Arkivoc 2019, v, 0-0
Suman, M. et al.
with water (100 mL). The pH of the solution was adjusted to 6.5-7.0 by the addition of aqueous 1N HCl and
stirred at room temperature for 1 h. The solution was filtrated to obtain solid, which was washed with water
(
50 mL) and dried under vacuum to get 9.57 g (95%) of compound 15 as a pale yellow solid. R
f
= 0.5 (SiO
). mp 173-175. IR (Neat) νmax: 3396.9, 3307.3, 3185.8, 2932.2, 2859.9, 2364.3, 2236.0,
678.7, 1612.2, 1564.9, 1509.9, 1412.6, 1331.6, 1310.3, 1266.0, 1194.6, 1110.8, 1052.9, 865.8, 781.9, 620.9,
2
,
1
1
5
2
1
0%MeOH/CH
Cl
2 2
-
1 1
62.1 cm . H NMR (500MHz, DMSO-d6):δ7.92 (brs, 1H), 7.80 (m, 4H), 7.60 (d, J 6.4Hz, 2H), 7.46 (d, J 6.4 Hz,
13
H), 7.30 (brs, 1H), 6.26 (d, J 3.2 Hz, 1H), 5.86 (d, J 2.8 Hz, 1H) ppm. C NMR (125 MHz, DMSO-d6): δ 168.2,
+
51.2, 148.2, 133.7, 132.7, 128.0, 127.6, 126.5, 119.4, 110.1, 73.7 ppm. HRMS (ESI):[M+H] calcd. for
C
4
(
15
H
13
N
2
O
2
253.0977, found 253.0965.
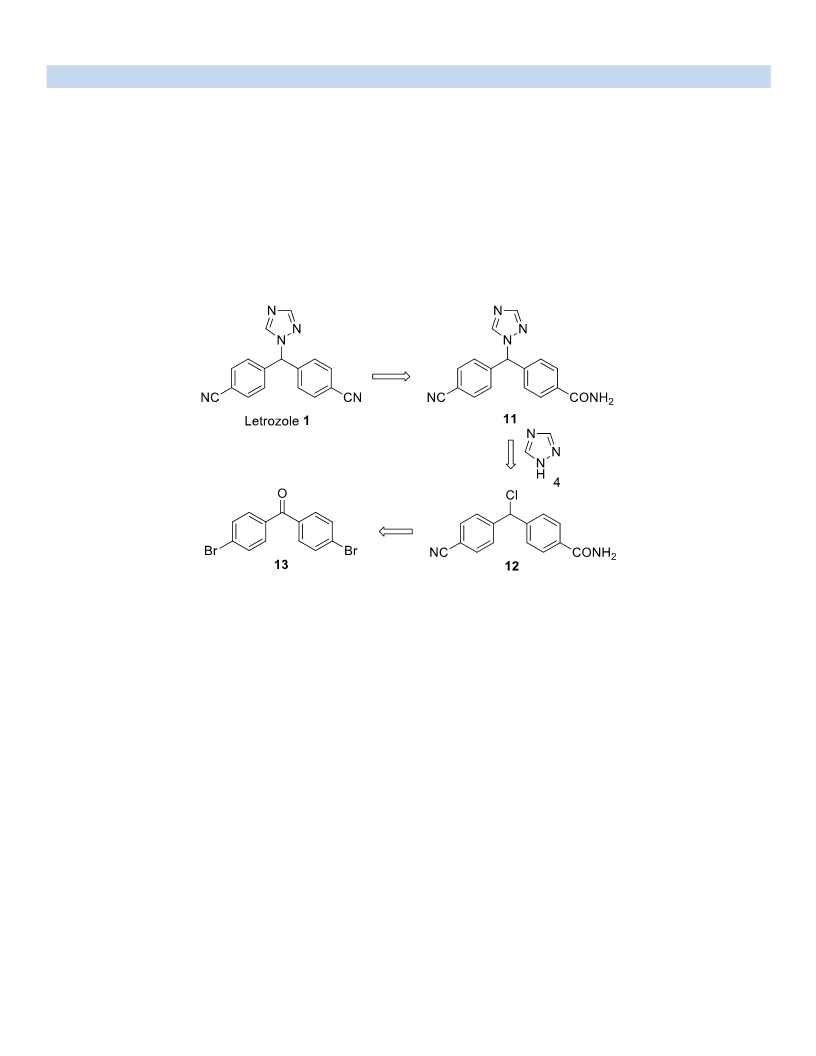
-(chloro(4-cyanophenyl)methyl)benzamide (12). A solution of compound 15 (9.0 g, 35.67 mmol) in CH
90 mL) was added thionyl chloride (21.22g, 178.38mmol) at 0 °C and the solution was warmed to 40 °C and
stirred for 4 h. TLC (2% MeOH/CH Cl ) showed consumption of compound 15. The reaction mixture was
concentrated under reduced pressure at 50°C and diluted with CH Cl (30 mL). Reaction mixture was
neutralized with saturated 10% aqueous NaHCO (30 mL) at 0 °C. The layers were separated and aqueous layer
was extracted with CH CH (2 x 20 mL) twice. The combined organic extracts were washed with water (30 mL)
and aqueous NaCl solution (30 mL) and dried over anhydrous Na SO . The filtrate was concentrated under
reduced pressure to give a crude material, which was purified by silica gel flash column chromatography (SiO
00-200 mesh, 2% MeOH/CH Cl ) to afford 9.06 g (94%) of compound 12 as a white solid. R = 0.6 (SiO , ethyl
acetate); mp 152-156 °C. IR (Neat): νmax: 3399.8, 3342.0, 3604.3, 2944.7, 2749.0, 2223.5, 1810.8, 1728.2,
2
Cl
2
2
2
2
2
3
2
2
2
4
2
,
1
2
2
f
2
-
1
1
1
1
d
C
651.7, 1499.3, 1251.5, 1103.0, 1013.4, 816.7, 755.9, 633.5 cm . H NMR (500MHz, DMSO-d6): δ 8.0 (brs,
H), 7.88 (m, 4H), 7.69 (d, J 6.8 Hz, 2H), 7.48 (d, J 6.8 Hz, 2H), 6.70 (brs, 1H). C NMR (125 MHz, DMSO-
13
+
6
):δ167.4, 145.8, 143.0, 134.3, 132.7, 128.4, 128.0, 127.3, 118.4, 111.0, 61.8. HRMS (ESI): [M+H] calcd. for
15
H
12
N
2
OCl 271.0638, found 271.0629.
