
Natural Product Research p. 1598 - 1604 (2016)
Update date:2022-08-16
Topics:
 Tran, Thu Huong
Tran, Thu Huong
 Le Huyen, Tram
Le Huyen, Tram
 Tran, Thi Minh
Tran, Thi Minh
 Nguyen, Tuan Anh
Nguyen, Tuan Anh
 Pham, Thanh Binh
Pham, Thanh Binh
 Nguyen Tien, Dat
Nguyen Tien, Dat
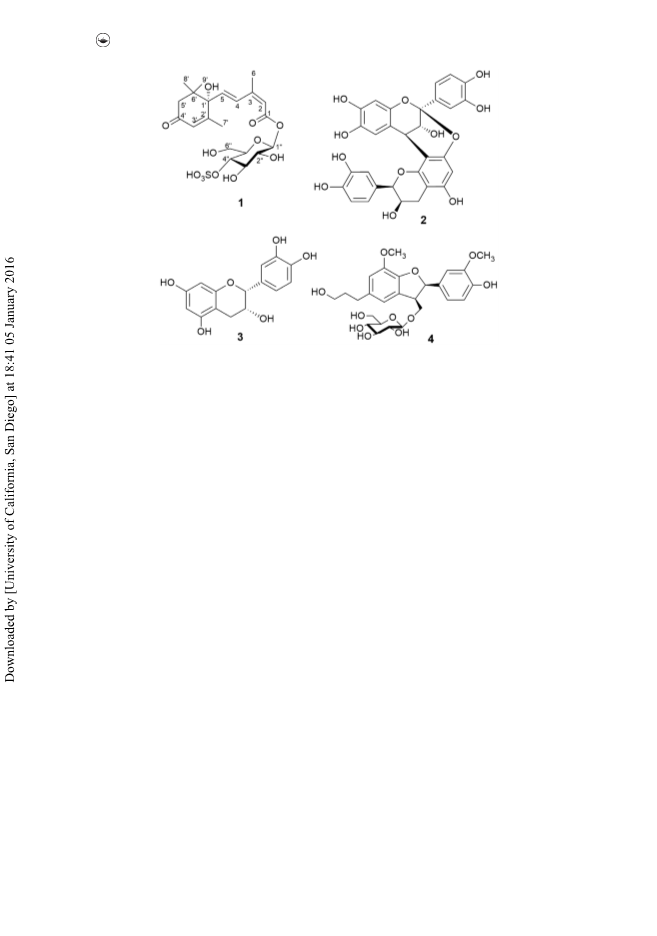
A megastigmane sulphoglycoside together with three phenolic compounds were isolated from the water-soluble fraction of the pericarps of Garcinia mangostana. The structure of the new compound was determined as 4-O-sulpho-β-d-glucopyranosyl abscisate (1) by spectroscopic data. Proanthocyanidin A2 (2) showed potent α-glucosidase inhibitory and DPPH scavenging activities with IC50values of 3.46 and 11.6 μM, respectively.
View More






ClickChem Technology Co., Limited
Contact:+86-0310-6519966/0531-52893837
Address:No.750 Shunhua Road, High-Tech Zone, Jinan city, Shandong China
Frapp's Chemical (NFTZ) Co.,Ltd
Contact:+86-576-86137892
Address:General Chamber of Commercial Building, 159 Wanchang Middle Road, Wenling, Zhejiang, China
VanderArk International Limited
Contact:86-10-82437576
Address:Qing He
shandong zhongke taidou chemical co.,ltd
Contact:86-531-88682301
Address:Jinan shandong Province CHina
Shanghai Dianyang Industry Co.,ltd
Contact:+86 21 6492 4669
Address:Chejing RD, Songjiang District, Shanghai, China
Doi:10.1016/S0040-4020(01)87304-X
(1986)Doi:10.1021/jacs.0c06866
(2020)Doi:10.1021/ja00818a073
(1974)Doi:10.1021/jp302600a
(2012)Doi:10.3390/molecules24122293
(2019)Doi:10.1021/acs.organomet.6b00935
(2017)