990
Can. J. Chem. Vol. 91, 2013
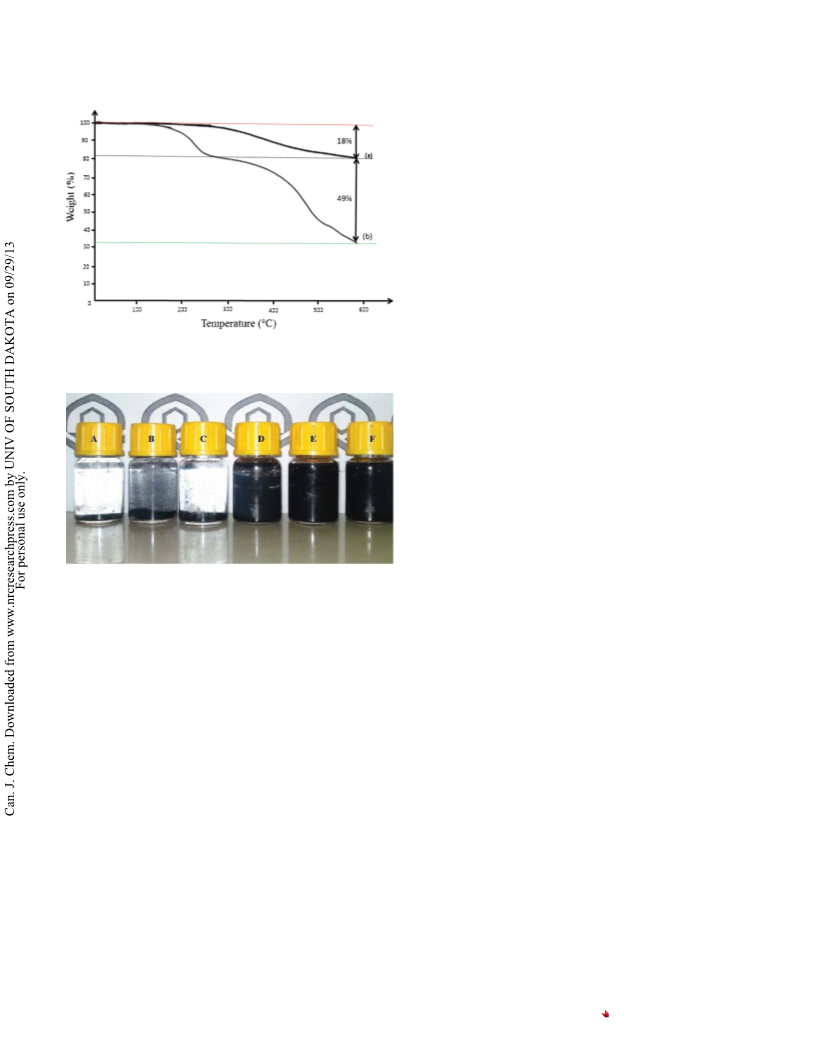
Fig. 6. Reusability of the catalyst in the synthesis of 6,6=-((4-chloro-
-nitrophenyl)methylene)bis(2-(tert-butyl)4-methylphenol) (4c).
␦ (ppm): 1.33 (18H, s, tert-butyl), 2.08 (6H, s, CH ), 6.04 (1H, s,
3
3
Ar CH), 6.30 (2H, s, OH), 6.85 (2H, d, J = 8.34 Hz, 2,6-H aldehyde),
3
6
.90 (2H, s, 3-H TBMP), 7.42 (2H, d, J = 8.32 Hz, 3,5-H aldehyde), 7.55
1
3
(
2H, s, 5-H TBMP). C NMR (125 MHz, DMSO-d ) ␦ (ppm): 20.86,
6
29.84, 34.39, 38.66, 38.94, 39.21, 39.49, 39.77, 40.05, 40.32, 42.60,
1
1
19.00, 125.67, 127.64, 127.69, 130.78, 130.99, 131.18, 137.58, 143.18,
50.57. MS: m/z = 494.18 (M ), 496.18 (M + 2). Anal. calcd. for
+
+
C29H35BrO : C 70.30, H 7.12%; found: C 70.15, H 7.30%.
