NOVEL BENZOTHIAZOLE IONIC LIQUIDS AS CATALYSTS FOR THE SYNTHESIS OF PARABENS 1481
EXPERIMENTAL
ν(O–H), 2977 ν(C–H), 1680 ν(C=O), 1605, 1445
ν(C–Harom), 1295 ν(C–O–C), 844 γ(1,4-Ph–H).
Materials and methods. All chemicals were of
analytical grade and used without further purification.
The melting points were determined in an open glass
capillary and are uncorrected. The IR spectra (400–
Isopropyl paraben (2c). White solid, mp 85–87°C
–1
(84–86°C [23]). IR spectrum (KBr), ν, cm : 3400
ν(O–H), 2987 ν(C–H), 1690 ν(C=O), 1587, 1502, 1446
ν(C–Harom), 1285 ν(C–O–C), 853 γ(1,4-Ph–H).
–1
4
000 cm ) were recorded on a Bruker Vector-22 FTIR
spectrometer for samples prepared as KBr pellets. The
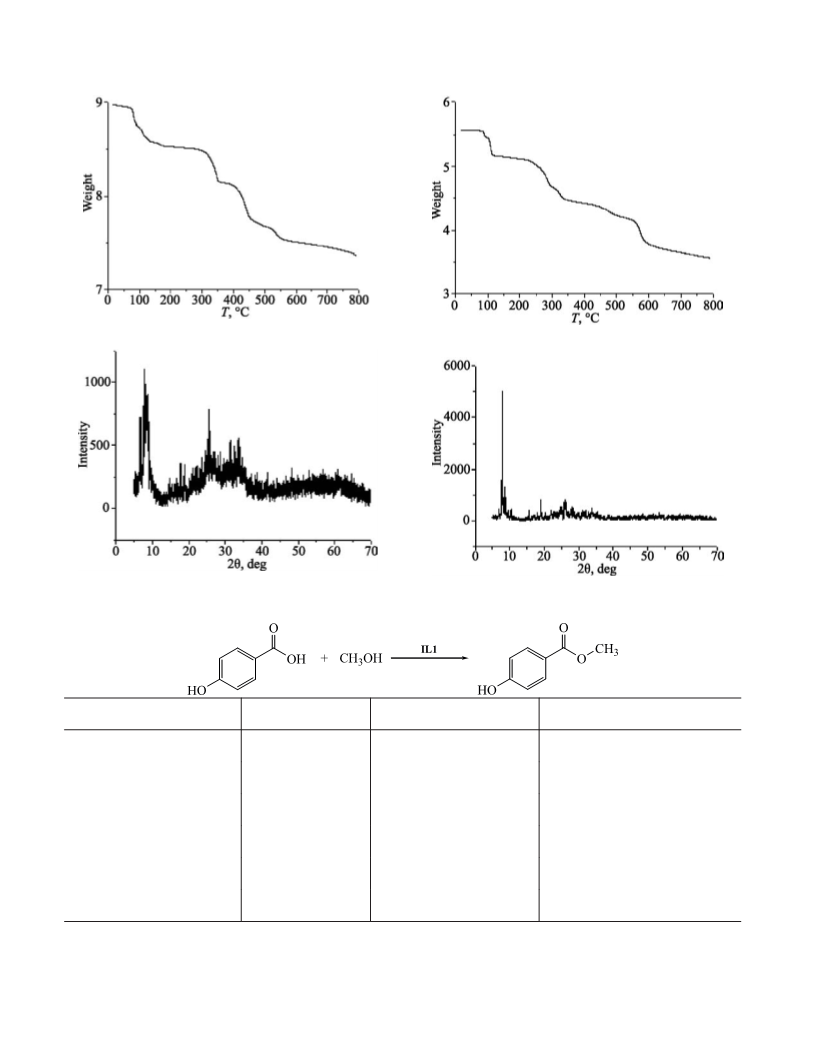
powder XRD patterns were obtained on a Bruker D8
Quest diffractometer. The TGA analysis was performed
Butyl paraben (2d). White solid, mp 66–67°C
68–69°C [24]). IR spectrum (KBr), ν, cm : 3381
ν(O–H), 2949, 2975 ν(C–H), 1681 ν(C=O), 1587, 1502,
1454 ν(C–Harom) 1285 ν(C–O–C), 844 γ(1,4-Ph–H).
–1
(
on a TA Discovery instrument in flowing N with a
2
heating rate of 10 min .The SEM images were obtained
on a FEI Philips Verios 460 microscope. The C, H,
O analyses were performed with a Fisons EA 1108
Isobutyl paraben (2e). White solid, mp 74–77°C
–1
(
76–78°C [25]). IR spectrum (KBr), ν, cm : 3241
ν(O–H), 2959, 2884 ν(C–H), 1680 ν(C=O), 1596, 1511,
454 ν(C–Harom), 1285 ν(C–O–C), 853 γ(1,4-Ph–H).
(CHNSO) elemental analyzer.
