Triarylmethane Dye Leuconitriles
J . Org. Chem., Vol. 66, No. 3, 2001 671
143.3, 133.1, 128.3, 128.1, 127.2, 127.0, 125.9, 125.6, 123.3(CN),
119.8, 116.8, 112.6, 111.7, 79.4(CH2), 78.0(C), 40.4, 40.1, 38.3;
IR (KBr) cm-1 2890, 2809, 2224(CN), 1613, 1508, 1360, 1325,
1197, 808; MS (EI, 25 and 70 eV) 412(b), 395, 381, 369(b), 291,
251, 184, 148, 134; MS (CI) 413, 412, 387, 386(b), 374, 373,
370(b), 369, 292, 253, 148. Anal. Calcd for C26H28N4O: C, 75.69;
H, 6.84; N, 13.58. Found: C, 74.98; H, 6.86; N, 13.20;
39.9; IR (KBr) cm-1 2905, 2895, 1664(CO), 1602, 1376, 1189,
818, 720; MS (EI) 239, 148(b), 104, 91, 77; mp ) 163-164 °C.
2-[1′′-(4′′′-N,N-Dim eth yla m in op h en yl)-(2′′-p h en yl)eth -
en yl]-4-(4′-N,N-d im eth yla m in op h en yl)p h en ol (26b): 1H
NMR (200 MHz, CDCl3) δ 7.49 (dd, J 1 ) 8.8 Hz, J 2 ) 2.2 Hz,
1H), 7.39 (d, J ) 9.0 Hz, 2H), 7.31 (d, J ) 9.0 Hz, 2H), 7.32 (d,
J ) 2.2 Hz, 1H), 7.23-7.12 (m, 6H), 6.97 (d, J ) 8.8 Hz, 1H),
6.74 (d, J ) 9.0 Hz, 2H), 6.67 (d, J ) 9.0 Hz, 2H), 2.97 (s, 6H),
2.94 (s, 6H); 13C NMR (APT, DEPT, and 13C, 50 MHz, C6D6) δ
151.6, 150.4, 149.5, 136.8, 136.3, 133.9, 129.1, 128.9, 128.8,
128.7, 128.2, 128.0, 127.20, 127.16, 127.11, 126.6, 126.4, 116.0,
112.9, 112.2, 40.7, 40.4; IR (KBr) cm-1 3430, 2895, 2800, 1604,
1521, 1355, 815; MS (EI) 434, 343, 312, 217, 216, 208, 171,
163, 134(b); HRMS (EI) calcd for C30H30N2O 434.2358, found
434.2349.
λ
max(CHCl3) ) 310 nm, 280 nm; HRMS (EI) calcd for C26H28N4O
412.2263, found 412.2266.
4-N,N-Dim eth yla m in oben zoyl cya n id e (10):89 1H NMR
(200 MHz, CDCl3) δ 7.95 (d, J ) 9.2 Hz, 2H), 6.69 (d, J ) 9.2
Hz, 2H), 3.17 (s, 6H); 13C NMR (APT and 13C, 50 MHz, CDCl3)
δ 164.4(CO), 155.5, 133.0, 121.7, 113.8, 111.2, 40.2; IR (KBr)
cm-1 2926, 2214(CN), 1645, 1605, 1544, 1390, 1293, 1200, 824;
MS (EI) 174, 173(b), 148, 118, 77; mp ) 170-171 °C.
5-[(4′′-N,N-Dim eth yla m in o)-r-cya n oben zyl]-4-(4′-N,N-
4-Ben zyl-4,6-bis(4′-N,N-d im eth yla m in oph en yl)-1-m eth -
yl-1,2-d ih yd r o-3,1,4-ben zoxa zin e (8b): 1H NMR (200 MHz,
CDCl3) δ 7.47 (d, J ) 9.0 Hz, 2H), 7.43 (d, J ) 2.0 Hz, 1H),
7.39 (dd, J 1 ) 8.8 Hz, J 2 ) 2.0 Hz, 1H), 7.36 (d, J ) 8.8 Hz,
2H), 7.15-6.94 (m, 5), 6.81 (d, J ) 9.0 Hz, 2H),), 6.79 (d, J )
8.8 Hz, 1H), 6.67 (d, J ) 8.8 Hz, 2H), 4.35 (d, J ) 9.2 Hz, 1H),
4.25 (d, J ) 9.2 Hz, 1H), 3.69 (d, J ) 13.8 Hz, 1H), 3.38 (d, J
) 13.6 Hz, 1H), 2.98 (s, 6H), 2.93 (s, 6H), 2.58 (s, 3H); 13C NMR
(APT, DEPT, and 13C 50 MHz) CDCl3 δ 149.6, 149.4, 145.2,
137.5, 133.2, 131.5, 131.2, 128.8, 127.2, 127.1, 127.0, 126.3,
125.8, 125.2, 118.0, 113.0, 111.7, 81.9[Ar2(Bn)C-O-], 77.0(N-
CH2-O), 48.8(PhCH2-), 40.8, 40.5, 39.3; MS (EI) 477, 434, 386,
193, 170, 134(b), 91; HRMS (EI) calcd for C32H35N3O 477.2780,
found 477.2771.
