excellent yields. With this family of com-
pounds in hand, we then proceeded to optimize the conditions
for the cleavage of the substituted 2-aminopyrimidines 3a–j to
yield the derived guanidine compounds.
An initial screening of acidic conditions, using 1a as a model
substrate, established that the optimal conditions to cleave the
pyrimidine ring, generating the desired guanidines as their
hydrochloride salts was the use of 4 M aqueous HCl at 80 °C. The
different N-substituted 2-amino-4,6-dimethoxy pyrimidines 3a–i,
⇑
Corresponding author. Tel.: +353 1 896 3731.
0040-4039/Ó 2015 Elsevier Ltd. All rights reserved.

 Shaw, Julian W.
Shaw, Julian W.
 Barbance, Laure
Barbance, Laure
 Grayson, David H.
Grayson, David H.
 Rozas, Isabel
Rozas, Isabel