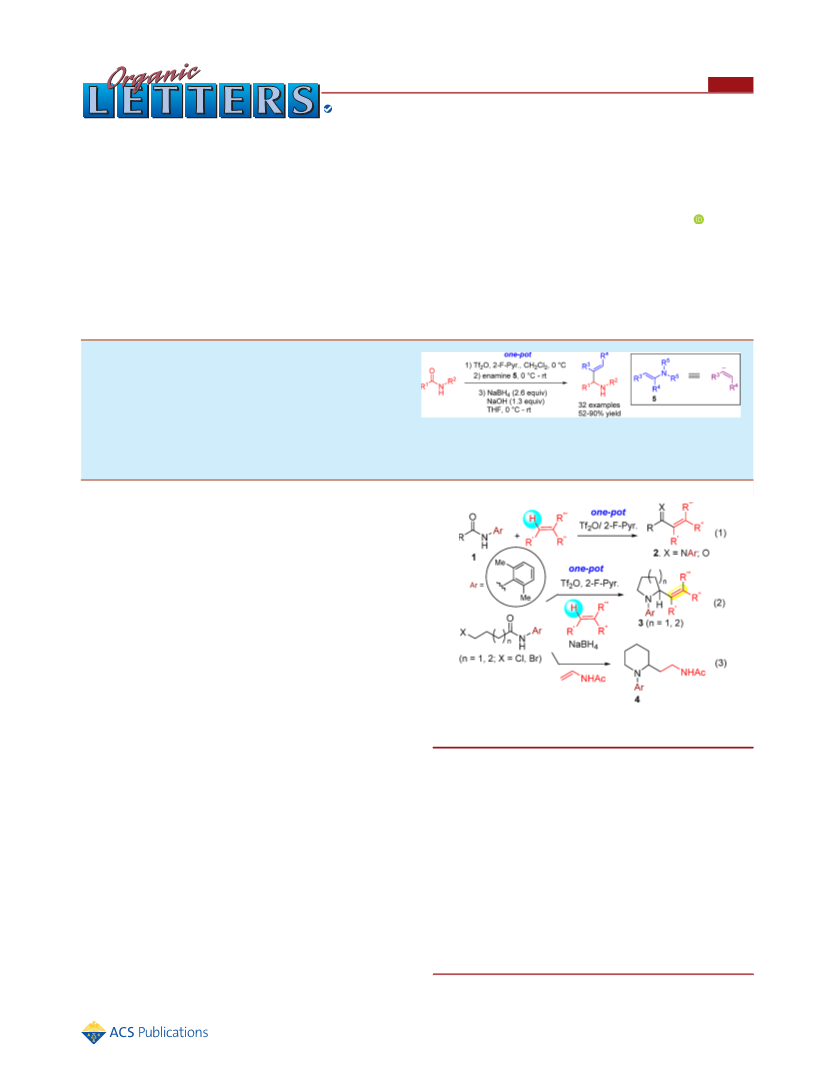
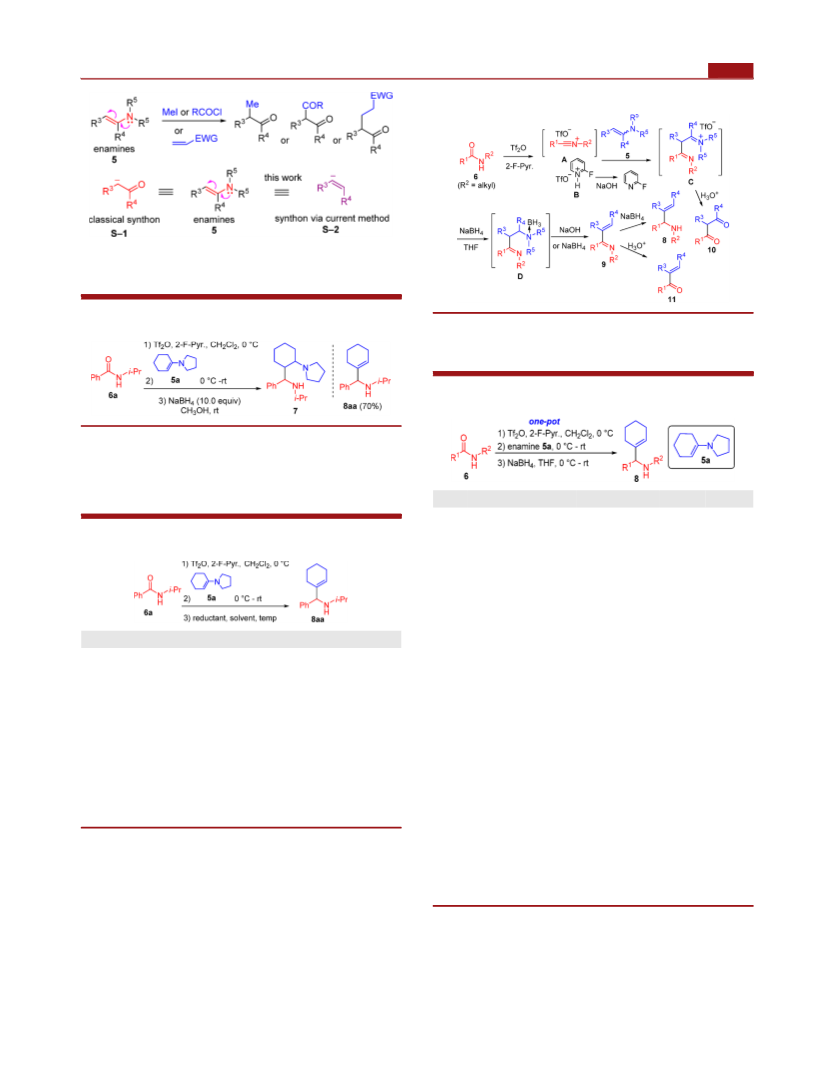
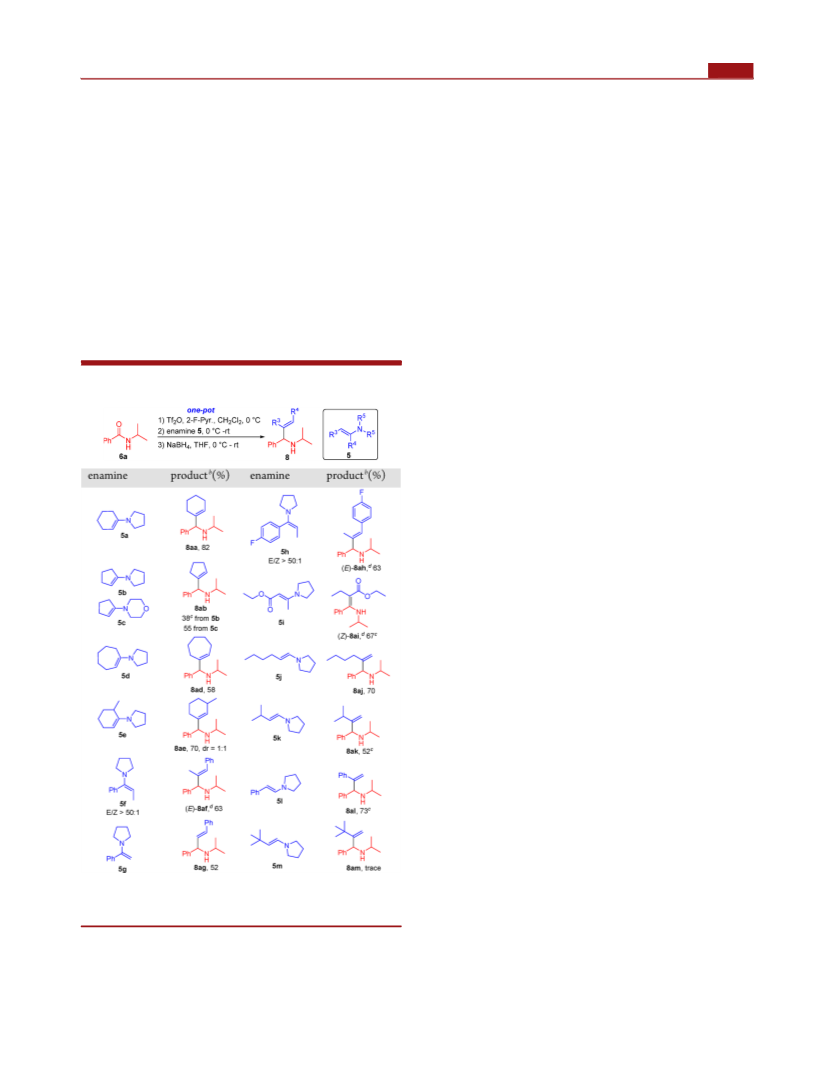
Organic Letters
Letter
Soda, Y.; Oishi, T.; Sato, T.; Chida, N. J. Am. Chem. Soc. 2017, 139,
18386.
AUTHOR INFORMATION
■
Corresponding Authors
(5) (a) Xiao, K.-J.; Luo, J.-M.; Ye, K.-Y.; Wang, Y.; Huang, P.-Q. Angew.
Chem., Int. Ed. 2010, 49, 3037. (b) Xiao, K.-J.; Wang, A.-E; Huang, P.-Q.
Angew. Chem., Int. Ed. 2012, 51, 8314. (c) Wang, A.-E; Chang, Z.; Sun,
W.-T.; Huang, P.-Q. Org. Lett. 2015, 17, 732. (d) Huang, P.-Q.; Huang,
Y.-H.; Xiao, K.-J.; Wang, Y.; Xia, X.-E. J. Org. Chem. 2015, 80, 2861.
