The Journal of Organic Chemistry
ARTICLE
(50 mL), stirred for 24 h in contact with air. The yellow solid formed was
collected and crystallized from acetonitrile-DMF 4:1 yielding 2.20 g
(77%) of a solid still containing DMF, which was removed by drying
under reduced pressure: mp >227-228 °C (lit.81 mp 206-208 °C); 1H
NMR (300 MHz, DMSO-d6) δ 7.04-7.08 (m, 1H), 7.28-7.31 (m,
2H), 7.33-7.51 (m, 1H), 12.16 (s, 1H); 13C NMR (75.5 MHz, DMSO-
d6) δ 136.3 (s), 127.0 (s), 124.9 (s), 124.6 (d), 120.3 (d), 120.2 (d),
119.3 (s), 112.2 (d).
’ AUTHOR INFORMATION
Corresponding Author
*E-mail: jan.bergman@ki.se.
Present Addresses
§School of Chemical Science and Engineering, Royal Institute of
Technology, Teknikringen 30, 100 44 Stockholm, Sweden.
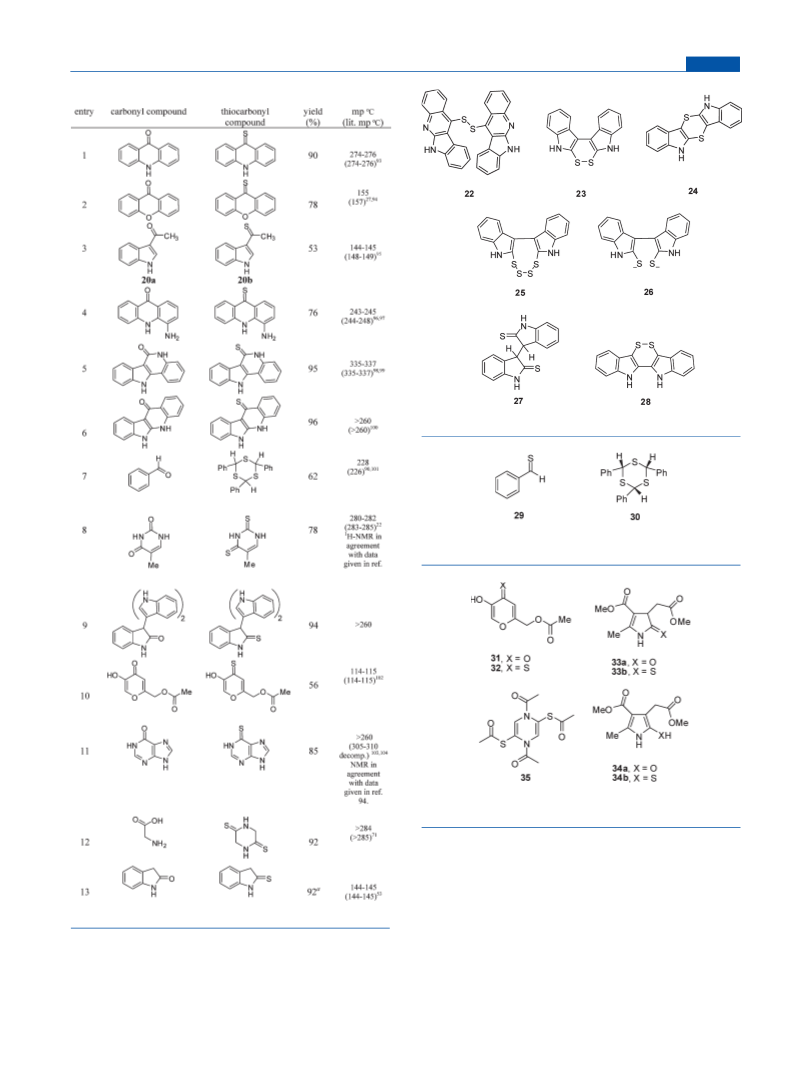
Cyclodisulfide 23 by Thionation of Oxindole at 160 °C
(Table 3, Entry 13). Oxindole (1.33 g, 10 mmol) and the reagent 3
(1.52 g, 4 mmol) were warmed with dimethyl sulfone (4. 0 g) and then
heated at 160 °C for 5 min. The melt was allowed to cool and then
heated with water. The solid formed was crystallized from acetonitrile-
DMF 4:1 yielding 1.37 g (92%): mp >227-228 °C. This material was
identical with that obtained via reductive cleavage of the tetrasulfide 25.
