Semisynthesis of Rosmanol and Its Derivatives
J ournal of Natural Products, 2002, Vol. 65, No. 7 989
Me-16 and Me-17), 2.00 (1H, td, H-1R), 2.25 (1H, s, H-5), 3.07
(1H, hept, J ) 7.0 Hz, H-15), 3.17 (1H, br d, H-1â), 3.66 (3H,
s, -OCH3), 4.27 (1H, d, J ) 3.18 Hz, H-6), 4.71 (1H, d, J )
3.18 Hz, H-7), 5.48 (1H, br s, Ar-OH), 6.00 (1H, br s, Ar-OH),
6.80 (1H, s, H-14); EIMS m/z 360 [M]+ (100), 314 (81), 298
(80), 284 (88), 269 (84), 245(93), 228 (38), 215 (93); HREIMS
m/z 360.1931 (calcd for C21H28O5 360.1926).
graphed over silica gel with dichloromethane/acetone (98:2)
to obtain 16-hydroxyrosmanol15 (10) (17.0 mg, 43%): 1H NMR
(300 MHz) δ 0.92 (3H, s, Me-19), 1.02 (3H, s, Me-18), 1.32 (3H,
d, J ) 7.0 Hz, Me-17), 2.21 (1H, s, H-5), 3.09 (1H, sext, J )
7.0 Hz, H-15), 3.29 (1H, br d, J ) 14.6 Hz, H-1â), 3.76 (1H,
dd, J 1 ) 2.7 Hz, J 2 ) 9.5 Hz, H-16), 3.99 (1H, dd, J 1 ) 2.7 Hz,
J 2 ) 9.5 Hz, H-16), 4.51 (1H, d, J ) 3.4 Hz, H-6), 4.72 (1H, d,
J ) 3.4 Hz, H-7), 6.37 (1H, s, Ar-OH), 6.69 (1H, s, H-14), 8.98
(1H, s, Ar-OH); EIMS m/z 362 [M]+ (25), 344 (4), 300 (25), 267
(39), 213 (43), 97 (42), 83 (55), 57 (93), 55 (100); HREIMS m/z
362.1694 (calcd for C20H26O6 362.1659).
12,16-Ep oxyca r n osol (11). A solution of triphenylphos-
phine (221.8 mg, 0.846 mmol) in dichloromethane (10 mL) was
added dropwise to a solution of N-bromosuccinimide (145.7 mg,
0.819 mmol) in dichloromethane (7.5 mL), and the mixture
was stirred at room temperature for 5 min. Pyridine (0.03 mL,
0.372 mmol) was then added dropwise to the solution followed
by addition of 16-hydroxycarnosol (9) (93.8 mg, 0.271 mmol).
The reaction mixture was stirred at room temperature for 6
days, then poured into a saturated aqueous sodium bicarbon-
ate solution. The product was extracted with ethyl acetate,
washed with water and brine, and dried over anhydrous
sodium sulfate. The crude product was chromatographed over
silica gel using dichloromethane/acetone (99:1) as eluent to give
11 (59.7 mg, 67%) as a white solid: mp 138-140 °C; UV
(EtOH) λmax (log ꢀ) 209 (1.80), 268 (2.92) nm; IR (film) νmax
3424, 2957, 1739, 1476, 1393, 1339, 1115, 756 cm-1; 1H NMR
(300 MHz, see Table 1); 13C NMR (75 MHz, see Table 1); EIMS
m/z 328 [M]+ (14), 284 (50), 282 (100), 280 (35), 267 (69), 213
(40), 165 (11), 111 (21), 55 (50); HREIMS m/z 328.1647 (calcd
for C20H24O4 328.1675).
