Chemistry - An Asian Journal
10.1002/asia.201600673
COMMUNICATION
Commun. 2013, 4, 1446; d) Y. Lin, Y. Li, X. Zhan, Chem. Soc. Rev.
2012, 41, 4245-4272; e) R. S. Kularatne, H. D. Magurudeniya, P. Sista,
M. C. Biewer, M. C. Stefan, J. Polym. Sci., Part A: Polym. Chem. 2013,
51, 743-768.
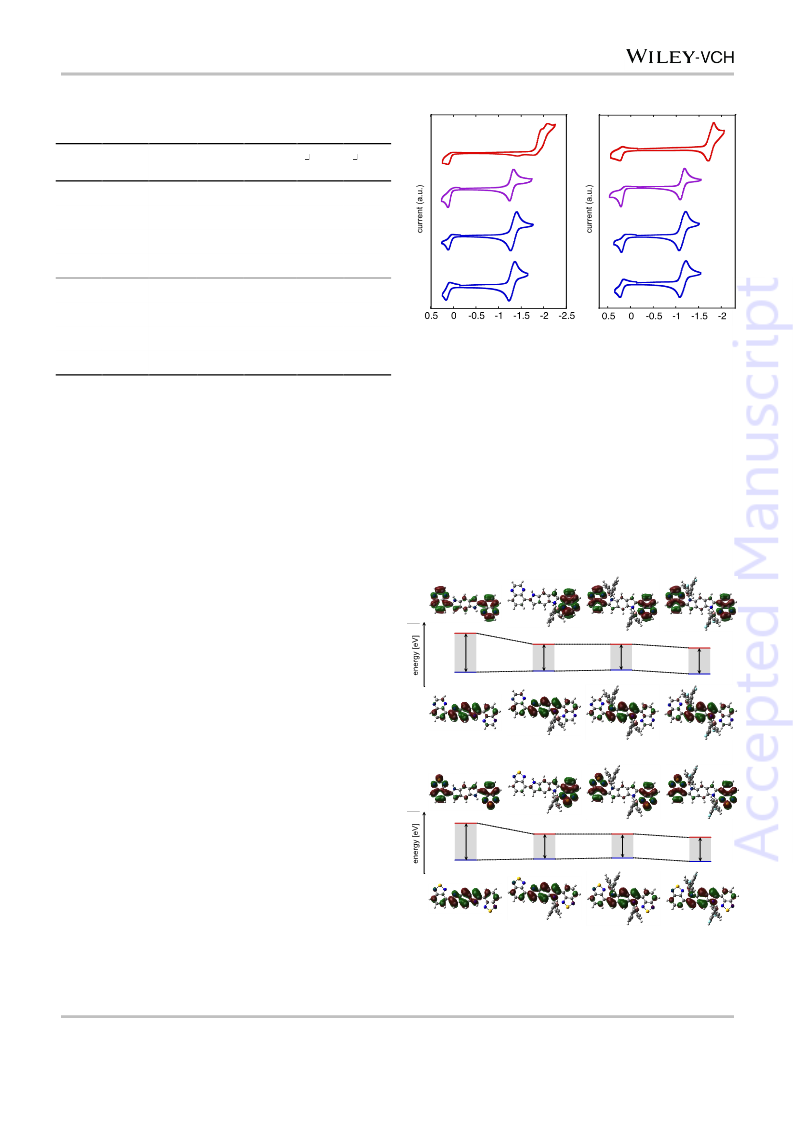
little changed (1.44 eV vs. 1.47 eV), as reflected in their
absorption maxima mentioned above. These trends seen for the
HOMO and LUMO levels are consistent with the experimental
values obtained from the DPV measurements (see above). The
theoretical calculations of BT-based molecules (Fig. 4b) showed
a similar trend to those of the Qx analogues, while the HOMO
and LUMO levels were lower, and the HOMO–LUMO gaps were
a little smaller.
[4]
[5]
a) C. J. Brabec, M. Heeney, I. McCulloch, J. Nelson, Chem. Soc. Rev.
2011, 40, 1185-1199; b) Y.-J. Cheng, S.-H. Yang, C.-S. Hsu, Chem.
Rev. 2009, 109, 5868-5923; c) Z.-G. Zhang, J. Wang, J. Mater. Chem.
2012, 22, 4178-4187.
a) H. Z. Chen, Y. D. Jin, R. S. Xu, B. X. Peng, H. Desseyn, J. Janssens,
P. Heremans, G. Borghs, H. J. Geise, Synth. Met. 2003, 139, 529-534;
b) L. Chunchatprasert, P. V. R. Shannon, J. Chem. Soc. Perkin Trans.