(e) Huang, P.-Q.; Ou, W.; Han, F. Chem. Commun. 2016, 52, 11967.
(f) Huang, P.-Q.; Huang, Y.-H.; Geng, H.; Ye, J.-L. Sci. Rep. 2016, 6,
28801. (g) Huang, P.-Q.; Huang, Y.-H.; Wang, S.-R. Org. Chem. Front.
2017, 4, 431.
ORCID
Author Contributions
§C.-C.Y. and T.-T.C. contributed equally to this work.
(6) Baraznenok, I. L.; Nenajdenko, V. G.; Balenkova, E. S. Tetrahedron
2000, 56, 3077.
Notes
The authors declare no competing financial interest.
(7) (a) Stork, G.; Terrell, R.; Szmuszkovicz, J. J. Am. Chem. Soc. 1954,
76, 2029. (b) Stork, G.; Landesman, H. J. Am. Chem. Soc. 1956, 78, 5128.
(c) Stork, G.; Landesman, H. J. Am. Chem. Soc. 1956, 78, 5129.
(d) Stork, G.; Brizzolara, A.; Landesman, H.; Szmuszkovicz, J.; Terrell,
R. J. Am. Chem. Soc. 1963, 85, 207.
(8) (a) The Chemistry of Enamines; Rappoport, Z., Ed.; Wiley-VCH:
Weinheim, 1994. (b) Enamines: Synthesis, Structure and Reactions, 2nd
ed.; Cook, A. G., Ed.; Dekker: New York, 1998.
(9) Dalko, P. I.; Moisan, L. Angew. Chem., Int. Ed. 2004, 43, 5138.
(10) (a) Johannsen, M.; Jørgensen, K. A. Chem. Rev. 1998, 98, 1689.
(b) Grange, R. L.; Clizbe, E. A.; Evans, P. A. Synthesis 2016, 48, 2911.
(c) Groenendaal, B.; Ruijter, E.; Orru, V. A. Chem. Commun. 2008, 5474.
