
ACS Catalysis p. 10375 - 10380 (2020)
Update date:2022-08-17
Topics:
 Grimm, Christopher
Grimm, Christopher
 Lazzarotto, Mattia
Lazzarotto, Mattia
 Pompei, Simona
Pompei, Simona
 Schichler, Johanna
Schichler, Johanna
 Richter, Nina
Richter, Nina
 Farnberger, Judith E.
Farnberger, Judith E.
 Fuchs, Michael
Fuchs, Michael
 Kroutil, Wolfgang
Kroutil, Wolfgang
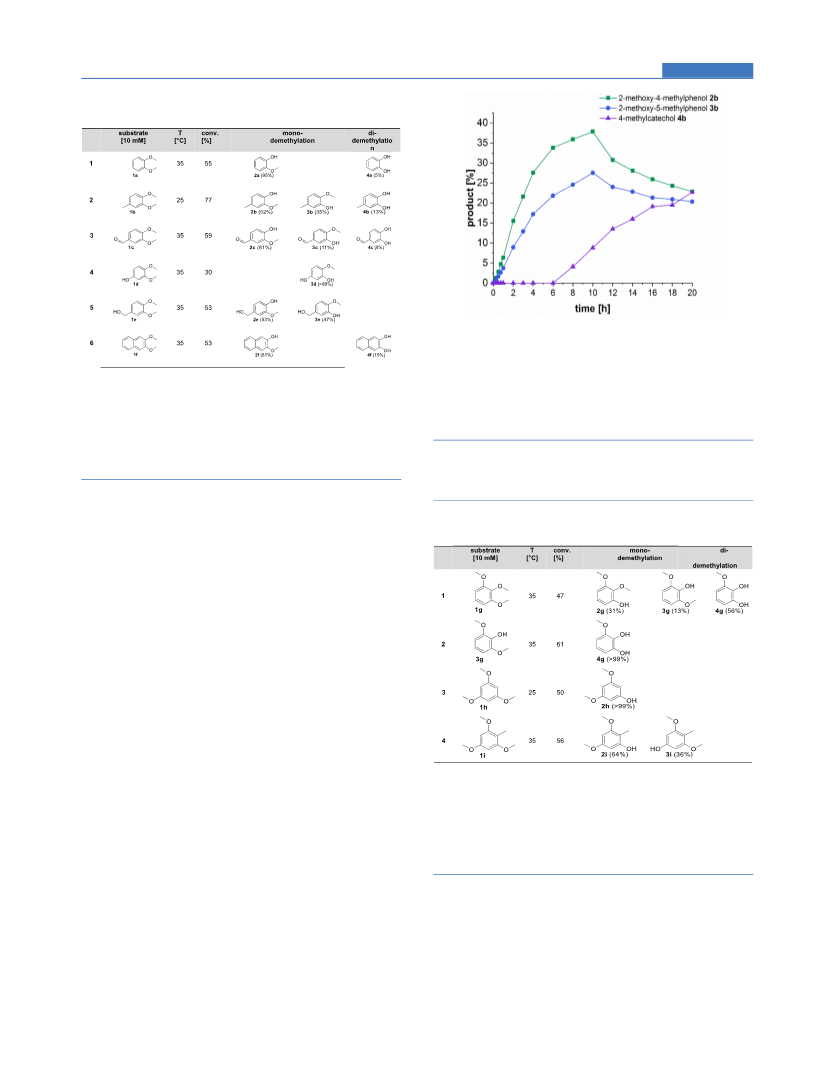
The cleavage of aryl methyl ethers is a common reaction in chemistry requiring rather harsh conditions; consequently, it is prone to undesired reactions and lacks regioselectivity. Nevertheless, O-demethylation of aryl methyl ethers is a tool to valorize natural and pharmaceutical compounds by deprotecting reactive hydroxyl moieties. Various oxidative enzymes are known to catalyze this reaction at the expense of molecular oxygen, which may lead in the case of phenols/catechols to undesired side reactions (e.g., oxidation, polymerization). Here an oxygen-independent demethylation via methyl transfer is presented employing a cobalamin-dependent veratrol-O-demethylase (vdmB). The biocatalytic demethylation transforms a variety of aryl methyl ethers with two functional methoxy moieties either in 1,2-position or in 1,3-position. Biocatalytic reactions enabled, for instance, the regioselective monodemethylation of substituted 3,4-dimethoxy phenol as well as the monodemethylation of 1,3,5-trimethoxybenzene. The methyltransferase vdmB was also successfully applied for the regioselective demethylation of natural compounds such as papaverine and rac-yatein. The approach presented here represents an alternative to chemical and enzymatic demethylation concepts and allows performing regioselective demethylation in the absence of oxygen under mild conditions, representing a valuable extension of the synthetic repertoire to modify pharmaceuticals and diversify natural products.
View More




Shanghai Mokai Pharmaceutical Co.,Ltd
Contact:021-60257269
Address:Rm506,No.915,Zhenbei Road,Shanghai,200333,China
Zhejiang Quzhou Jiancheng Silicone Co., Ltd.(Shanghai Jiancheng Industial and Trade Co, Ltd)
Contact:18957018777 +86-570-3888777
Address:The company production base address: Quzhou City, Zhejiang Province high-tech industrial park Nianhua Road 37
SHAANXI FUJIE PHARMACEUTICAL CO.,LTD
website:http://www.fujiepharm.com
Contact:+86-29-63650906
Address:Yuanqu Yi Road, Qinghe Food Industrial Park, Sanyuan County, Shaanxi Province, China
Weifang Arylchem Chemical Co., LTD
Contact:86-536-5217866
Address:Development Zone, Shouguang, Shandong Province
Contact:+86 21 34123252
Address:14, 4580 Dushi, Shanghai, China
Doi:10.1016/j.jcat.2006.07.015
(2006)Doi:10.1039/c9ra06620a
(2019)Doi:10.1021/acs.orglett.5b01224
(2015)Doi:10.1016/S0021-9517(03)00184-2
(2003)Doi:10.1039/c1cc12434j
(2011)Doi:10.1271/bbb.63.2168
(1999)