1
Synthesis of ionic liquids IL1 and IL2 (general
Isopentyl paraben (2f). White solid, mp 58–60°C
procedure). Asolution of heteropolyacid (H PW O or
3
12 40
–1
(
58–59°C [26]). IR spectrum (KBr), ν, cm : 3260
H PMo O ) (1 mmol) in ethanol (20 mL) was cooled
5
12 41
ν(O–H), 2950 ν(C–H), 1680 ν(C=O), 1596, 1511, 1454
ν(C–Harom), 1285 ν(C–O–C), 853 γ(1,4-Ph–H).
to 0–5°C, and then a solution of benzothiazole (1 mmol)
in ethanol (5 mL) was added dropwise, the mixture
was stirred at room temperature until completion of the
reaction (by TLC) ,filtered. The solid was washed with
ethyl acetate and dried to obtain the target product.
Isooctyl paraben (2g). White solid, mp 38–42°C. IR
–
1
spectrum (KBr), ν, cm : 3485 ν(O–H), 2958 ν(C–H),
718 ν(C=O), 1633, 1554, 1489 ν(C–Harom), 1239 ν(C–
1
O–C), 834 γ(1,4-Ph–H). Found, %: C 71.95; H 8.90;
[
HBth]H PW O (IL1). Yellow powder, yield
2 12 40
O 19.21. C H O . Calculated, %: C 71.97; H 8.86;
1
5
22
3
–
1
7
0.4%, mp > 250°C. IR spectrum (KBr), ν, cm : 3516
O 19.17.
ν(N–H), 3086 ν(=C–H), 1698 ν(C=N), 1430 ν(aromatic
C–H), 1216 ν(C–S), 795 γ(1,2-Ph–H).
Decyl paraben (2h). White solid, mp 31–33°C (30–
–
1
3
4°C [27]). IR spectrum (KBr), ν, cm : 3485 ν(O–H),
[
HBth]H PMoO
(IL2). White powder, yield
4
41
2940 ν(C–H), 1718 ν(C=O), 1587, 1445 ν(C–Harom),
267 ν(C–O–C), 853 γ(1,4-Ph–H).
–
1
7
1.2%, mp > 250°C. IR spectrum (KBr), ν, cm : 3502
1
ν(N–H), 3065 ν(=C–H), 1697 ν(C=N), 1431 ν(aromatic
C–H), 1262 ν(C–S), 809 γ(1,2-Ph–H).
Synthesis of dodecyl paraben (2i) and hexadecyl
paraben 2j (general procedure). p-Hydroxybenzoic
acid (0.1 mol), toluene (10 mL) and IL1 or IL2 were
added in succession to alcohol dodecyl or hexadecyl
Synthesis of paraben compounds 2a–2h (gene-
ral procedure). A mixture of p-hydroxybenzoic acid
(10 mmol), alcohol 1a–1g, and IL1 or IL2 was refluxed
(
0.11 mol). The mixture was refluxed until completion
until completion of the reaction (by TLC). The alcohol
was evaporated, and the residue was extracted with
ethyl acetate (3 × 10 mL). The organic phases were
combined, dried, and concentrated. The precipitate
was recrystallized from methanol to obtain the target
product.
of the reaction (by TLC). The solvent was evaporated,
and the residue was extracted with ethyl acetate
(3 × 10 mL). The organic phases were combined, dried,
and concentrated. The precipitate was recrystallized
from methanol to obtain the target product.
Dodecyl paraben (2i). White solid, mp 39–41°C
–1
Methyl paraben (2a). White solid, mp 126–128°C
(36–38°C [15]). IR spectrum (KBr), ν, cm : 3390
ν(O–H), 2945, 2911 ν(C–H), 1688 ν(C=O), 1595, 1511,
1465 ν(C–Harom), 1275 ν(C–O–C), 852 γ(1,4-Ph–H).
–
1
(
126–127°C [21]). IR spectrum (KBr), ν, cm : 3292
ν(O–H), 3037 ν(Ph–H), 2958 ν(C–H), 1679 ν(C=O),
593, 1514, 1438 ν(C–Harom), 1278 ν(C–O–C), 850
1
Hexadecyl paraben (2j). White solid, mp 61–63°C
γ(1,4-Ph–H).
–1
(
64–65°C [28]). IR spectrum (KBr), ν, cm : 3391
Ethyl paraben (2b). White solid, mp 116–118°C
116–118°C [22]). IR spectrum (KBr), ν, cm : 3212
ν(O–H), 2921 ν(C–H), 1690 ν(C=O), 1596, 1511, 1465
ν(C–Harom), 1261 ν(C–O–C), 862 γ(1,4-Ph–H).
–
1
(
RUSSIAN JOURNAL OF ORGANIC CHEMISTRY Vol. 56 No. 8 2020

 Liu, B.
Liu, B.
 Liu, Y.
Liu, Y.
 Sun, J.
Sun, J.
 Yin, D.
Yin, D.