Organic Process Research & Development 2006, 10, 941−943
Production of Piperonal, Vanillin, and p-Anisaldehyde via Solventless
Supported Iodobenzene Diacetate Oxidation of Isosafrol, Isoeugenol, and
Anethol Under Microwave Irradiation
Heiddy Marquez Alvarez,*,† Dayse P. Barbosa,†,§ Alini Tinoco Fricks,† Donato A. G. Aranda,‡ Ricardo H. Valde´s,‡,§ and
O. A. C. Antunes*,†,§
Instituto de Qu´ımica, UFRJ, CT Bloco A 641, Cidade UniVersita´ria, Rio de Janeiro RJ 21941-590, Brazil, Escola de
Qu´ımica, UFRJ, Rio de Janeiro, 21941-590 Brazil, Nu´cleo de Pesquisas de Produtos Naturais, UFRJ, CCS Bloco H,
Cidade UniVersita´ria, Rio de Janeiro, RJ 21941-590, Brazil
Abstract:
catalytic oxygenation reactions. However, polymeric PhIO
is insoluble in most organic media. PhIO is prepared by the
hydrolysis of PhI(OAc)2 which is soluble in most organic
solvents.5
Recently, several groups reported the direct use of
PhI(OAc)2 as a terminal oxidant, for example in the oxidation
of alcohols to carbonyl compounds using alumina-supported
PhI(OAc)2 under microwave irradiation,6 chemoselective
oxidation of alcohols by CrIII(salen)X,4a and oxygenation of
olefins by iron(III)-porphyrin catalysis in the presence of a
small amount of water in organic solvent.7
A novel experimental procedure to obtain carbonyl compounds
under microwave irradiation from activated olefins and sup-
ported iodobenzene diacetate is described. By varying the
reaction conditions it is possible to generate the corresponding
aldehydes in reasonable-to-excellent yields and selectivities. The
methodology is simple, clean, and reproducible and presents
short reaction times. Using isosafrol, isoeugenol, and anethol it
was possible to produce piperonal, vanillin and p-anisaldehyde,
respectively.
Whereas the epoxidation of alkenes metalloporphyrins/
PhI(OAc)2 is already known, the selective cleavage of CdC
double bonds leading to aldehydes, ketones, or carboxylic
acids has thus far never (to our knowledge) been reported
so far.
Due to the fact that piperonal, p-anisaldehyde, and vanillin
are used extensively within the flavor and fragrance industry
and because the present process includes, among other routes,
ozonolysis followed by ozonide reduction with sulphur or
zinc, a highly energy-demanding and environmentally un-
friendly process, we describe in the present paper the
oxidation of isopropenylbenzenes to the corresponding
aldehydes using solventless supported PhI(OAc)2 as reaction
media.
Introduction
The oxidation of olefins to carbonyl compounds is an
important reaction in organic synthesis and several methods
are available to accomplish this conversion under a variety
of reaction conditions.1 In recent years, organic reactions on
solid supports assisted by microwave irradiation have gained
special attention because of their enhanced selectivity, milder
reaction conditions, and associated ease of manipulation.2
Iodosobenzene (PhIO)3 and iodobenzene diacetate [PhI(O-
Ac)2]4 are commonly used single oxygen atom donors in
* Corresponding authors. (H.M.A.) E-mail: heiddy2000@yahoo.com; tele-
phone: +55 21 25627248; fax: +55 21 25627559. E-mail: (O.A.C.A.):
E-mail: octavio@iq.ufrj.br; telephone: +55 21 25627818; fax: +55 21
25627559.
† Instituto de Qu´ımica, UFRJ.
‡ Escola de Qu´ımica, UFRJ.
Results and Discussion
§ Nu´cleo de Pesquisas de Produtos Naturais.