(11) (a) Lu, Z.; Ma, S. Angew. Chem., Int. Ed. 2008, 47, 258. (b) Trost,
B. M.; Zhang, T.; Sieber, J. D. Chem. Sci. 2010, 1, 427. (c) Sundararaju,
B.; Achard, M.; Bruneau, C. Chem. Soc. Rev. 2012, 41, 4467.
(12) Ramirez, T. A.; Zhao, B.; Shi, Y. Chem. Soc. Rev. 2012, 41, 931.
ACKNOWLEDGMENTS
■
Financial support for this work was provided by the National Key
R&D Program of China (2017YFA0207302), the National
Natural Science Foundation of China (21332007 and
21472153), the Program for Changjiang Scholars and Innovative
Research Team in University (PCSIRT) of Ministry of
Education, the Fundamental Research Funds for the Central
Universities (20720150044), and the Natural Science Founda-
tion of Fujian Province of China (2014J01062).
DEDICATION
■
This paper is dedicated to the memory of the late Professor
Gilbert Stork.
(13) Muller, T. E.; Hultzsch, K. C.; Yus, M.; Foubelo, F.; Tada, M.
̈
Chem. Rev. 2008, 108, 3795.
(14) Masson, G.; Housseman, C.; Zhu, J. Angew. Chem., Int. Ed. 2007,
46, 4614.
REFERENCES
■
(1) The amide linkage: Structural Significance in Chemistry,
Biochemistry and Materials Science; Greenberg, A., Breneman, C. M.,
Liebman, J. F., Eds.; Wiley: New York, 2003.
(2) For reviews, see: (a) Seebach, D. Angew. Chem., Int. Ed. 2011, 50,
96. (b) Pace, V.; Holzer, W. Aust. J. Chem. 2013, 66, 507. (c) Pace, V.;
Holzer, W.; Olofsson, B. Adv. Synth. Catal. 2014, 356, 3697. (d) Sato, T.;
Chida, N. Org. Biomol. Chem. 2014, 12, 3147. (e) Ruider, S. A.; Maulide,
N. Angew. Chem., Int. Ed. 2015, 54, 13856. (f) Kaiser, D.; Maulide, N. J.
Org. Chem. 2016, 81, 4421. (g) Sato, T.; Chida, N. Yuki Gosei Kagaku
Kyokaishi 2016, 74, 599.
(3) For selected examples on transformation of secondary amides, see:
(a) Movassaghi, M.; Hill, M. D. J. Am. Chem. Soc. 2006, 128, 4592.
(b) Medley, J. W.; Movassaghi, M. J. Org. Chem. 2009, 74, 1341.
(c) Pelletier, G.; Bechara, W. S.; Charette, A. B. J. Am. Chem. Soc. 2010,
132, 12817. (d) Bechara, W. S.; Pelletier, G.; Charette, A. B. Nat. Chem.
2012, 4, 228. (e) Oda, Y.; Sato, T.; Chida, N. Org. Lett. 2012, 14, 950.
(15) Wei, Y.; Shi, M. Chem. Rev. 2013, 113, 6659.
(16) (a) Skucas, E.; Ngai, M.-Y.; Komanduri, V.; Krische, M. J. Acc.
Chem. Res. 2007, 40, 1394. (b) Petasis, N. A.; Akritopoulou, I.
Tetrahedron Lett. 1993, 34, 583. For a review see: (c) Candeias, N. R.;
Montalbano, F.; Cal, P. M. S. D.; Gois, P. M. P. Chem. Rev. 2010, 110,
6169.
(17) Liu, T.-L.; Wang, C.-J.; Zhang, X. Angew. Chem., Int. Ed. 2013, 52,
8416.
(18) Perlmutter, P. Conjugate Addition Reactions in Organic Synthesis;
Pergamon: New York, 1992.
(19) (a) Hirasawa, Y.; Kobayashi, J.; Morita, H. Org. Lett. 2006, 8, 123.