d im eth yla m in op h en yl)p yr oca tech ol (14): for X-ray crys-
1
tallography structure report, see ref 54; H NMR (200 MHz,
CD3COCD3) δ 8.12 (s, 2H), 7.11 (d, J ) 8.8 Hz, 2H), 6.99 (d, J
) 8.8 Hz, 2H), 6.84 (s, 1H), 6.78 (d, J ) 8.8 Hz, 2H), 6.73 (s,
1H), 6.68 (d, J ) 8.8 Hz, 2H), 5.21 (s, 1H), 2.96 (s, 6H), 2.90
(s, 6H); 13C NMR (APT, DEPT, and 13C, 50 MHz, CDCl3) δ
150.9, 150.7, 145.5, 145.3, 134.8, 130.7, 128.7, 126.9, 125.3,
121.8, 117.9, 116.0, 113.3, 113.0, 40.5, 40.4, 38.2(CH); IR (KBr)
cm-1 3489, 3257, 2881, 2800, 2255, 1608, 1514, 1441, 1353,
1095, 820; MS (EI) 387(b), 265, 251, 194, 193, 171, 159, 122;
mp ) 247-248 °C (decomp.).
4,4′-Bis(N,N-dim eth ylam in o)dicyan ostilben e (15):90 mix-
ture of E and Z isomers; 1H NMR (400 MHz, CDCl3) δ 7.30 (d,
J ) 9.2 Hz, 4H), 6.56 (d, J ) 9.2 Hz, 4H), 3.01 (s, 12H); 13C
NMR (APT, DEPT, and 13C, 50 MHz, CDCl3) δ 151.2, 130.5,
120.2, 118.544, 118.380, 111.4, 39.9; IR (KBr) cm-1 2902, 2821,
2217, 2208, 1603, 1515, 1371, 1197, 818; MS (EI) 316(b), 315,
301, 286, 229, 157, 149. λmax(MeCN), nm 474, 392, 336, 270,
200; mp ) 293-294 °C.
Bis(4-N,N-d im et h yla m in op h en yl)-(4-N-m et h yl-N-for -
m yla m in op h en yl)a ceton itr ile (18): 1H NMR (200 MHz,
CDCl3) δ 8.51 (s, 1H), 7.30 (d, J ) 8.8 Hz, 2H), 7.12 (d, J ) 8.8
Hz, 2H), 7.05 (d, J ) 9.1 Hz, 4H), 6.66 (d, J ) 9.1 Hz, 4H),
3.31 (s, 3H), 2.94 (s, 12H); 13C NMR (APT, DEPT, and 13C, 50
MHz, CDCl3) δ 162.1(CHO), 149.8, 141.3, 139.8, 129.9, 129.2,
127.6, 123.8, 121.6, 111.9, 55.3, 40.2, 31.8(HCON-Me); IR (KBr)
cm-1 2888, 2804, 2233, 1679, 1608, 1519, 1357, 1112, 813; MS
(EI) 412, 292, 278(b), 262, 251, 206, 190; mp ) 167-169 °C
(decomp.); HRMS (EI) calcd for C26H28N4O 412.2263, found
412.2264.
