Communication
RSC Advances
Reactions were carried out under the optimum reaction would result in the formation of the soluble compound FeCl3,
conditions and the results are summarized in Table 6 (entry 1– which was powerful homogeneous Friedel–Cras catalyst.
5). As expected, the benzoylation of mesitylene proceeded more However, the obtained result indicated that the Friedel–Cras
effectively than m-xylene due to the presence of more electron acylation over Fe2O3/HY would be a well heterogeneously cata-
donating groups. This result was quite consistent with the lysed reaction. It could be due to the strong interaction of Fe2O3
conventional Friedel–Cras acylation reactions.1 However, with HY zeolite, which inhibited the reaction of Fe2O3 with
chlorobenzene was sluggish in the acylation and the conversion benzoyl chloride.
was low since the electron withdrawing nature of chloro group.
Furthermore, Fe2O3/HY was employed in the acylations of m-
xylene with phenylacetyl chloride and 4-chlorobenzoyl chloride,
4. Conclusions
respectively (Table 6, entry 6, 7). It was found that the conver- In summary, iron oxide supported on HY zeolite was found to
sion of benzoyl chloride was 99.5%, while that of 4-chlor- be an efficient, stable and reusable solid acid catalyst for Frie-
obenzoyl chloride was 91.5% at the same reaction conditions. del–Cras acylation reaction and exhibited excellent catalytic
This was mainly due to the synergistic effect of electron with- performance in the acylation of m-xylene with benzoyl chloride.
drawing induction and donating conjugation of chloride atom. 99.5% conversion of benzoyl chloride and 94.5% selectivity of
Moreover, the conversion of phenylacetyl chloride was slightly 2,4-dimethylphenyl-acetophenone were achieved. The catalysts
lower than that of benzoyl chloride. It was attributed to the were characterized by XRD, BET, XPS, NH3-TPD, Py-IR and the
difficulty of forming phenylacetyl cation.
results revealed that the catalytic activity of Fe2O3/HY was
enhanced by the increase of Lewis acidic sites. Furthermore, it
was found that the catalytic activity increased with the SAR
decrease of HY zeolite. The inuences of iron oxide load,
temperature, molar ratio and catalyst dose were investigated
and optimized. The reusability tests of the catalyst showed that
this catalyst can be used ve runs without appreciable loss in
catalytic activity.
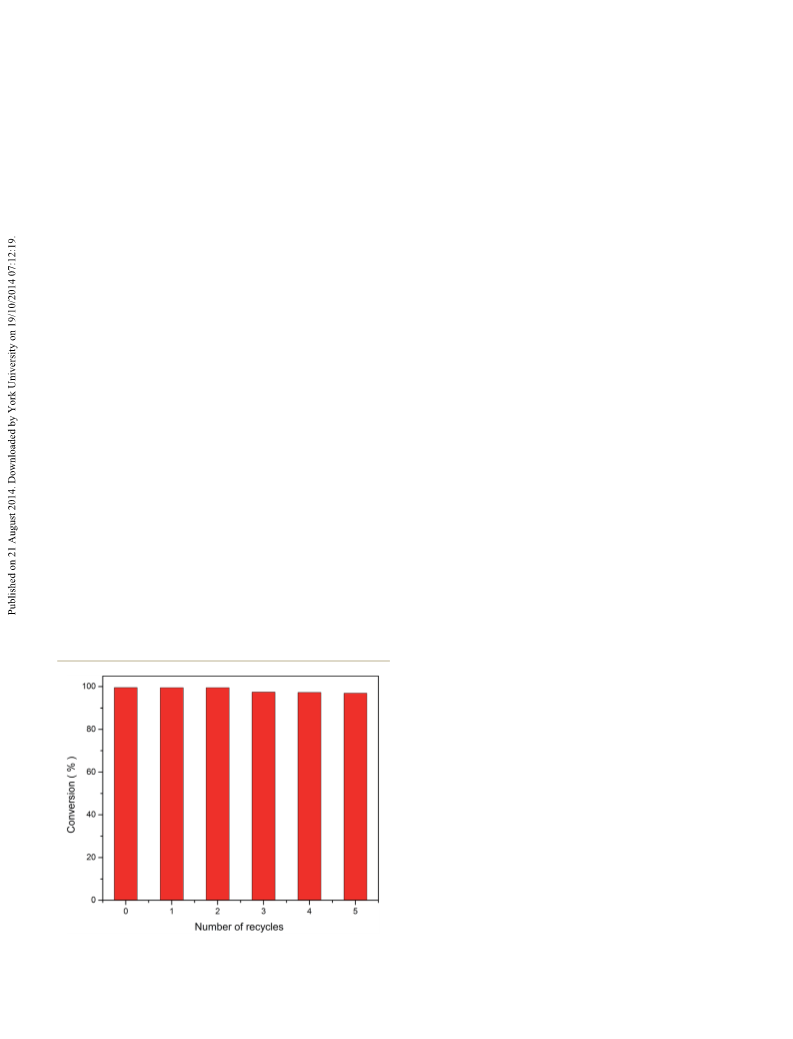
3.6 Reusability of the catalyst
