
Advanced Synthesis and Catalysis p. 1499 - 1506 (2015)
Update date:2022-08-12
Topics:
 Touchy, Abeda S.
Touchy, Abeda S.
 Kon, Kenichi
Kon, Kenichi
 Onodera, Wataru
Onodera, Wataru
 Shimizu, Ken-Ichi
Shimizu, Ken-Ichi
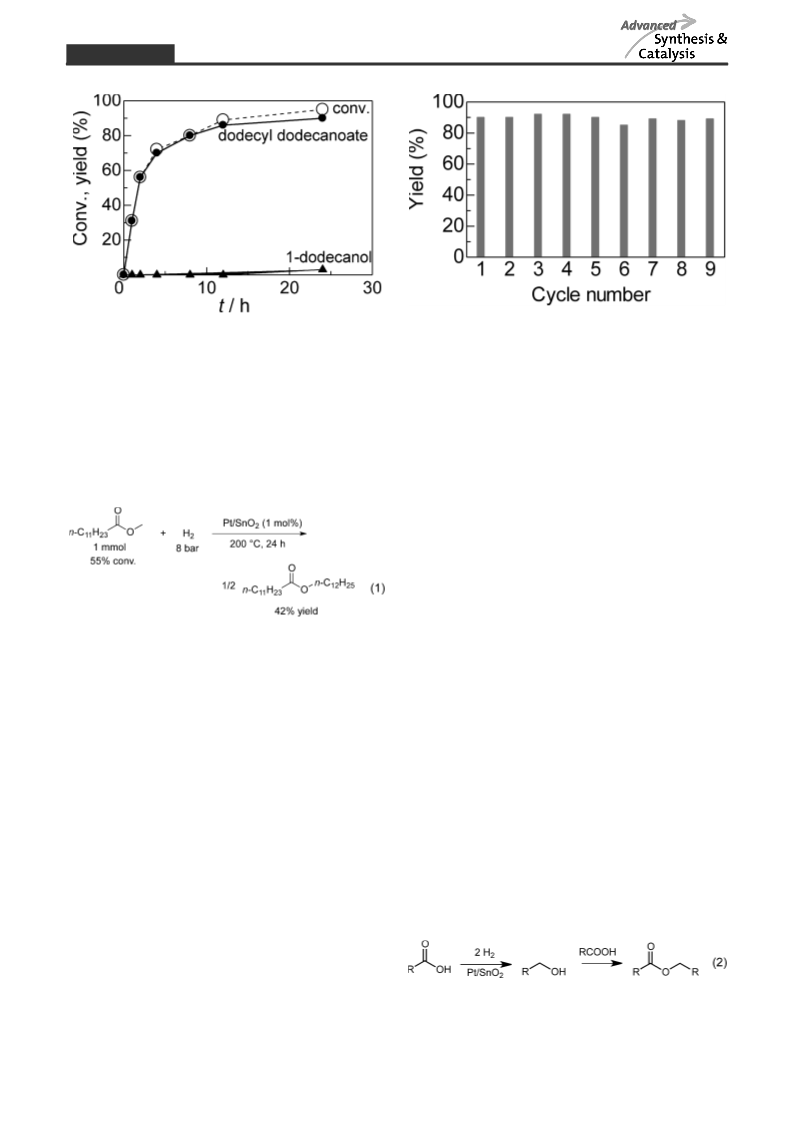
Supported metal catalysts have been tested for an unprecedented reductive dimerization of carboxylic acids to esters under 8 bar hydrogen in solvent-free conditions. Among various metal-loaded tin oxide catalysts, platinum-loaded tin dioxide (Pt/SnO2) shows the highest ester yield for the reaction of dodecanoic acid. Among Pt catalysts on various supports, Lewis acidic oxides, especially SnO2, show high activity. The most active catalyst, 5 wt% Pt/SnO2 reduced at 100°C, is effective for the reductive esterification of various carboxylic acids, and the catalyst is reusable for nine cycles, demonstrating the first successful example for the title reaction. Infrared (IR) studies of a model compound (formic acid) on some metal oxides indicate a strong Lewis acid-base interaction between SnO2 and the carbonyl oxygen. For Pt/SnO2 catalysts with different Pt particle sizes, the activity increases with decreasing size of Pt metal. A cooperative catalysis of the Pt metal nanoparticles and the Sn4+ Lewis acid sites is proposed.
View More


Jintan City Mego Chemical Co., Ltd
Contact:+86-0519-82814387
Address:23# Dengguan Town, Jintan, Jiangsu Province, China
Shanghai Kangxin Chemical Co., Ltd
Contact:+86 21 60717227
Address:118,Ganbai Village,Waigang Town,Jiading District,Shanghai
Sichuan Sangao Biochemical Co., Ltd
Contact:+86-28-84874233
Address:19F, Bldg.2, Shudu Center, Tianfu 2nd St., Hi-tech zone, Chengdu 610041, Sichuan Province, China.
Contact:0510-85393305
Address:1619 Huishan Avenue, Huishan District, Wuxi,
website:http://www.sjzthld.com
Contact:86-311-66699812
Address:NO.600 ZHONGSHAN EAST ROAD SHIJIAZHUANG
Doi:10.1021/jo052368q
(2006)Doi:10.1016/j.tetlet.2014.11.009
(2014)Doi:10.1002/zaac.201100316
(2012)Doi:10.1023/A:1002829018667
(2000)Doi:10.1002/chem.201801668
(2018)Doi:10.1016/j.molcata.2014.07.021
(2014)