Bis(4-N ,N -d im e t h yla m in op h e n yl)-(4-N -m e t h yla m i-
n op h en yl)a ceton itr ile (17): 1H NMR (200 MHz, CDCl3) δ
7.06 (d, J ) 9.2 Hz, 4H), 7.02 (d, J ) 9.0 Hz, 2H), 7.64 (d, J )
9.2 Hz, 4H), 6.52 (d, J ) 9.0 Hz, 2H), 2.93 (s, 12H), 2.80 (s,
3H); 13C NMR (APT, DEPT, and 13C, 50 MHz, CDCl3) δ 149.6,
148.5, 129.88, 129.52, 129.35, 129.03, 124.6, 111.90, 111.85,
55.1, 40.4, 30.6 (ArNH-Me); IR (KBr) cm-1 3396, 2884, 2803,
2229, 1609, 1519, 1354, 1184, 817; MS (EI) 384(b), 340, 278,
264(b), 248, 219, 190; mp ) 228-229 °C.
3-[1′-(4′′-N,N-Dim et h yla m in op h en yl)-(2′-p h en yl)et h -
en yl]-N,N,N,N-tetr a m eth ylben zid in e (9b). E and Z isomers
were separated and had identical NMR spectra; 1H NMR (200
MHz, C6D6) δ 7.78 (d, J ) 2.2 Hz, 1H), 7.66 (dd, J 1 ) 8.8 Hz,
J 2 ) 2.2 Hz, 1H), 7.49 (d, J ) 9.0 Hz, 4H), 7.39-7.30 (m, 2H),
7.12-6.85 (m, 5H), 6.55 (d, J ) 9.0 Hz, 2H), 6.54 (d, J ) 9.0
Hz, 2H), 2.57 (s, 6H), 2.50 (s, 6H), 2.48 (s, 6H); 13C NMR (APT,
DEPT, and 13C, 50 MHz, C6D6) δ 150.7, 150.3, 149.7, 143.2,
139.0, 134.6, 132.9, 131.5, 131.0, 129.7, 129.4, 128.24, 128.20,
126.6, 126.3, 124.8, 118.8, 113.4, 112.5, 42.9, 40.3, 40.1; one
isomer: MS (EI) 461, 446, 370(b), 355, 326, 223, 185, 177, 162,
134, 91; the other isomer: MS (EI) 461, 446, 370, 355, 326,
223, 185, 177, 162, 134(b), 91; mp ) 176-178 °C (decomp);
HRMS (EI) calcd for C32H35N3O 461.2831, found 461.2827.
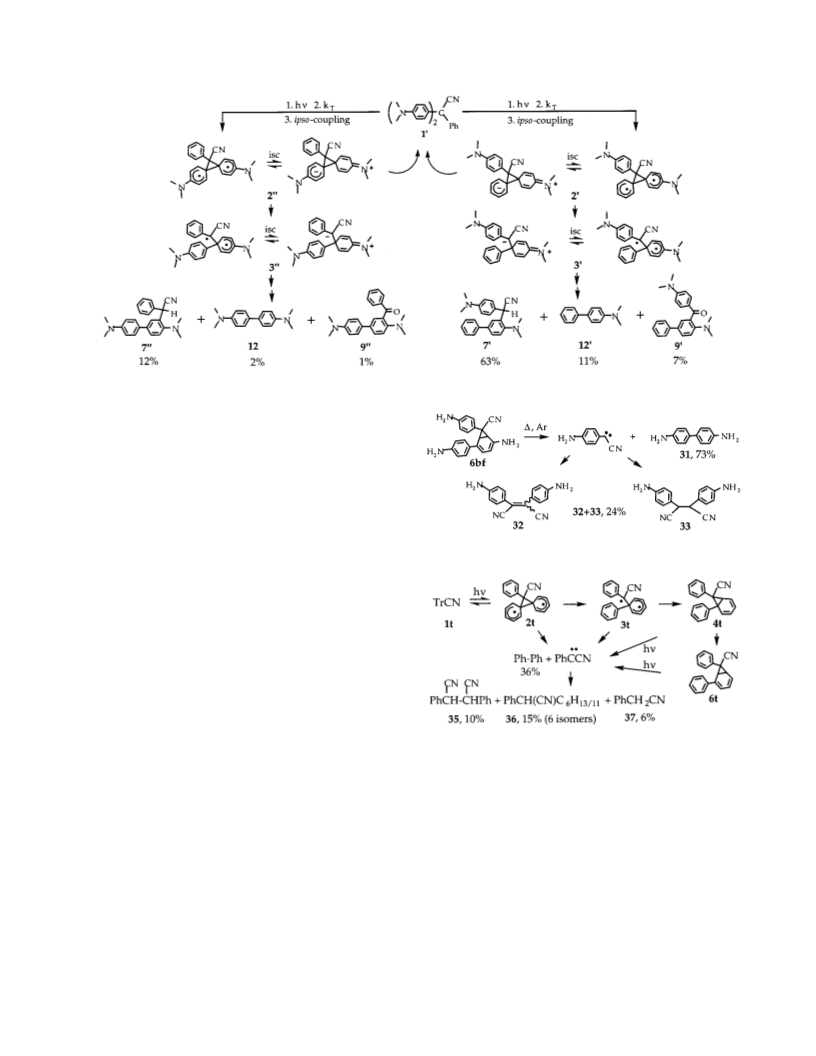
2-[(4′-N,N-Dim et h yla m in o)-r-cya n ob en zyl]-4-p h en yl-
N,N-d im eth yla n ilin e (7′): 1H NMR (200 MHz, CDCl3) δ
7.56-7.16 (m, 7H), 7.44 (dd, J 1 ) 9.2 Hz, J 2 ) 2.2 Hz, 1H),
7.28 (d, J ) 9.2 Hz, 2H), 6.74 (d, J ) 9.0 Hz, 2H), 6.01 (s, 1H),
2.93 (s, 6H), 2.64 (s, 6H); 13C NMR (APT, DEPT, and 13C, 50
MHz, CDCl3) δ 150.2, 149.8, 138.3, 136.5, 132.8, 128.75, 128.74,
127.6, 127.5, 127.4, 126.99, 126.95, 121.9, 120.8, 112.6, 45.8,
40.4, 36.3(CH); MS (EI) 355(b), 340, 313, 310, 269, 178, 170,
