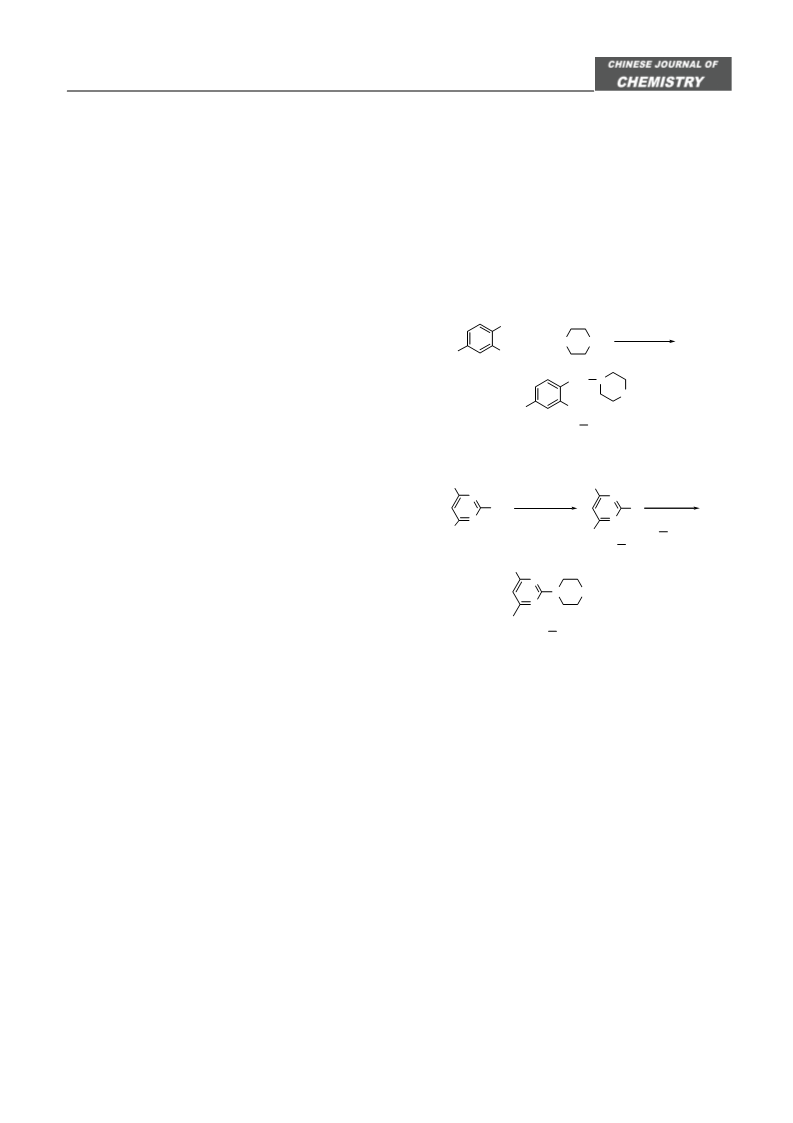Wang et al.
FULL PAPER
1
3
3
1
1
2
5
H), 2.30 (s, 3H); C NMR (101 MHz, CDCl
67.53, 162.65, 161.51, 157.16, 148.86, 148.04, 147.85,
46.71, 118.57, 112.53, 109.38, 68.84, 50.54, 43.52,
4.29, 11.07. Anal. calcd for C18
.57, N 28.12; found C 54.03, H 5.65, N 28.08.
3
) δ:
50.44, 43.51, 24.29. Anal. calcd for C18H F N OS: C
19 3 8
47.78, H 4.23, N 24.77; found C 47.53, H 4.23, N 24.40.