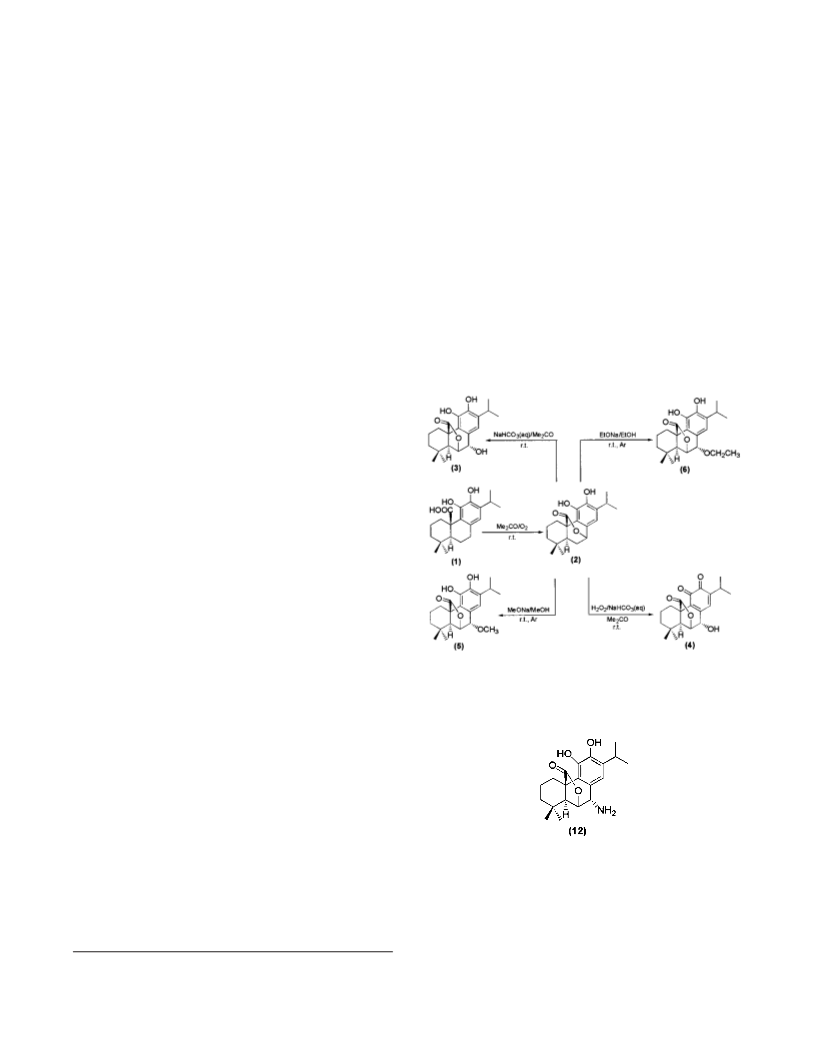
Tr ea tm en t of Ca r n osol (2) w ith Sod iu m Eth oxid e. A
solution of sodium (48.9 mg, 2.13 mmol) in ethanol (10 mL)
was stirred at room temperature under nitrogen. When the
sodium was dissolved, a solution of carnosol (2) (49 mg, 0.148
mmol) in ethanol (5 mL) was added. After 12 h, the reaction
mixture was acidified with dilute hydrochloric acid, the ethanol
was evaporated under reduced pressure, and the product was
extracted with ethyl acetate, washed with brine, and dried over
anhydrous sodium sulfate. The crude reaction product was
chromatographed over silica gel using dichloromethane/
acetone (98:2) as eluent to yield 7-ethoxyrosmanol12 (6) (28.2
mg, 51%): 1H NMR (300 MHz) δ 0.92 (3H, s, Me-19), 1.01 (3H,
s, Me-18), 1.22 (6H, d, J ) 7.0 Hz, Me-16 and Me-17), 1.33
(3H, t, -OCH2-CH3), 1.99 (2H, td, H-1R), 2.28 (1H, s, H-5),
3.11 (1H, hept, J ) 7.0 Hz, H-15), 3.20 (1H, br d, H-1â ), 3.84
(2H, q, -OCH2-CH3), 4.36 (1H, d, J ) 3.20 Hz, H-6), 4.66 (1H,
d, J ) 3.20 Hz, H-7), 5.59 (1H, br s, Ar-OH), 6.15 (1H, br s,
Ar-OH), 6.79 (1H, s, H-14); EIMS m/z 374 [M]+ (100), 328 (87),
300 (29), 231 (24), 215 (68), 69 (40), 55 (51); HREIMS m/z
374.2084 (calcd for C22H30O3 374.2075).
Oxid a tion of Rosm a n ol (3) w ith P yr id in iu m Ch lor o-
ch r om a te. Rosmanol (3) (39.5 mg, 0.11 mmol) in dichlo-
romethane (15 mL) was treated at room temperature and
under magnetic stirring with pyridinium chlorochromate (47.8
mg, 0.22 mmol). After 2.5 h the reaction was completed. The
mixture was filtered through a short pad of Florisil with
suction, the filtrate was evaporated on the rotary evaporator,
and the crude product was chromatographed on silica gel using
n-hexane/dioxane (1:1) as eluent to give in increasing order of
polarity galdosol13 (7) (3.9 mg, 21%) and rosmaquinone12 (4)
(6.1 mg, 32%). Galdosol (7): 1H NMR (300 MHz) δ 0.98 (3H,
s, Me-19), 1.12 (3H, s, Me-18), 1.18 and 1.29 (each 3H, d, J )
7.0 Hz, Me-16 and Me-17), 2.46 (1H, s, H-5), 3.20 (1H, hept, J
) 7.0 Hz, H-15), 4.72 (1H, s, H-6), 7.69 (1H, s, H-14).
Rosmaquinone (4): the spectroscopic data are given above.
Red u ction of Ga ld osol (7) w ith Sod iu m Bor oh yd r id e.
