
Journal of Chemical Thermodynamics p. 1283 - 1296 (1999)
Update date:2022-08-10
Topics:
 Eli, Wumanjiang
Eli, Wumanjiang
 Chen, Wenhai
Chen, Wenhai
 Xue, Qunji
Xue, Qunji
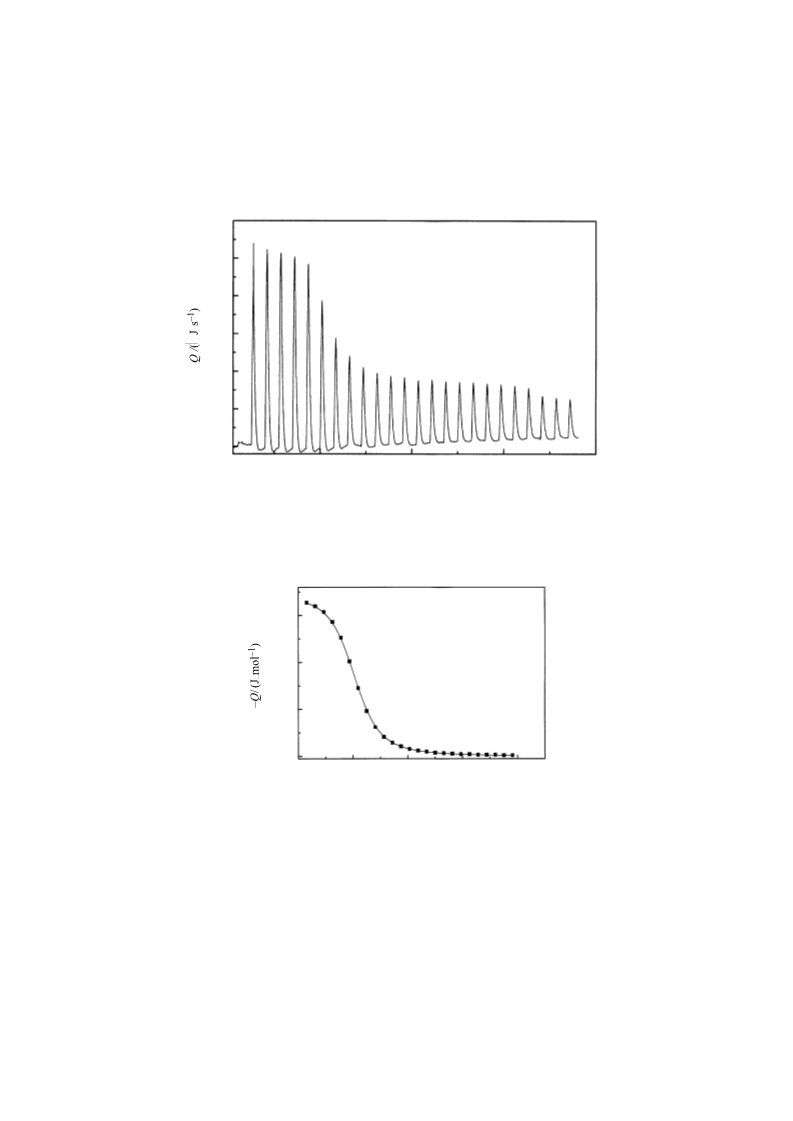
The association of a series of anionic surfactants (CnH2n+1SO4Na, n = 6, 8, 10, 12, 14) with β-cyclodextrin was studied by means of the isothermal titration calorimeter (i.t.c.) at T = 298.15 K. For these types of inclusion complexes, the results agreed well with a 1:1 association mode. Apparent values for the association constants, and changes in the standard molar Gibbs energies, enthalpies, and entropies were derived for the association process. The results indicated that the association of surfactants with β-cyclodextrin is characterized by both favourable enthalpy and favourable entropy changes. The results also demonstrated that the longer the alkyl chain of the anionic surfactant, the greater the association constant with β-cyclodextrin.
View More









Antaeus Bio-technology Co., LTD
Contact:021-31252569
Address:shanghai pudong
Contact:+86-18653358619
Address:zibo
BrightGene Bio-Medical Technology Co., Ltd.
website:https://en.bright-gene.com/
Contact:+86-512-62551801
Address:Building C25 - C31, No. 218 Xinghu Road, Suzhou Industrial Park, Suzhou, Jiangsu, China.
Yicheng Goto Pharmaceuticals Co.,Ltd.
Contact:+86 710 3423122
Address:5th Floor,East Gate of Building #2,Servo-Industrial Park,1st Qilin Road,Xiangyang,Hubei,China
Luojiang Chenming Biological Products Co
Contact:+86 15000297032
Address:GROUP NO.4, HE SHENG VILLAGE, PANLONG TOWN,
Doi:10.1021/ja01374a036
(1930)Doi:10.1271/bbb.63.238
(1999)Doi:10.1021/ol991151d
(1999)Doi:10.1016/j.jpcs.2009.11.012
(2010)Doi:10.1039/b605091c
(2006)Doi:10.1016/j.molstruc.2019.127269
(2020)