2
6
,6=-(Phenylmethylene)bis(2-(tert-butyl)4-methylphenol) (Table 2,
compound 4g)
−
1
mp 176–178 °C. IR (KBr) (cm ): 3562, 3495, 3001, 2930, 1870,
1
1443, 1215, 873, 645. H NMR (500 MHz, DMSO-d ) ␦ (ppm): 1.33
6
(
18H, s, tert-butyl), 2.07 (6H, s, CH ), 6.06 (1H, s, Ar CH), 6.33 (2H, s,
3 3
OH), 6.89 (2H, s, 3-H TBMP), 6.92 (2H, d, J = 6.2 Hz, 3,5-H aldehyde),
7
7
2
.17 (1H, t, J = 7.00 Hz, 4-H aldehyde), 7.24 (2H, d, 3,5-H aldehyde),
1
3
.53 (2H, s, 5-H TBMP). C NMR (125 MHz, DMSO-d ) ␦(ppm):
6
0.88, 29.93, 34.38, 38.65, 38.93, 39.21, 39.49, 39.77, 40.05, 40.32,
TBMP), 7.72 (1H, s, 2-H aldehyde), 8.06 (1H, d, J = 7.3 Hz, 4-H alde-
42.99, 125.47, 125.87, 127.41, 127.83, 127.90, 128.99, 131.29, 137.34,
143.46, 150.62. MS: m/z = 416.27 (M ). Anal. calcd. for C29H36O :
1
3
+
hyde). C NMR (125 MHz, DMSO-d ) ␦ (ppm): 20.85, 29.82, 34.40,
6
2
38.65, 38.93, 39.20, 39.48, 39.76, 40.04, 40.32, 42.94, 121.60, 123.18,
C 83.61, H 8.71%; found: C 83.44, H 8.66%.
1
1
7
25.90, 127.76, 127.91, 129.32, 130.77, 135.76, 137.96, 146.43, 147.60,
+
6,6=-(p-Tolylmethylene)bis(2-(tert-butyl)4-methylphenol) (Table 2,
compound 4h)
50.66. MS: m/z = 461.54 (M ). Anal. calcd. for C29H35NO : C 75.46, H
4
.64, N 3.03%; found: C 75.55, H 7.69, N 3.00%.
−
1
mp 195–197 °C. IR (KBr) (cm ): 3542, 3514, 2948, 2916, 1470,
1
6
4
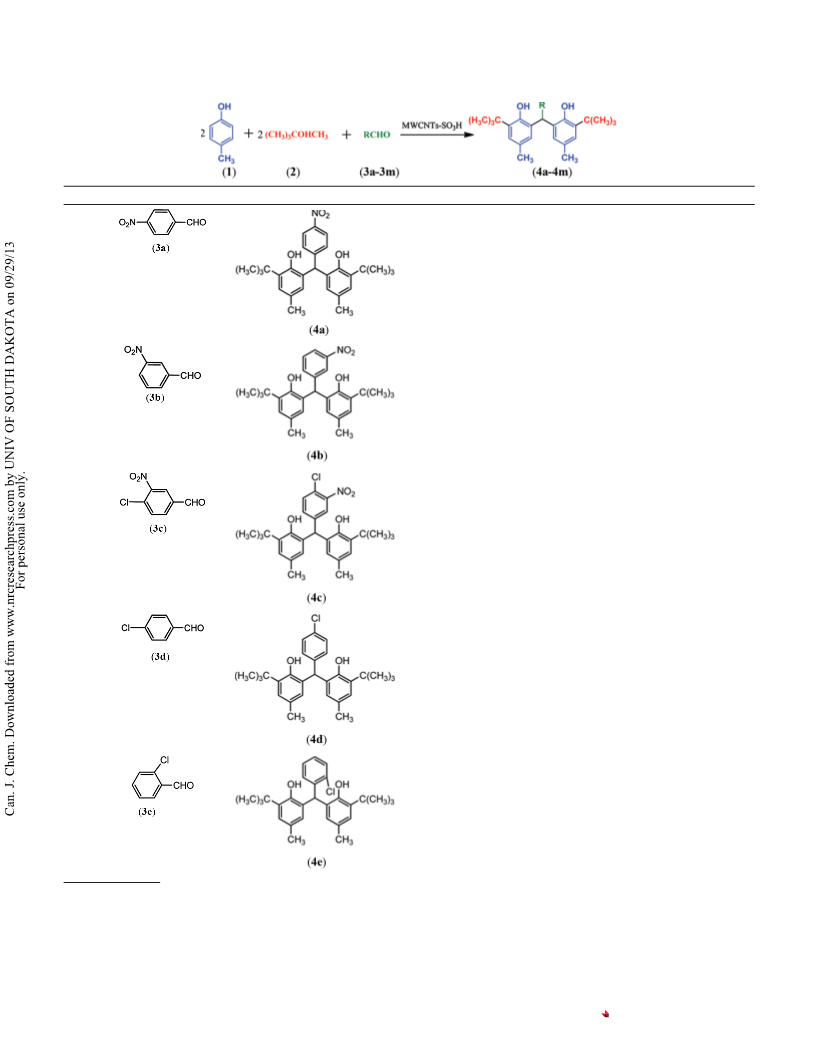
,6=-((4-Chloro-3-nitrophenyl)methylene)bis(2-(tert-butyl)
-methylphenol) (Table 2, compound 4c)
1141, 1320, 1178, 847, 760, 666. H NMR (500 MHz, DMSO-d ) ␦
6
(ppm): 1.33 (18 H, s, tert-butyl), 2.07 (6H, s, CH ), 2.25 (3H, s, CH -
3
3
−1
mp 201–203 °C. IR (KBr) (cm ): 3564, 3522, 3000, 2951, 2914,
aldehyde), 5.99 (1H, s, Ar CH), 6.33 (2H, s, OH), 6.80 (2H, d, J =
3
1
1
530, 1470, 1364, 1185, 1155, 1051, 842, 766, 664, 482. H NMR
7.82 Hz, 3,5-H aldehyde), 6.88 (2H, s, 3-H TBMP), 7.04 (2H, d, J =
1
3
(500 MHz, DMSO-d ) ␦ (ppm): 1.34 (18H, s, tert-butyl), 2.09 (6H, s,
7.82 Hz, 2,6-H aldehyde), 7.48 (2H, s, 5-H TBMP). C NMR (125 MHz,
6
CH ), 6.20 (1H, s, Ar CH), 6.30 (2H, s, OH), 6.94 (2H, s, 3-H TBMP),
DMSO-d ) ␦(ppm): 20.57, 20.90, 29.83, 34.38, 38.66, 38.94, 39.22,
3
3
6
7