Galdosol (7) (3.9 mg, 0.011 mmol) was dissolved in methanol
(2 mL) and treated with sodium borohydride (19.3 mg, 0.51
mmol). The mixture was stirred at room temperature for 4.5
h, then the mixture was cooled in an ice-bath, acidified with a
solution of 5% hydrochloric acid, extracted with ethyl acetate,
and dried over anhydrous sodium sulfate. The solvent was
removed under reduced pressure, and the crude product was
purified by preparative TLC using n-hexane/ethyl acetate (3:
2) as eluent to yield epirosmanol14 (8) (3.4 mg, 90%): 1H NMR
(300 MHz) δ 0.95 (3H, s, Me-19), 1.00 (3H, s, Me-18), 1.23 (6H,
d, J ) 7.0 Hz, Me-16 and Me-17), 1.97 (1H, s, H-5), 3.08 (1H,
hept, J ) 7.0 Hz, H-15), 3.19 (1H, br d, H-1â), 4.77 (2H,
overlapping signals, H-6 and H-7), 5.50 (1H, br s, Ar-OH), 6.18
(1H, br s, Ar-OH), 7.04 (1H, s, H-14); EIMS m/z 346 [M]+ (24),
300 (9), 284 (34), 231 (38), 215 (100), 199 (43), 14 (44); HREIMS
m/z 346.1779 (calcd for C20H26O5 346.1778).
Ack n ow led gm en t. We thank FEDER (Grant No. IFD97-
0602) and the CYTED Project IV.11 for financial support.
Refer en ces a n d Notes
(1) Chang, H. M.; But, P. P. H. Pharmacology and Applications of Chinese
Materia Medica; World Scientific Publishing: Singapore, 1986; Vol.
1; p 255.
(2) Scott, G. Chem. Br. 1985, 648.
(3) Bergwein, K. Am. Perfum. Cosmet. 1968, 83, 41.
(4) Ikram, M.; Haq, I. Fitoterapia 1980, 51, 231.
(5) Inatani, R.; Nakatani, N.; Fuwa, H. Agric. Biol. Chem. 1983, 47, 521.
(6) Gonza´lez, A. G.; Abad, T.; J ime´nez, I. A.; Ravelo, A. G.; Luis, J . G.;
Aguiar, Z.; San Andre´s, L.; Plasencia, M.; Herrera, J . R.; Moujir, L.
Biochem. Syst. Ecol. 1989, 17, 293.
(7) Moujir, L.; Gutie´rrez-Navarro, A. M.; San Andre´s, L.; Luis, J . G.
Phytochemistry 1993, 34, 1493.
(8) Moujir, L.; Gutie´rrez-Navarro, A. M.; San Andre´s, L.; Luis, J . G.
Phytother. Res. 1996, 10, 172.
(9) Baillie, A. C.; Thomson, R. H. J . Chem. Soc. (C) 1968, 48.
(10) Nibuji, N.; Reiko, I. Agric. Biol. Chem. 1984, 48, 2081.
(11) Gonza´lez, A. G.; Fraga, B. M.; Luis, J . G.; Herrera, J . R.; Ravelo, A.
G. Phytochemistry 1985, 24, 1853.
(12) Gonza´lez, A. G.; San Andre´s, L.; Luis, J . G.; Herrera, J . R.; Ravelo,
A. G. Can. J . Chem. 1989, 67, 208.
(13) Gonza´lez, A. G.; Fraga, B. M.; Luis, J . G.; Ravelo, A. G. Experientia
1971, 29, 1471.
(14) Nakatani, N.; Inatani, R. Agric. Biol. Chem. 1984, 48, 2081.
(15) Luis, J . G.; San Andre´s, L. Phytochemistry 1993, 33, 638.
(16) Luis, J . G.; San Andre´s, L.; Perales, A. Tetrahedron 1993, 49, 4993.
(17) Gonza´lez, A. G.; Rodr´ıguez, C. M.; Luis, J . G. J . Chem. Res. (S) 1988,
114.
(18) Wenkert, E.; Fuchs, A.; McChesney, J . C. J . Org. Chem. 1965, 30,
2931.
(19) Gonza´lez, A. G.; Aguiar, Z. E.; San Andre´s, L.; Luis, J . G. Phytochem-
istry 1992, 31, 1297.
(20) Luis, J . G.; San Andre´s, L.; Quin˜ones, W. Q. Tetrahedron Lett. 1994,
35, 179.
(21) Kakisawa, H.; Hayashi, T.; Yamazaki, T. Tetrahedron Lett. 1969, 301.
(22) Olah, G. A.; Gupta, B. G.; Malhorta, R.; Narang, S. C. J . Org. Chem.
1980, 45, 1638.
Tr ea tm en t of 16-Hyd r oxyca r n osol (9) w ith Aqu eou s
Sod iu m Bica r bon a te. 16-Hydroxycarnosol (9) (39.2 mg, 0.11
mmol) in acetone (5 mL) was treated with aqueous sodium
bicarbonate (5%, 10 mL, 6 mmol) for 15.5 h. The solvent was
evaporated under reduced pressure, the mixture was cooled
in an ice-bath, water was added, and the product was extracted
with ethyl acetate, washed with brine, and dried over anhy-
drous sodium sulfate. The crude reaction was chromato-
(23) Tius, M. A.; Fauq, A. H. J . Am. Chem. Soc. 1986, 108, 1035.
NP010565O

 Marrero, Joaquin G.
Marrero, Joaquin G.
 Andres, Lucia S.
Andres, Lucia S.
 Luis, Javier G.
Luis, Javier G.