1 1996, 1787-1795; c) F. R. Japp, A. N. Meldrum, J. Chem. Soc. 1899,
1043-1046; d) D. A. Kinsley, S. G. P. Plant, J. Chem. Soc. 1958, 1-7; e)
G. K. B. Prasad, A. Burchat, G. Weeratunga, I. Watts, G. I. Dmitrienko,
Tetrahedron Lett. 1991, 32, 5035-5038.
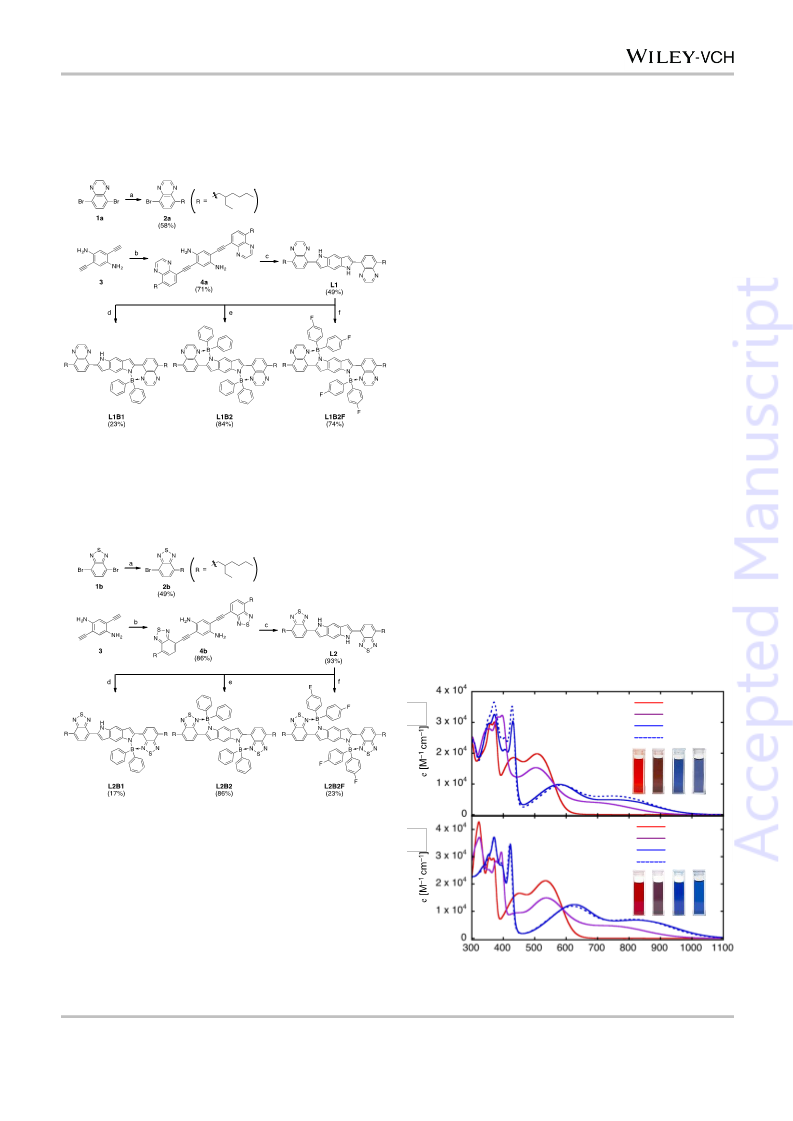
In summary, BDP-based D–A-type boron complexes
represent a new class of NIR-absorbing materials. The BDP
donor unit allows the D–A-type molecules to be further
functionalized to change the photophysical and electrochemical
properties of the compounds. Boron complexation caused both a
significant red-shift of the NIR absorption and lowering of the
LUMO energy levels compared with those of the parent D–A-
type molecules. In addition, further tuning of the absorption
wavelength and energy levels was made possible by changing
the number of boryl substituents and installation of a fluorine
group on the diarylboron atoms.
[6]
[7]
a) H. Tsuji, Y. Yokoi, C. Mitsui, L. Ilies, Y. Sato, E. Nakamura, Chem.
Asian J. 2009, 4, 655-657; b) H. Tsuji, Y. Yokoi, S. Furukawa, E.
Nakamura, Heterocycles 2015, 90, 261-270.
a) A. Wakamiya, T. Taniguchi, S. Yamaguchi, Angew. Chem. 2006, 118,
3242-3245; Angew. Chem., Int. Ed. 2006, 45, 3170-3173; b) A. Job, A.
Wakamiya, G. Kehr, G. Erker, S. Yamaguchi, Org. Lett. 2010, 12, 5470-
5473; c) A. Wakamiya, S. Yamaguchi, Bull. Chem. Soc. Jpn. 2015, 88,
1357-1377; d) Y.-L. Rao, S. Wang, Inorg. Chem. 2011, 50, 12263-
12274; e) D. Li, H. Zhang, Y. Wang, Chem. Soc. Rev. 2013, 42, 8416-
8433; f) G. C. Welch, R. Coffin, J. Peet, G. C. Bazan, J. Am. Chem.