.17 (1H, d, J = 7.9 Hz, 6-H aldehyde), 7.52 (1H, s, 5-H aldehyde), 7.63
39.49, 39.77, 40.05, 40.33, 42.59, 125.44, 127.41, 127.78, 128.57,
128.88, 131.39, 134.77, 137.26, 140.30, 150.52. MS: m/z = 430.32 (M ).
1
3
+
(
(
1H, d, J = 1.3 Hz, 2-H aldehyde), 7.64 (2H, s, 5-H TBMP). C NMR
125 MHz, DMSO-d ) ␦ (ppm): 20.84, 29.83, 34.41, 38.66, 38,94,
Anal. calcd. for C30H38O : C 83.67, H 8.89%; found: C 83.64, H
6
2
39.22, 39.49, 39.77, 40.05, 40.33, 42.63, 122.49, 125.62, 126.01,
8.96%.
1
27.71, 128.03, 130.56, 131.10, 134.36, 138.12, 145.53, 147.14, 150.65.
+
+
6,6=-(m-Tolylmethylene)bis(2-(tert-butyl)4-methylphenol) (Table 2,
compound 4i)
MS: m/z = 495.22 (M ), 497.28 (M + 2). Anal. calcd. for C29H34ClNO4:
C 70.22, H 6.91, N 2.82%; found: C 70.55, H 7.09, N 2.85%.
−
1
mp 166–168 °C. IR (KBr) (cm ): 3552, 3524, 2948, 2906, 1466,
1
6
,6=-((4-Chlorophenyl)methylene)bis(2-(tert-butyl)4-methylphenol)
1141, 1318, 1168, 847, 760, 656. H NMR (500 MHz, DMSO-d ) ␦ (ppm):
6
(Table 2, compound 4d)
1.33 (18H, s, tert-butyl), 2.07 (6H, s, CH ), 2.16 (3H, s, CH -aldehyde),
3
3
−1
mp 203–205 °C. IR (KBr) (cm ): 3564, 3517, 2993, 2913, 2861,
6.01 (1H, s, Ar CH), 6.34 (2H, s, OH), 6.65 (1H, d, J = 7.95 Hz, 6-H
3
1
1441, 1410, 1174, 1090,1015, 873, 762. H NMR (500 MHz, DMSO-d ) ␦
aldehyde), 6.90 (2H, s, 3-H TBMP), 6.98 (1H, t, J = 7.60 Hz, 5-H
6
1
3
(ppm): 1.38 (18H, s, tert-butyl), 2.08 (6H, s, CH ), 6.06 (1H, s, Ar CH),
aldehyde), 7.48 (2H, s, 5-H TBMP). C NMR (125 MHz, DMSO-d ) ␦
3
3
6
6
.30 (2H, s, OH), 6.90 (2H, s, 3-H TBMP), 6.92 (2H, d, J = 7.4 Hz, 2,6-H
(ppm): 20.89, 21.06, 29.35, 29.83, 34.07, 34.38, 38.65, 38.93, 39.21,
39.49, 39.77, 40.04, 40.32, 42.95, 125.46, 126.05, 126.66, 126.80,
127.40, 127.77, 127.86, 129.70, 131.28, 136.82, 137.29, 143.38, 150.55,
aldehyde), 7.29 (2H, d, J = 7.4 Hz, 3,5-H aldehyde), 7.54 (2H, s, 5-H
TBMP). C NMR (125 MHz, DMSO-d ) ␦ (ppm): 20.85, 29.83, 34.38,
1
3
6
+
38.65, 38.93, 39.21, 39.49, 39.77, 40.04, 40.32, 42.54, 125.66, 127.63,
153.44. MS: m/z = 430.14 (M ). Anal. calcd. for C30H38O : C 83.67, H
2
1
27.68, 127.86, 130.49, 130.76, 131.04, 137.57, 142.71, 150.58. MS:
8.89%; found: C 83.74, H 8.86%.