3,30-Bithio-oxindole, 27. The solution obtained from reductive
cleavage of the tetrasulfide 25 was acidified with AcOH, which resulted
in quick formation of the title compound as a yellow precipitate, 2.52 g
(85%). The precipitate was recrystallized from acetonitrile, mp 180 °C
dec. This molecule is sensitive toward aerial oxidation: 1H NMR (300
MHz, DMSO-d6) δ 4.66 (s, 2H), 6.85-6.91 (m, 4H), 6.96-6.98 (m,
2H), 7.07-7.13 (m, 2H), 13.06 (s, 2H); 13C NMR (75.5 MHz, DMSO-
d6) δ 60.8 (d), 110.4 (d), 123.0 (d), 123.4 (d), 128.6 (d), 130.2 (s),
144.2 (s), 204.3 (s). Anal. Calcd for C16 H12N2S2: C, 64.60; H, 4.08; N,
9.43. Found: C, 64.26; H, 3.99; N, 9.31.
’ REFERENCES
(1) Klingsberg, E.; Papa, D. J. Am. Chem. Soc. 1951, 73, 4988–4989.
(2) (a) Schumacher, G. Ph.D. Thesis, University of Mainz, Germany,
1968. (b) Hoffman, H.; Schumacher, G. Tetrahedron Lett. 1967, 31,
2963–2966.
(3) Scheibye, S. Ph.D. Thesis, University of Aarhus, Denmark, 1981.
(4) Cava, M. P.; Levinson, M. I. Tetrahedron 1985, 41, 5061–5087.
(5) Jesberger, M.; Davis, T. P.; Berner, L. Synthesis 2003, 1929–
1958.
(6) (a) Ozturk, T.; Erdal, E.; Olcay, M. Chem. Rev. 2007, 107, 5210–
5278 and references cited therein. (b) Ozturk, T.; Erdal, E.; Olcay, M.
Chem. Rev. 2010, 110, 3419–3478 and references cited therein.
(7) Shaumann, E. Compr. Org. Synth. I 1991, 6, 419–434.
(8) Meisel, M.; Grunze, H. Z. Anorg. Allg. Chem. 1967, 360, 277–283.
(9) Foreman, M. St. J.; Woollins, J. D. J. Chem. Soc., Dalton Trans.
2000, 1533–1543.
(10) Polshettiwar, V.; Kaushik, M. P. J. Sulfur Chem. 2006, 27, 353–
386.
(11) Fluck, E.; Binder, H. Z. Anorg. Allg. Chem. 1967, 354, 113–129.
(12) Brunel, E.; Monzur, J.; Retuert, J. J. Chem. Res., Miniprint 1981,
3437–3445.
Methyl 5-Mercapto-4-(2-methoxy-2-oxoethyl)-2-methyl-
1H-pyrrole-3-carboxylate, 34b. The diester 33a(2.13g, 10 mmol)
and the reagent 3 (1.14 g, 4 mmol) were heated at reflux temperature in
acetonitrile (50 mL) for 1 h. After concentration to 25 mL, water was
added and the solid formed collected and crystallized from 2-propanol,
1.85 g (81%): mp 185-187 °C; IR νmax 3273, 2954, 1742, 1724, 1707,
(13) Weintraub, P. M. Int. J. Sulfur Chem. 1973, 8, 321–327.
(14) S€oder, A. Ger. Pat. 2,822,113, 1979.
1681, 1562, 1440, 1341, 1269, 1200, 1173, 1117, 1080, 1003, 782 cm-1
;
(15) Stratman, J. Pharm. Acta Helv. 1989, 64, 252–255.
(16) Brillon, D. Sulfur Rep. 1992, 12, 297–338.
(17) Goel, O. P.; Krolls, U. Synthesis 1987, 162–164.
(18) Charette, A. D.; Grenon, M. J. Org. Chem. 2003, 68, 5792.
(19) Scheeren, J. W.; Ooms, P. H. J.; Nivard, R. J. F. Synthesis 1973,
149–151.
(20) Curphey, T. J. Org. Chem. 2002, 67, 6461–6473.
(21) Foreman, M. S. J.; Slawin, A. M. Z.; Woollins, J. D. Heteroatom
Chem. 1999, 10, 651–657.
(22) Lapucha, A. R. Synthesis 1987, 256–258.
(23) Cho, D.; Ahn, J.; Castro, K. A. D.; Ahn, H.; Rhee, H. Tetra-
hedron 2010, 66, 5583–5588.
(24) Kaneko, K.; Katayama, H.; Wakabayashi, T.; Kumonoka, T.
Synthesis 1988, 152–158.
