Organic Letters
Letter
Eur. J. Org. Chem. 2001, 2001, 3975. (d) Han, Y. H.; Zhou, T.; Sui, Y.;
Hua, R. Org. Process Res. Dev. 2014, 18, 1229.
Scheme 9. Conversion of 1 into Congeners 2 and 3
(
6) For the preparation of (R)-10 by the silylation of (S)-glycidol, see:
Donohoe, T. J.; Ironmonger, A.; Kershaw, N. M. Angew. Chem., Int. Ed.
008, 47, 7314. For the preparation of (S)-glycidol by the base-
2
promoted cyclization of (S)-3-chloro-1,2-propanediol, see: Furrow, M.
E.; Schaus, S. E.; Jacobsen, E. N. J. Org. Chem. 1998, 63, 6776.
(7) The concentration of TsOH (0.5 mg/mL in methanol) was
important to achieve the selective removal of the TBS-group.
(
(
8) Frigerio, M.; Santagostino, M. Tetrahedron Lett. 1994, 35, 8019.
9) (a) Kraus, G. A.; Roth, B. J. Org. Chem. 1980, 45, 4825. (b) Bal, B.
S.; Childers, W. E., Jr.; Pinnick, H. W. Tetrahedron 1981, 37, 2091.
10) (a) Tamura, Y.; Yakura, T.; Haruta, J.; Kita, Y. J. Org. Chem. 1987,
2, 3927. For contributions from our laboratory, see: (b) Yasui, Y.;
(
5
Koga, Y.; Suzuki, K.; Matsumoto, T. Synlett 2004, 615. (c) Yasui, Y.;
Suzuki, K.; Matsumoto, T. Synlett 2004, 619. For reviews, see:
(d) Pouysegu, L.; Deffieux, D.; Quideau, S. Tetrahedron 2010, 66, 2235.
(e) Roche, S. P.; Porco, J. A., Jr. Angew. Chem., Int. Ed. 2011, 50, 4068.
(11) Patrick, T. B.; Neumann, J.; Tatro, A. J. Fluorine Chem. 2011, 132,
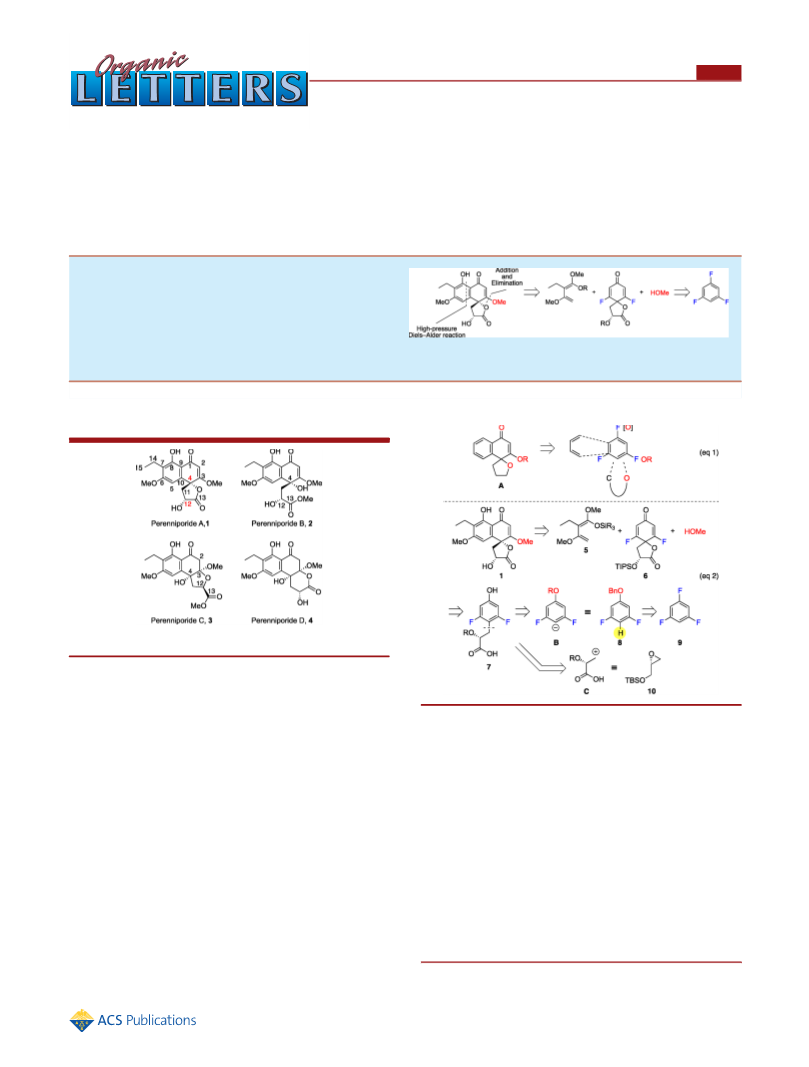
an oxa-Michael addition to give perenniporide C (3) as a
colorless oil. The physical data of the synthetic materials 2 and 3
also coincided with the reported data, respectively.