+
+
m/z = 450.23 (M ), 452.03 (M + 2). Anal. calcd. for C29H35ClO : C
7
2
6
(
,6=-((3-Hydroxyphenyl)methylene)bis(2-(tert-butyl)4-methylphenol)
Table 2, compound 4j)
mp 196–198 °C. IR (KBr) (cm ): 3544, 3497, 3001, 2957, 2914,
7.22, H 7.82%; found: C 77.55, H 7.59%.
−
1
6
,6=-((2-Chlorophenyl)methylene)bis(2-(tert-butyl)4-methylphenol)
1
(Table 2, compound 4e)
1599, 1444, 1254, 1176, 868, 766, 466. H NMR (500 MHz, DMSO-d )
␦ (ppm): 1.28 (18H, s, tert-butyl), 2.09 (6H, s, CH ), 5.91 (1H, s, Ar CH),
6
−1
mp 193–195 °C. IR (KBr) (cm ): 3577, 3515, 3000, 2962, 2914,
3
3
1
2
866, 1440, 1360, 1182, 1045, 869, 757, 663. H NMR (500 MHz,
6.35 (2H, s, OH), 6.35 (2H, d, J = 7.65 Hz, 2,6-H aldehyde), 6.56 (1H,
d, J = 7.25 Hz, 6-H aldehyde), 6.89 (2H, s, 3-H TBMP), 7.03 (1H, t, J =
7.76 Hz, 5-H aldehyde), 7.49 (2H, s, 5-H TBMP), 9.16 (1H, s, OH
DMSO-d ) ␦(ppm): 1.32 (18H, s, tert-butyl), 2.16 (6H, s, CH ), 6.23 (1H, s,
Ar CH), 6.20 (2H, s, OH), 6.61 (1H, t, J = 6.9 Hz, 4-H aldehyde), 6.92
6
3
3
1
3
(
7
2H, s, 3-H TBMP), 7.20 (2H, m, 5,6-H aldehyde), 7.39 (1H, d, J =
.08 Hz, 3-H aldehyde), 7.46 (2H, s, 5-H TBMP). 13C NMR (125 MHz,
DMSO-d ) ␦ (ppm): 20.91, 29.78, 34.38, 38.64, 38.92, 39.20, 39.48,
aldehyde). C NMR (125 MHz, DMSO-d ) ␦ (ppm): 20.92, 29.85,
6
34.40, 38.63, 38.91, 39.19, 39.47, 39.75, 40.03, 40.30, 42.84, 113.06,
116.15, 119.83, 125.46, 127.44, 127.86, 128.80, 131.30, 137.31, 144.86,
150.52, 152.02. MS: m/z = 432.23 (M ). Anal. calcd. for C29H36O : C
80.52, H 8.39%; found: C 80.74, H 8.26%.
6
+
3
9.76, 40.04, 40.32, 41.06, 125.79, 127.46, 129.83, 137.53, 150.79. MS:
3
+
+
m/z = 450.87 (M ), 452.29 (M + 2). Anal. calcd. for C29H35ClO : C
2
7
7.22, H 7.82%; found: C 77.15, H 7.80%.
6
,6=-Methylenebis(2-(tert-butyl)4-methylphenol) (Table 2,
6
,6=-((4-Bromophenyl)methylene)bis(2-(tert-butyl)4-methylphenol)
compound 4m)
−
1
(Table 2, compound 4f)
mp 129–131 °C. IR (KBr) (cm ): 3533, 3490, 3004, 2957, 2904,
−1
1
mp 210–212 °C. IR (KBr) (cm ): 3560, 3517, 3026, 2944, 2862,
1599, 1444, 1214, 1176, 868, 766, 466. H NMR (500 MHz, DMSO-d ) ␦
(ppm): 1.30 (18H, s, tert-butyl), 2.11 (6H, s, CH ), 5.84 (2H, s, Ar CH ),
6
1
1441, 1251, 1216, 1175, 1010, 873, 825, 424. H NMR (500 MHz, DMSO-d )
6
3
2
2
Published by NRC Research Press

 Fareghi-Alamdari, Reza
Fareghi-Alamdari, Reza
 Golestanzadeh, Mohsen
Golestanzadeh, Mohsen
 Agend, Farima
Agend, Farima
 Zekri, Negar
Zekri, Negar