(b) Wrasidlo, W.; Mielgo, A.; Torres, V. A.; Barbero, S.; Stoletov, K.;
Suyama, T. L.; Klemke, R. L.; Gerwick, W. H.; Carson, D. A.; Stupack, D.
G. Proc. Natl. Acad. Sci. U. S. A. 2008, 105, 2313.
(20) Stutz, A. Angew. Chem., Int. Ed. Engl. 1987, 26, 320.
̈
(21) Marín, M. T.; Margarit, M. V.; Salcedo, G. E. Farmaco 2002, 57,
(f) Cyr, P.; Reg
17, 3386. (g) Pace, V.; de la Vega-Hernan
Org. Lett. 2016, 18, 2750. (h) Regnier, S.; Bechara, W. S.; Charette, A. B.
J. Org. Chem. 2016, 81, 10348. (i) Tona, V.; Maryasin, B.; de la Torre, A.;
Sprachmann, J.; Gonzalez, L.; Maulide, N. Org. Lett. 2017, 19, 2662. See
́
nier, S.; Bechara, W. S.; Charette, A. B. Org. Lett. 2015,
723.
(22) Skoda, E. M.; Davis, G. C.; Wipf, P. Org. Process Res. Dev. 2012, 16,
26.
(23) (a) Myers, A. G.; Yang, B. H.; Chen, H.; McKinstry, L.; Kopecky,
D. J.; Gleason, J. L. J. Am. Chem. Soc. 1997, 119, 6496. (b) Ren, J.;
Workman, D. B.; Squires, R. R. Angew. Chem., Int. Ed. Engl. 1997, 36,
2230.
́
dez, K.; Urban, E.; Langer, T.
́
́
also: (j) Li, X.-W.; Lin, F.-G.-R.; Huang, K.-M.; Wei, J.-L.; Li, X.-Y.;
Wang, X.-Y.; Geng, X.-Y.; Jiao, N. Angew. Chem., Int. Ed. 2017, 56,
12307.
(4) For selected examples on transformation of tertiary amides, see:
(a) White, K. L.; Mewald, M.; Movassaghi, M. J. Org. Chem. 2015, 80,
7403. (b) Katahara, S.; Kobayashi, S.; Fujita, K.; Matsumoto, T.; Sato,
T.; Chida, N. J. Am. Chem. Soc. 2016, 138, 5246. (c) Tinnis, F.; Volkov,
A.; Slagbrand, T.; Adolfsson, H. Angew. Chem., Int. Ed. 2016, 55, 4562.
(d) Pace, V.; Murgia, I.; Westermayer, S.; Langer, T.; Holzer, W. Chem.
Commun. 2016, 52, 7584. (e) Kaiser, D.; de la Torre, A.; Shaaban, S.;
Maulide, N. Angew. Chem., Int. Ed. 2017, 56, 5921. (f) Shi, H.-Y.;
Michaelides, I.; Darses, B.; Jakubec, P.; Nguyen, Q. N. N.; Paton, R. S.;
Dixon, D. J. J. Am. Chem. Soc. 2017, 139, 17755. (g) Trillo, P.; Slagbrand,
T.; Tinnis, F.; Adolfsson, H. Chem. Commun. 2017, 53, 9159. (h) de la
Torre, A.; Kaiser, D.; Maulide, N. J. Am. Chem. Soc. 2017, 139, 6578.
(i) Yoritate, M.; Takahashi, Y.; Tajima, H.; Ogihara, C.; Yokoyama, T.;
D
Org. Lett. XXXX, XXX, XXX−XXX

 Wang, Ai-E
Wang, Ai-E
 Yu, Cun-Cun
Yu, Cun-Cun
 Chen, Ting-Ting
Chen, Ting-Ting
 Liu, Yong-Peng
Liu, Yong-Peng
 Huang, Pei-Qiang
Huang, Pei-Qiang