156, 117, 91; HRMS (EI) calcd for C24H25N3 355.2048, found
355.2048.
3-(r-Cya n oben zyl)-N,N,N,N-tetr a m eth ylben zid in e (7′′):
1H NMR (200 MHz, CDCl3) δ 7.60 (d, J ) 2.2 Hz, 1H), 7.56-
7.16 (m, 9H), 6.65 (d, J ) 8.8 Hz, 2H), 5.92 (s, 1H), 2.88 (s,
6H), 2.66 (s, 6H); 13C NMR (APT, DEPT, and 13C, 50 MHz,
CDCl3) δ 151.3, 149.8, 140.2, 137.8, 133.4, 128.6, 128.2, 127.5,
127.4, 127.1, 126.9, 123.7, 121.6, 121.3, 112.5, 45.7, 40.3,
35.4(CH); MS (EI) 355, 340, 296, 234, 192, 178, 170, 156,
134(b), 121, 91; HRMS (EI) calcd for C24H25N3 355.2048, found
355.2050.
Ack n ow led gm en t. We thank Drs. George S. Ham-
mond, Michael A. J . Rodgers, Anthony A. Gorman, and
Thomas H. Kinstle for insightful discussions. We are
grateful to Dr. M. A. J . Rodgers for the use of LFP set
up and Drs. A. A. Gorman and Ian Hamblett for the
pulse-radiolysis study. This research was supported by
the McMaster Endowment, the National Science Foun-
dation (DMR-9526755), and the Office of Naval Re-
search (Navy N00014-97-1-0834).
Su ppor tin g In for m ation Available: Tables S1-S3 (yields
of minor products; photophysical parameters of electron ac-
ceptors; observations in SS photolysis), Schemes S1-S8 (trap-
ping with EtSH; irradiation of 1 in CHCl3, 1 and benzophe-
none, 1 and DCA, and 1t; photochemistry of CV radical;
valence tautomerism of 6 and 4; R,R-elimination), Figures S1-
S8 (UV-vis of 1 irradiated in different solvents; kinetics of
31*, 2, and 3; quenching of 1* by oxygen; dark reaction and
thermal bleaching; ORTEP drawings of 1, 9, and 14). This
material is available free of charge via the Internet at
http://pubs.acs.org.
r-P h en yl-(4-N,N-d im eth yla m in o)a cetop h en on e (10b):
91 1H NMR (200 MHz, CDCl3) δ 7.93 (d, J ) 9.2 Hz, 2H), 7.34-
7.14 (m, 5H), 6.63 (d, J ) 9.2 Hz, 2H), 4.18 (s, 2H), 3.03 (s,
6H); 13C NMR (APT and 13C, 50 MHz, CDCl3) δ 195.7(CO),
153.3, 135.7, 130.8, 129.3, 128.5, 126.4, 124.4, 110.6, 44.9(CH2),
J O000374A

 Jarikov
Jarikov
 Neckers
Neckers