The separation and reusability of catalysts are quite important
for acylations. So the recycling experiments were performed
over Fe2O3/HY and the results are presented in Fig. 7. The
catalyst was separated from the reaction mixture by simple
ltration, washed with ethanol and reused aer drying at 110 ꢁC
for 5 h. It was seen from Fig. 7 that the conversion of benzoyl
chloride displayed a slight drop aer the third run. The
decrease of benzoyl chloride conversion was probably attributed
to the unavoidable trace mechanical loss of catalyst during
work-up procedures. These results indicated that Fe2O3/HY
catalyst exhibited excellent stability, no signicant deactivation
of Fe2O3/HY catalyst was detected during its reuse in the
acylation.
References
1 P. H. Gore, in Aromatic Ketone Synthesis in Friedel–Cras and
Related Reactions, ed. G. A. Olah, John Wiley & Sons Inc.,
London, 1964, vol. III, part 1, p. 1.
2 H. G. Franck, J. W. Stadelhofer, Industrial Aromatic
Chemistry, Springer, Berlin, 1988.
3 S. Gmouh, H. Yang and M. Vaultier, Org. Lett., 2003, 5, 2219–
2222.
4 J. Ross and J. Xiao, Green Chem., 2002, 4, 129–133.
Moreover, the acylation of m-xylene with benzoyl chloride
over Fe2O3 would be a homogeneously catalysed reaction. It
5 D. Das and S. Cheng, Appl. Catal., A, 2000, 201, 159–168.
ˇ
´
´
´
6 M. Bejblova, D. Prochazkova and J. Cejka, ChemSusChem,
2009, 2, 486–499.
´
´ ´
7 J. Farkas, S. Bekassy and B. Agai, Synth. Commun., 2000, 30,
2479–2485.
8 T. Yamato, C. Hideshima and G. K. Surya Prakash, J. Org.
Chem., 1991, 56, 2089–2091.
9 K. Arata, H. Nakamura and M. Shouji, Appl. Catal., A, 2000,
197, 213–219.
10 T. Okuhara, N. Mizuno and M. Misono, Adv. Catal., 1996, 41,
113–252.
11 M. B. Gawande, P. S. Branco and I. D. Nogueira, Green Chem.,
2013, 15, 682–689.
12 R. V. Jagadeesh, A. E. Surkus and H. Junge, Science, 2013,
342, 1073–1076.
13 M. B. Gawande, P. S. Branco and R. S. Varma, Chem. Soc.
Rev., 2013, 42, 3371–3393.
14 B. Chiche, A. Finiels, C. Gauthier, P. Geneste, J. Graille and
D. Pioch, J. Org. Chem., 1986, 51, 2128–2130.
15 V. N. Sheemol, B. Tyagi and R. V. Jasra, J. Mol. Catal. A:
Chem., 2004, 215, 201–208.
Fig. 7 Reusability of Fe2O3/HY catalyst. m-xylene : benzoyl chloride ¼
4 : 1; catalyst dose ¼ 5 wt%; T¼ 130 ꢁC.
This journal is © The Royal Society of Chemistry 2014
RSC Adv., 2014, 4, 36951–36958 | 36957

 Mu, Manman
Mu, Manman
 Chen, Ligong
Chen, Ligong
 Liu, Yunlong
Liu, Yunlong
 Fang, Wangwang
Fang, Wangwang
 Li, Yang
Li, Yang