Soc. 2009, 131, 10802-10803; g) G. C. Welch, G. C. Bazan, J. Am.
Chem. Soc. 2011, 133, 4632-4644; h) D. Cruriel, M. Más-Montoya, L.
Usea, A. Espinosa, R. A. Orenes, P. Molina, Org. Lett. 2012, 14, 3360-
3363; i) D. L. Crossley, I. A. Cade, E. R. Clark, A. Escande, M. J.
Humphries, S. M. King, I. Vitorica-Yrezabal, M. J. Ingleson, M. L.
Turner, Chem. Sci. 2015, 6, 5144-5151; j) D. L. Crossley, J. Cid, L. D.
Curless, M. L. Turner, M. J. Ingleson, Organometallics 2015, 34, 5767-
5774.
Acknowledgements
S.F. thanks MEXT for KAKENHI No. 26870144, and E.N. thanks
MEXT for KAKENHI No. 15H05754. We thank K. Inoue for
spectroscopic measurements of film samples.
Keywords: boron • donor–acceptor type molecule • energy level
tuning • heterocycles • near-infrared absorption
[8]
a) T. Bura, N. Leclerc, S. Fall, P. Lévêque, T. Heiser, P. Retailleau, S.
Rihn, A. Mirloup, R. Ziessel, J. Am. Chem. Soc. 2012, 134, 17404-
17407; b) H.-Y. Lin, W.-C. Huang, Y.-C. Chen, H.-H. Chou, C.-Y. Hsu, J.
T. Lin, H.-W. Lin, Chem. Commun. 2012, 48, 8913-8915; c) L. Xiao, H.
Wang, K. Gao, L. Li, C. Liu, X. Peng, W.-Y. Wong, W.-K. Wong, X. Zhu,
Chem. Asian. J. 2015, 10, 1513-1518; d) D. Suresh, C. S. B. Gomes, P.
S. Lopes, C. A. Figueria, B. Ferreira, P. T. Gomes, R. E. D. Paolo, A. L.
Maçanita, M. T. Duarte, A. Charas, J. Morgado, Diogo Vila-Viçosa, M. J.
Calhorda, Chem. Eur. J. 2015, 21, 9133-9149.
[1]
[2]
a) G. Qian, Z. Y. Wang, Chem. Asian J. 2010, 5, 1006-1029; b) J.
Fabian, H. Nakazumi , M. Matsuoka, Chem. Rev. 1992, 92, 1197-1226.
a) E. Bundgaard, F. C. Krebs, Sol. Energy Mater. Sol. Cells 2007, 91,
954-985; b) R. Kroon, M. Lenes, J. C. Hummelen, P. W. M. Blom, B. de
Boer, Polym. Rev. 2008, 48, 531-582; c) M. C. Scharber, D.
Mühlbacher, M. Koppe, P. Denk, C. Waldauf, A. J. Heeger, C. J.
Brabec, Adv. Mater. 2006, 18, 789-794; d) G. Dennler, M. C. Scharber,
T. Ameri, P. Denk, K. Forberich, C. Waldauf, C. J. Brabec, Adv. Mater.
2008, 20, 579-583; e) E. Ay, S. Furukawa, E. Nakamura, Org. Chem.
Front. 2014, 1, 991.
[9]
K. Okuma, J. Seto, K. Sakaguchi, S. Ozaki, N. Nagahora, K. Shioji,
Tetrahedron Lett. 2009, 50, 2943-2945.
[10] S. Hermánek, Chem. Rev. 1992, 92, 325-362.
[11] D. Suresh, C. S. B. Gomes, P. S. Lopes, C. A. Figueira, B. Ferreira, P.
T. Gomes, R. E. Di Paolo, A. L. Maçanita, M. T. Duarte, A. Charas, J.
Morgado, D. Vila-Viçosa, M. J. Calhorda, Chem. Eur. J. 2015, 21,
9133-9149.
[3]
a) J. Peet, J. Y. Kim, N. E. Coates, W. L. Ma, D. Moses, A. J. Heeger,
G. C. Bazan, Nature Mater. 2007, 6, 497-500; b) Y. Sun, G. C. Welch,
W. L. Leong, C. J. Takacs, G. C. Bazan, A. J. Heeger, Nature Mater.
2012, 11, 44-48; c) J. You, L. Dou, K. Yoshimura, T. Kato, K. Ohya, T.
Moriarty, K. Emery, C.-C. Chen, J. Gao, G. Li, Y. Yang, Nature
For internal use, please do not delete. Submitted_Manuscript

 Nakamura, Tomoya
Nakamura, Tomoya
 Furukawa, Shunsuke
Furukawa, Shunsuke
 Nakamura, Eiichi
Nakamura, Eiichi