G7: The compound was obtained in 60.5% yield as
white crystals; m.p. 132-134 ℃. H NMR (400 MHz,
1
H
22
N
8
OS: C 54.25, H
3
CDCl ) δ: 10.21 (s, 1H), 7.71 (br s, 1H), 7.12 (d, J=3.2
G2: The compound was obtained in 65.2% yield as a
white solid; m.p. 146-147 ℃. H NMR (400 MHz,
Hz, 1H), 6.60 (br s, 1H), 6.24 (s, 1H), 5.27 (s, 2H),
1
3.88-3.87 (m, 4H), 2.88-2.87 (m, 4H), 2.26 (s, 6H);
1
3
CDCl
3
) δ: 10.37 (s, 1H), 7.66 (d, J=1.5 Hz, 1H), 7.03
d, J=3.4 Hz, 1H), 6.58 (dd, J=3.5, 1.7 Hz, 1H), 6.23
s, 1H), 5.15 (s, 2H), 3.93-3.78 (m, 4H), 2.93-2.78
C NMR (101 MHz, CDCl
150.65, 147.55, 147.39, 138.79 (q, J =42.42 Hz,
Triazole-C ), 119.55, 116.78 (q, J=272.7 Hz, CF ),
112.77, 109.04, 70.17, 50.50, 43.45, 24.06. Anal. calcd
OS: C 48.92, H 4.54, N 24.02; found C
3
) δ: 167.09, 165.11, 161.56,
(
(
(
3
3
1
3
m, 4H), 2.42 (s, 3H), 2.25 (s, 6H); C NMR (101 MHz,
) δ: 166.90, 162.58, 161.52, 148.75, 147.99,
47.74, 146.70, 118.54, 112.54, 108.79, 68.81, 50.57,
3.46, 24.05, 11.06. IR (KBr) v: 2939, 2877, 1569, 1502,
CDCl
3
21 3 8
for C19H F N
1
4
1
48.80, H 5.01, N 23.85.
G8: The compound was obtained in 64.1% yield as
−1
1
448, 1167 cm . Anal. calcd for C19
H N
24 8
OS: C 55.32,
white crystals; m.p. 140-142 ℃. H NMR (400 MHz,
H 5.86, N 27.16; found C 55.20, H 5.88, N 27.02.
CDCl ) δ: 10.50 (s, 1H), 8.28 (d, J=4.8 Hz, 2H), 7.63
3
G3: The compound was obtained in 68.5% yield as
yellow crystals; m.p. 147-149 ℃. H NMR (400 MHz,
(d, J=4.4 Hz, 1H), 7.59 (d, J=4.4 Hz, 1H), 7.17 (t, J=
1
4.4 Hz, 1H), 6.47 (t, J=4.8 Hz, 1H), 5.26 (s, 2H),
1
3
DMSO-d
d, J=4.8 Hz, 1H), 7.82 (d, J=2.8 Hz, 1H), 7.27 (dd,
J=4.9, 3.8 Hz, 1H), 6.48 (d, J=4.8 Hz, 1H), 5.09 (s,
6
) δ: 9.94 (s, 1H), 8.17 (d, J=4.8 Hz, 1H), 7.96
3.87-3.85 (m, 4H), 2.90-2.87 (m, 4H); C NMR
(
(101 MHz, CDCl ) δ: 165.20, 161.49, 157.74, 156.12,
3
138.77 (q, J=42.42 Hz, Triazole-C ), 136.41, 135.18,
3
2
3
1
1
2
5
H), 3.74-3.72 (m, 4H), 2.75-2.72 (m, 4H), 2.32 (s,
132.78, 128.15, 116.78 (q, J=273.7 Hz, CF ), 109.96,
3
1
3
H), 2.24 (s, 3H); C NMR (101 MHz, CDCl
3
) δ:
70.09, 50.36, 43.54. Anal. calcd for C H F N S : C
17
17
3
8 2
67.57, 162.72, 161.51, 157.17, 154.37, 147.78, 137.39,
34.25, 131.42, 128.02, 109.40, 68.85, 50.54, 43.53,
4.32, 11.02. Anal. calcd for C18H N S : C 52.15, H
22 8 2
4
4.92, H 3.77, N 24.65; found C 44.60, H 4.03, N 24.23.
G9: The compound was obtained in 62.7% yield as
1
white crystals; m.p. 129-131 ℃. H NMR (400 MHz,
.35, N 27.03; found C 52.11, H 5.20, N 26.98.