-((4-cyanophenyl)(1H-1,2,4-triazol-1-yl)methyl)benzamide (11) and Isomer 4,4'-(4H-1,2,4-triazol-4-
4
ylmethylene)bisbenzonitrile (17). A mixture of compound 12 (5.0 g, 18.46 mmol) and 1H-1,2 4-triazole 4 (3.18
g, 46.17 mmol) was heated under neat conditions at 110 °C-120 °C for 6h. After which time, TLC (10%
MeOH/CH
temperature and diluted with water (20 mL) and extracted with CH
organic extracts were washed with water (30 mL), aqueous NaCl solution (30 mL) and dried over anhydrous
Na SO . The filtrate was concentrated under reduced pressure to give a crude material which was purified by
silica gel column chromatography (SiO , 100-200 mesh, 1% MeOH/CH Cl ) to afford 4.20 g (75%) of compound
. mp: 130-134 °C. IR (Neat):νmax: 3429.7, 2923.0, 1629.0,
2
Cl
2
) showed complete consumption of compound 12. The reaction mixture was cooled to room
2
Cl (2 x 30 mL) twice. The combined
2
2
4
2
2
2
1
1
8
1 as a white solid. R
552.4, 1382.7, 1219.2, 1019.1, 772.3, 678, 6 cm . H NMR (500MHz, DMSO-d
.0 (brs, 1H), 7.88 (dd, J 6.4, 2.4 Hz, 4H), 7.41 (m, 3H), 7.32 (3, 3H), 7.34 (s, 1H), 7.31 (d, 2H). C NMR (125
f
= 0.4 (SiO
2
, 10% MeOH/CH
2
1
Cl
2
-
1
6
): δ 8.71 (brs, 1H), 8.11 (s, 1H),
13
MHz, DMSO-D
HRMS (ESI): [M+H] calcd. for C17
6
) δ: 167.3, 152.2, 144.9, 143.9, 140.9, 134.2, 132.7, 129.0, 128.0, 127.9, 118.4, 110.9, 64.3;
+
H
14
N O 304.1198, found 304.1213
5
Regioisomer 4,4'-(4H-1,2,4-triazol-4-ylmethylene)bisbenzonitrile (17). White solid (0.3 g), mp 138-142 °C, IR
(
7
(
1
Neat):νmax: 3356.2, 3189.4, 2229.8, 1669.4, 1613.5, 1569.1, 1507.4, 1416.7, 1390.7, 1167.9, 1066.6, 862.2,
-
1 1
48.4, 668.3, 622.0, 556.4 cm ; H NMR (500MHz, DMSO-D ):δ8.62 (s, 2H), 8.02 (brs, 1H), 7.91 (m, 4H), 7.41
6
1
3
m, 3H),7.29 (d, J 6.4 Hz,2H), 7.19 (s, 1H). C NMR (125 MHz, CDCl
3
):δ167.2, 143.7, 142.9, 140.7, 134.5, 133.0,
O 304.1198, found 304.1204.
+
28.8, 128.2,127.8, 118.4, 111.3, 61.1. HRMS (ESI): [M+H] calcd. for C17
H
N
14 5
Letrozole (1). A solution of compound 11 (3.0 g, 9.8 mmol) in 1,4-dioxane (3.0 mL) was treated with
trifluoroacetic anhydride (4.15 g, 19.7mmol) at 0 °C and the solution was warmed to room temperature and
stirred for 1h. After which time, TLC (10% MeOH/CH
2
Cl ) showed complete consumption of compound 11. The
2
Page 6
©ARKAT USA, Inc

 Suman
Suman
 Vijayabhaskar
Vijayabhaskar
 NageswaraRao
NageswaraRao
 Syam Kumar
Syam Kumar
 VenkateswaraRao
VenkateswaraRao