1H NMR (300 MHz, DMSO-d6) δ 2.43 (s, 3H), 3.17 (s, 2H), 3.38 (s,
1H), 3.49 (s, 3H), 3.64 (s, 3H), 11.90 (s 1H); 13C NMR (75.5 MHz,
DMSO-d6) δ 13.4 (q), 30.6 (d), 50.4 (q), 51.4 (q), 111.2 (s), 117.1 (s),
126.9 (s), 139.9 (s), 164.4 (s), 171.1 (s)Anal. Calcd for C10 H13NO4S: C,
49.37; H, 5.38; N 5.75. Found: C, 49.25; H, 5.46; N, 5.61.
3-(1H-Indol-3-yl)-3,30-biindoline-2-thione (Table 3, Entry 9).
3-(1H-Indol-3-yl)-3,30-biindolin-2-one105 (728 mg, 2 mmol), the reagent 3
(228 mg, 0.6 mmol), and dimethyl sulfone (3.05 g) were heated (165-
170 °C) for 20 min. The melt was allowed to cool and then heated in water
for 10 min. The solid formed was collected, 713 mg (94%): mp >260 °C;
1H NMR (300 MHz, DMSO-d6) δ 7.09-7.15 (m, 2H), 7.18-7.20 (m,
5H), 7.24-7.30 (m, 7H), 13.00 (s, 1H); 13C NMR (75.5 MHz, DMSO-d6)
δ 72.7 (s), 111.2 (d), 124.4 (d), 126.5 (d), 127.5 (d), 128.6 (d), 128.7 (d),
129.0 (d), 129.1 (d), 129.1 (d), 139.2 (s), 143.0 (s), 143.5 (s), 145.3 (s,
2C), 208.4 (s). Anal. Calcd for C24H17N3S: C, 75.96; H, 4.51; N, 11.07.
Found: C, 76.10; H, 4.46; N, 11.00.
ꢀ
(25) Janosik, T.; Bergman, J.; Stensland, B.; Stalhandske, C. J. Chem.
Soc., Perkin Trans. 1 2002, 330–334.
(26) Fluck, E.; Binder, H. Angew. Chem. 1966, 78, 666.
(27) Scheibye, S.; Shabana, R.; Lawesson, S. O.; R€omming, C.
Tetrahedron 1982, 38, 993–1001.
(28) Smolders, R. R.; Hanuise, J.; Coomans, R.; Proietto, V.; Voglet,
N.; Waefelaer, A. Synthesis 1982, 493–494.
’ ASSOCIATED CONTENT
(29) Ley, S. V.; Leach, A. G.; Storer, R. I. J. Chem. Soc., Perkin Trans. 1
2001, 358–361.
(30) Jagodzinski, T. S. Chem. Rev. 2003, 103, 197–227.
(31) Kaleta, Z.; Tarkanyi, G.; A, G.; Kalman, F.; Nagy, T.; Soos, T.
Org. Lett. 2006, 8, 1093–1095.
(32) Filippi, J. J.; Fernandez, X.; Lizzani-Cuvelier, L.; Loiseau, A. M.
Tetrahedron Lett. 2003, 44, 6647–6650.
(33) Seijas, J. A.; Vꢀazquez-Tato, M. P.; Martínez, M. M.; Rodríguez-
Parga, J. Green Chem. 2002, 4, 390–391.
(34) Prokopcova, H.; Kappe, O. C. Adv. Syn. Catal. 2007, 349, 448–
452.
(35) Kappe, O. C.; Prokopcova, H. Angew. Chem. 2009, 48, 2276–2286.
(36) Nuzhat, A.; Jamshed, H.; Kappe, O. C. J. Org. Chem. 2009, 74,
5118–5121.
S
Supporting Information. The crystal structural data of 3
b
and 11a have been deposited at Cambridge Data Centre and
allocated the deposition numbers CCDC 789665 and CCDC
789666. CIFs for compounds 3 and 11a are included. Experi-
mental details for 6H-indolo[2,3-b]quinolin-11-one and com-
pounds 13a and 33a (Table 1, entries 2, 3, and 8). Compound
characterization data for 6H-indolo[2,3-b]quinolin-11-one and
compounds 13a and 33a (Table 1, entries 2, 3, 8, and 11; Table 3,
entries 3, 7, and 10). Copies of 1H and 13C NMR spectra for all
1
new compounds. Copies of H NMR are provided for most
compounds in the tables. This material is available free of charge
1552
dx.doi.org/10.1021/jo101865y |J. Org. Chem. 2011, 76, 1546–1553

 Bergman, Jan
Bergman, Jan
 Pettersson, Birgitta
Pettersson, Birgitta
 Hasimbegovic, Vedran
Hasimbegovic, Vedran
 Svensson, Per H.
Svensson, Per H.