CDCl
3
) δ: 10.50 (s, 1H), 8.14 (d, J=4.8 Hz, 1H), 7.63
G4: The compound was obtained in 71.8% yield as
(
d, J=3.6 Hz, 1H), 7.59 (d, J=3.6 Hz, 1H), 7.16 (br s,
1
light yellow crystals; m.p. 143-145 ℃. H NMR (400
1
H), 6.36 (d, J=4.8 Hz, 1H), 5.27 (s, 2H), 3.87-3.85
1
3
MHz, DMSO-d
6
) δ: 9.95 (s, 1H), 7.96 (d, J=4.0 Hz,
H), 7.81 (s, 1H), 7.27 (d, J=3.3 Hz, 1H), 6.35 (s, 1H),
.09 (s, 2H), 3.73 (br s, 4H), 2.72 (br s, 4H), 2.31 (s,
(
(
1
1
1
m, 4H), 2.88-2.86 (m, 4H), 2.31 (s, 3H); C NMR
101 MHz, CDCl ) δ: 167.67, 165.19, 161.48, 157.21,
56.11, 138.75 (q, J=42.42 Hz, Triazole-C ), 136.43,
1
5
3
1
1
1
3
3
1
3
3
H), 2.19 (s, 6H); C NMR (101 MHz, CDCl ) δ:
35.15, 132.77, 128.14, 116.78 (q, J=272.7 Hz, CF
09.57, 70.13, 50.44, 43.50, 24.30. Anal. calcd for
: C 46.14, H 4.09, N 23.92; found C 45.88,
3
),
66.99, 162.76, 161.63, 154.31, 147.73, 137.44, 134.18,
31.38, 128.01, 108.83, 68.91, 50.65, 43.52, 24.06,
1.00. IR (KBr) v: 2913, 2843, 1600, 1575, 1496, 1157
18 19 3 8 2
C H F N S
H 4.47, N 23.92.
−
1
cm . Anal. calcd for C19
6.15; found C 53.17, H 5.48, N 25.94.
G5: The compound was obtained in 60.6% yield as
24 8 2
H N S : C 53.25, H 5.64, N
G10: The compound was obtained in 66.1% yield as
2
1
white crystals; m.p. 150-151 ℃. H NMR (400 MHz,
CDCl
3
) δ: 10.49 (s, 1H), 7.63 (d, J=3.6 Hz, 1H), 7.59
1
white crystals; m.p. 141-143 ℃. H NMR (400 MHz,
CDCl ) δ: 10.21 (s, 1H), 8.28 (d, J=4.7 Hz, 2H), 7.71
d, J=1.3 Hz, 1H), 7.12 (d, J=3.5 Hz, 1H), 6.60 (dd,
J=3.5, 1.7 Hz, 1H), 6.47 (t, J=4.7 Hz, 1H), 5.26 (s,
(
d, J=3.6 Hz, 1H), 7.17 (t, J=3.6 Hz, 1H), 6.24 (s, 1H),
3
5
6
1
1
.27 (s, 2H), 3.87 (br s, 4H), 2.88 (br s, 4H), 2.26 (s,
(
13
H); C NMR (101 MHz, CDCl
61.57, 156.10, 138.71 (q, J=42.42 Hz, Triazole-C
36.44, 135.15, 132.76, 128.14, 116.78 (q, J=272.7 Hz,
), 109.03, 70.17, 50.52, 43.47, 24.06. Anal. calcd for
: C 47.29, H 4.39, N 23.22; found C 47.65,
3
) δ: 167.09, 165.19,
3
),
13
2
H), 3.87-3.85 (m, 4H), 2.90-2.87 (m, 4H);
C
NMR (101 MHz, CDCl
50.65, 147.54, 147.43, 139.52 (m, Triazole-C
16.79 (q, J=272.7 Hz, CF
3
) δ: 165.15, 161.52, 157.73,
), 119.46,
), 112.76, 109.96, 70.12,
0.37, 43.55. Anal. calcd for C17 OS: C 46.57, H
CF
3
1
1
5
3
3
19 21 3 8 2
C H F N S
3
H 4.05, N 22.95.
17 3 8
H F N
.91, N 25.56; found C 46.67, H 4.05, N 25.23.
