F. Bigi et al. / Tetrahedron 56 (2000) 2709±2712
2711
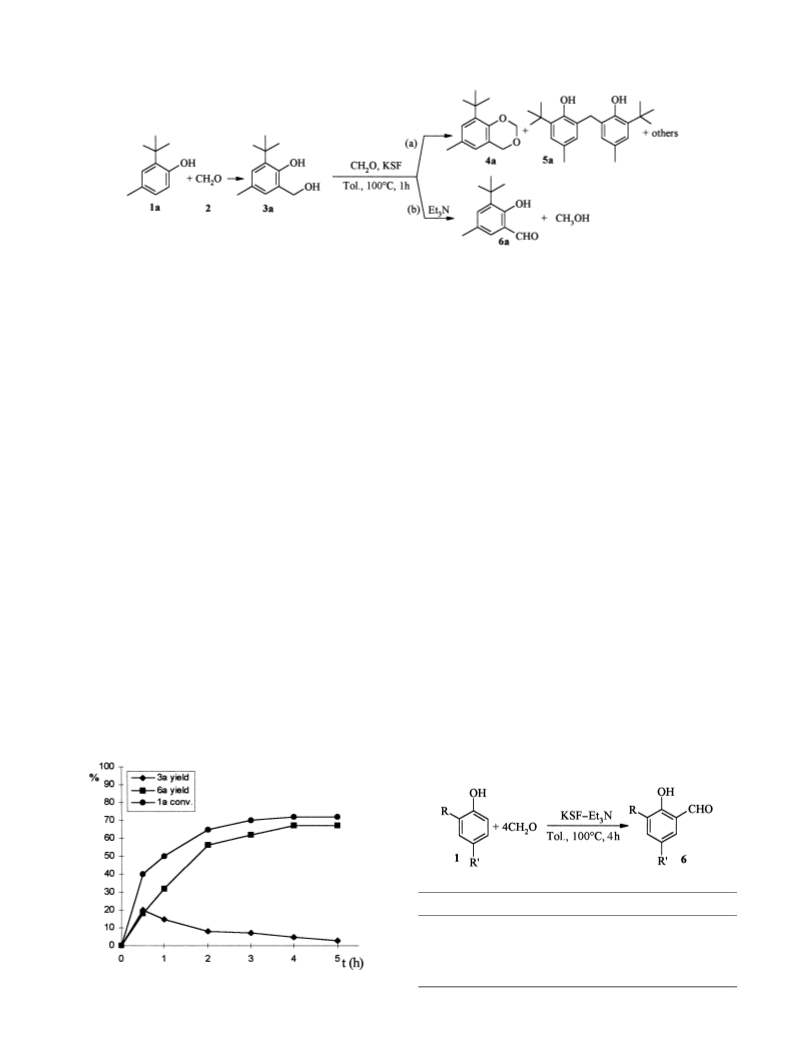
The formylation process was then extended to different
phenols. Interestingly, with monosubstituted phenols
[RCH3, (CH3)3C; R0H] the formylation exclusively
occurred at the ortho position (Table 1).
3-tertButyl-salicylic aldehyde (6d). Pale yellow oil, bp
247±248.58C (lit13a bp 2488C).
3-Methyl-5-decyl-salicylic aldehyde (6e). Pale yellow oil,
bp 37±388C/0.07 mm Hg; H NMR (300 MHz, CDCl3) d
1
The ortho-regioselectivity of the process could be ration-
alised, assuming the formation of an activated complex
resulting from an H-bond between formaldehyde and the
OH group of the phenol co-ordinated to the catalyst
surface.17
0.80 (3H, t, J6.6 Hz, CH3CH2), 1.19 (14H, br s, 7 CH2),
1.5 (2H, m, CH2CH2Ar), 2.17 (3H, s, CH3Ar), 2.47 (2H, t,
J7.7 Hz, CH2Ar), 7.08 (1H, d, J2.0 Hz, H-6), 7.13 (1H,
br s, H-4), 9.76 (1H, s, CHO), 11.01 (1H, s, OH); IR (NaCl)
3103 (OH), 1654 (CvO) cm21; MS m/z (M1) 276 (15%),
149 (100%), 41 (13%). Anal. calcd for C18H28O2: C, 78.2;
H, 10.2. Found: C, 78.1; H, 10.3.
Finally, we faced the problem of the catalyst recycle. After
®ltration and washing with methanol, the montmorillonite
KSF was effective only for one cycle, promoting the model
reaction in 36% yield.
Acknowledgements
In conclusion, the discovery and development of this
catalytic reaction have led to a new method for the direct
formylation of phenols in good yield and high selectivity
under environmentally friendly conditions.
Á
The authors thank the Ministero dell'Universita e della
Ricerca Scienti®ca e Tecnologica (MURST), Italy, the
Consiglio Nazionale delle Ricerche (CNR), Italy and the
University of Parma (National Project Stereoselezione in
Sintesi Organica. Metodologie ed Applicazioni) for ®nan-
cial support. The authors are also grateful to the Centro
Á
Interfacolta Misure (C.I.M.) for the use of NMR and Mass
Experimental
instruments and to Mr Pier Antonio Bonaldi for technical
assistance.