In summary, we have achieved the first total syntheses of
perenniporide A (1) and the congeners 2 and 3 and their epimers
using difluorodienone 6 derived from 1,3,5-trifluorobenzene (9).
The absolute structure of these natural products was assigned.
1
7
79.
12) (a) Casey, C. P.; Jones, C. R.; Tukada, H. J. Org. Chem. 1981, 46,
089. (b) Anderson, G.; Cameron, G. W.; Feutrill, G. I.; Read, R. W.
Tetrahedron Lett. 1981, 22, 4347.
(
2
(
(
13) Savard, J.; Brassard, P. Tetrahedron 1984, 40, 3455.
14) (a) Grieco, P. A.; Nunes, J. J.; Gaul, M. D. J. Am. Chem. Soc. 1990,
ASSOCIATED CONTENT
Supporting Information
■
*
S
1
(
1
12, 4595. (b) Grieco, P. A. Aldrichimica Acta 1991, 24, 59.
15) Dauben, W. G.; Kessel, C. R.; Takamura, K. H. J. Am. Chem. Soc.
980, 102, 6893. For a review, see: Matsumoto, K.; Hamana, H.; Iida, H.
Helv. Chim. Acta 2005, 88, 2033.
16) The high-pressure reaction was conducted by the super high-
NMR spectra for all new compounds (PDF)
(
pressure apparatus (PVS-1400R, TERAMECS Co., Ltd., Japan) at
RIKEN with the generous support of Prof. Sodeoka and Dr. Hirai.
1
AUTHOR INFORMATION
■
*
*
18) The acid sensitivity of the cycloadduct I was extremely high: it
(
Notes
quickly underwent decomposition upon attempted silica gel chromato-
graphic purification, giving the aromatized product 14 and other
unidentified products.
19) The double reaction did not occur, even with diene 18 (3.0
equiv); only the product 14 was obtained in 65% yield.
(b) Sparling, B. A.; Tucker, J. K.; Moebius, D. C.; Shair, M. D. Org. Lett.
015, 17, 3398. (c) Wipf, P.; Kim, Y.; Goldstein, D. M. J. Am. Chem. Soc.
(
The authors declare no competing financial interest.
(
ACKNOWLEDGMENTS
■
This work was supported by a Grant-in-Aid for Specially
Promoted Research (No. 23000006) from JSPS. We thank Prof.
Oliver Reiser, Mr. Martin Hofmann (Regensburg), Prof. Mikiko
Sodeoka, and Dr. Go Hirai (RIKEN) for helpful discussions and
generous offers to use the high-pressure reactors. A generous
donation of (S)- and (R)-3-chloro-1,2-propanediol from Sanyo
Fine Co. Ltd. is gratefully acknowledged.
2
1
995, 117, 11106.
(
21) If the steric bulkiness of the siloxy group in 6 is considered, the
left-side approach appears to be unfavorable. Nonetheless, however, the
major product 14a is formally arising from the left-side approach,
although the details are not clear at this moment. See also ref 20.
DEDICATION
■
Dedicated to Prof. Dieter Seebach on occasion of his 78th
birthday.
REFERENCES
■
(
2
1) Feng, Y.; Wang, L.; Niu, S.; Li, L.; Si, Y.; Liu, X.; Che, Y. J. Nat. Prod.
012, 75, 1339.
(
2) Caron, B.; Brassard, P. Tetrahedron 1991, 47, 4287.
(
3) (a) Rodriguez, J. R.; Agejas, J.; Bueno, A. B. Tetrahedron Lett. 2006,
4
(
2
(
7, 5661. (b) Seo, H.; Ohmori, K.; Suzuki, K. Chem. Lett. 2011, 40, 744.
c) Stadlbauer, S.; Ohmori, K.; Hattori, F.; Suzuki, K. Chem. Commun.
012, 48, 8425.
4) (a) Bridges, A. J.; Lee, A.; Maduakor, E. C.; Schwartz, C. E.
Tetrahedron Lett. 1992, 33, 7495. (b) Bridges, A. J.; Lee, A.; Maduakor,
E. C.; Schwartz, C. E. Tetrahedron Lett. 1992, 33, 7499. (c) Schlosser, M.
D
Org. Lett. XXXX, XXX, XXX−XXX

 Morita, Masao
Morita, Masao
 Ohmori, Ken
Ohmori, Ken
 Suzuki, Keisuke
Suzuki, Keisuke