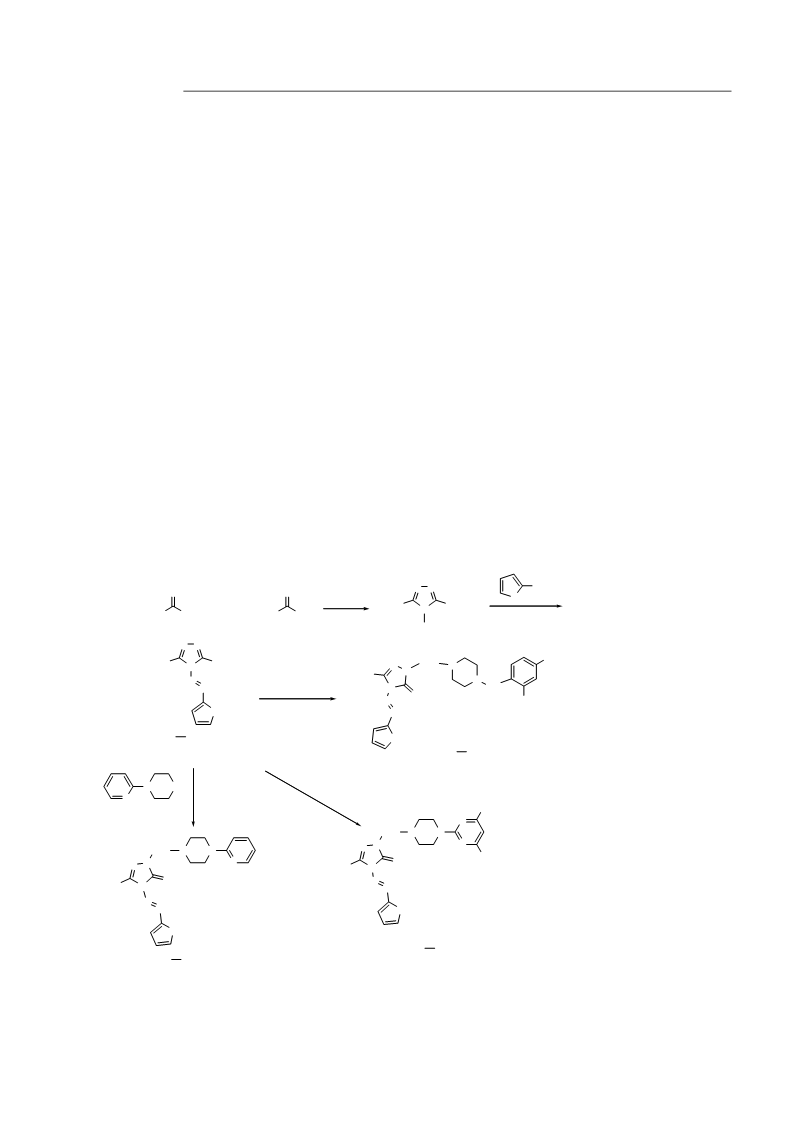
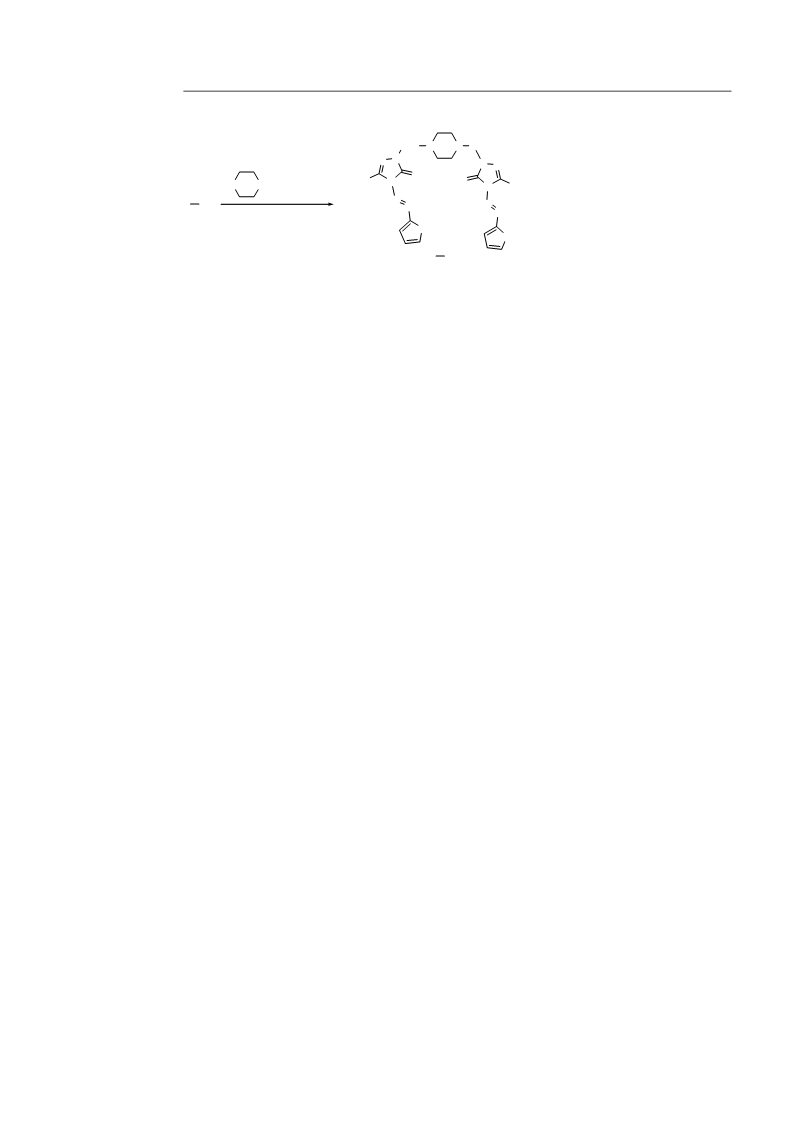
General synthetic procedures for 1-[(4-phenyl/(py-
ridin-2-yl)piperazin-1-yl)methyl]-4-(furan/thiophen-
G6: The compound was obtained in 69.2% yield as
1
2
1
-ylmethylene)amino-3-methyl/trifluoro-methyl-1H-
,2,4-triazole-5(4H)-thione H
white crystals; m.p. 126-128 ℃. H NMR (400 MHz,
CDCl
3
) δ: 10.21 (s, 1H), 8.14 (d, J=4.8 Hz, 1H), 7.70 (s,
H), 7.12 (d, J=3.2 Hz, 1H), 6.61 (bs, 1H), 6.36 (d, J=
.8 Hz, 1H), 5.27 (s, 2H), 3.87-3.86 (m, 4H), 2.88-
1
4
2
It was similar with that of F using 4-phenyl-pipera-
zine or 4-(pyridine-2-yl)piperazine as amine material
(Scheme 2).
13
3
.87 (m, 4H), 2.31 (s, 3H); C NMR (101 MHz, CDCl )
δ: 167.66, 165.14, 161.49, 157.20, 150.65, 147.54,
H1: The compound was obtained in 83.5% yield as
1
1
1
47.42, 138.83 (q, J=42.42 Hz, Triazole-C
3
), 119.47,
colorless crystals; m.p. 110-111 ℃. H NMR (400
16.79 (q, J=272.7 Hz, CF ), 112.76, 109.57, 70.15,
3
MHz, CDCl ) δ: 10.40 (s, 1H), 7.69 (d, J=1.5 Hz, 1H),
3
1128
www.cjc.wiley-vch.de
© 2015 SIOC, CAS, Shanghai, & WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim
Chin. J. Chem. 2015, 33, 1124—1134

 Wang, Baolei
Wang, Baolei
 Shi, Yanxia
Shi, Yanxia
 Zhan, Yizhou
Zhan, Yizhou
 Zhang, Liyuan
Zhang, Liyuan
 Zhang, Yan
Zhang, Yan
 Wang, Lizhong
Wang, Lizhong
 Zhang, Xiao
Zhang, Xiao
 Li, Yonghong
Li, Yonghong
 Li, Zhengming
Li, Zhengming
 Li, Baoju
Li, Baoju