Melting and boiling points were obtained on an Electro-
thermal melting point apparatus and are uncorrected. IR
spectra were recorded on a Nicolet PC5 spectrophotometer.
1H NMR spectra were recorded on a Bruker AC300 at
300 MHz. Chemical shifts are expressed in ppm relative
to TMS as internal standard. Mass spectra were obtained
on a Hewlett±Packard HP-5971 A instrument in EI mode
at 70 eV. Microanalyses were carried out by Dipartimento
di Chimica Generale ed Inorganica, Chimica Analitica,
References
1. (a) Lehnert, V. Tetrahedron 1972, 28, 663. (b) Nore, P.,
Honkanen, E. J. Heterocyclic Chem. 1980, 17, 985. (c) Kirk-
Othmer, Encyclopedia of Chemical Technology, 3rd ed., John
Wiley & Sons: New York, 1979; Vol. 7, pp 196±206. (d) Tietze,
L. F.; Beifuss, U. The Knoevenagel Reaction. In Comprehensive
Organic Synthesis, Trost, B. M., Fleming, I., Heathcock, C. H.,
Eds.; Pergamon Press: Oxford, 1991; Vol. 2, pp 341±394.
2. Burgstahler, A. W.; Worden, L. R. Org. Synth. 1966, 46, 28.
3. For the recent application of salen-type ligands see: (a) Larrow,
J. F.; Schaus, S. E.; Jacobsen, E. N. J. Am. Chem. Soc. 1996, 118,
7420. (b) Quan, R. W.; Li, Z.; Jacobsen, E. N. J. Am. Chem. Soc.
1996, 118, 8156. (c) Punniyamurthy, T.; Reddy, M. M.; Singh
Kalra, S. J.; Iqbal, J. Pure Appl. Chem. 1996, 68, 619.
Á
Chimica Fisica dell'Universita di Parma. TLC analyses
were performed on Merck 60 PF254 silica gel plates using
mixtures of hexane±ethyl acetate (5±10%). All the reagents
were of commercial quality from freshly opened containers.
Montmorillonite KSF (Fluka) was heated at 1308C for 10 h
before use.
Synthesis of salicylic aldehydes 6. General procedure
4. (a) Shuttleworth, R.; Fielden, J. M.; Levin, D. Eur. Pat. Appl.
EP536,960 1993 (C.A. 1993, 119, 116962v). (b) Levin, D. Eur.
Pat. Appl. EP529,870, 1993 (C.A. 1993, 119, 95098a).
A solution of the selected phenol (10 mmol), paraformalde-
hyde (40 mmol, 1.2 g), triethylamine (10 mmol, 1.0 g,
1.4 ml) and montmorillonite KSF (1.5 g) in toluene (5 ml),
was heated at 1008C in a small autoclave under ef®cient
stirring. After 4 h the reaction mixture was cooled to rt,
the catalyst was ®ltered and washed with boiling methanol
(3£50 ml); the solvents were distilled off and the crude was
puri®ed by ¯ash chromatography using as eluant mixtures
of hexane/ethyl acetate (5±10%). All the known products
(6a±d) gave spectral data consistent with the reported ones.
5. (a) Effenberger, F. Angew. Chem., Int. Ed. Engl. 1980, 19, 151.
(b) Olah, G. A.; Ohennesian, L.; Arvanaghi, M. Chem. Rev. 1987,
87, 671.
6. Casiraghi, G.; Casnati, G.; Puglia, G.; Sartori, G.; Terenghi,
M. G. J. Chem. Soc., Perkin Trans. 1 1980, 1862.
7. Furniss, B. S.; Hannaford, A. J.; Smith, P. W. G.; Tatchell, A. R.
Vogel's-Textbook of Practical Organic Chemistry, Wiley: New
York, 1989 (p 989).
8. See for example: (a) Balogh, M.; Laszlo, P. Organic Chemistry
Using Clays; Springer Verlag: New York, 1993. (b) Corma, A.
Chem. Rev. 1995, 95, 559. (c) Sheldon, R. A. Chemtech 1991,
566. (d) Arends, I. W. C. E.; Sheldon, R. A.; Wallan, M.;
Schuchardt, U. Angew. Chem., Int. Ed. Engl. 1997, 36, 1145.
(e) Clark, J. H.; Macquarrie, D. J. Chem. Soc. Rev. 1996, 303.
(f) Clark, J. H. Catalysis of Organic Reactions Using Supported
Inorganic Reagents; VCH: New York, 1994.
3-tertButyl-5-methyl-salicylic aldehyde (6a). Pale yellow
oil, bp 110±1128C/7 mm Hg (lit6 bp 128±1308C/
16 mm Hg).
3,5-Ditertbutyl-salicylic aldehyde (6b). Pale yellow solid,
mp 54±568C (lit18 mp 54±568C).
3-Methyl-salicylic aldehyde (6c). Pale yellow oil, bp 210±
2128C (lit13a bp 2118C).
9. (a) Sheldon, R. A.; Dakka, J. Catal. Today 1994, 19, 215.
(b) Chemistry of Waste Minimisation, Clark, J. H., Ed.; Chapman

 Bigi, Franca
Bigi, Franca
 Conforti, Maria Lina
Conforti, Maria Lina
 Maggi, Raimondo
Maggi, Raimondo
 Sartori, Giovanni
Sartori, Giovanni