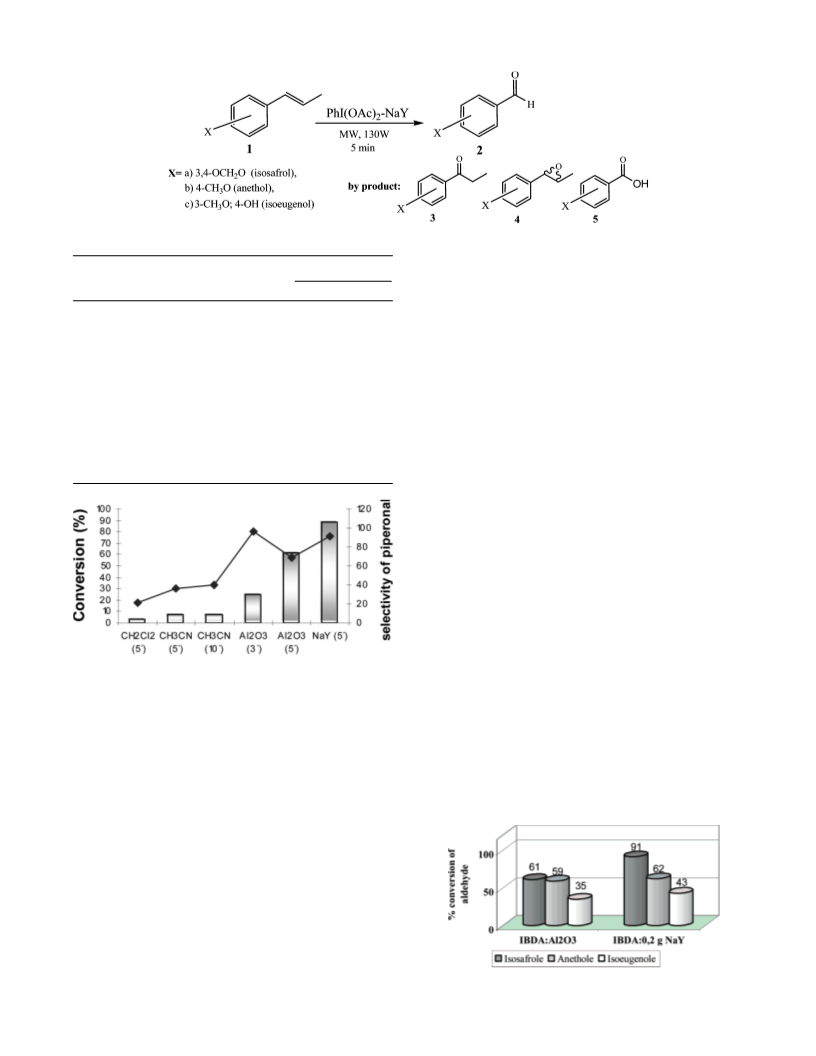
The oxidation of piperonal, p-anisaldehyde, and vanillin
to the corresponding aldehydes using solventless supported
PhI(OAc)2 as reaction media was carried out (Scheme 1).
(1) (a) Choudary, B. M.; Reddy, P. N. J. Mol. Catal. A 1995, 103, L1-L3. (b)
Gekhman, A. E.; Stolarov, I. P.; Moiseeva, N. I.; Rubaijlo, V. L.; Vargaftik,
M. N.; Moiseevi, I. I. lnorg. Chim. Acta 1998, 275-276, 453-461. (c)
Benhaliliba, H.; Derdour, A.; Bazureau, J.-P.; Texier-Bouilet, F.; Hamefin,
J. Tetrahedron Lett. 1998, 39, 541-542. (d) Costa, P. R. R. Qu´ım. NoVa
2000, 23(3), 357-369.
(3) (a) Samsel, E. G.; Srinivasan, K.; Kochi, J. K. J. Am. Chem. Soc. 1985,
107, 7606-7617. (b) Srinivasan, K.; Kochi, J. K. Inorg. Chem. 1985, 24,
4671-4679. (c) Bressan, M.; Morvillo, A. Inorg. Chem. 1989, 28, 950-
953. (d) Meunier, B. Chem. ReV. 1992, 92, 1411. (e) Ryan, K. M.; Bousquet,
C.; Gilheany, D. G. Tetrahedron Lett. 1999, 40, 3613-3616. (f) Guimara˜es,
C. A.; Santos, M. C.; Moraes, M. Qu´ım. NoVa 2004, 27, 199-205.
(4) (a) Adam, W.; Hajra, S.; Herderich, M.; Saha-Mo¨ller, C. R. Org. Lett. 2000,
2, 2773-2776. (b) Karthikeyan, G.; Perumal, P. T. Synlett 2003, 14, 2249-
2250. (c) Li, Z.; Xia, C.-G.; Ji, M. Appl. Catal., A 2003, 252, 17-21. (d)
Park, S.-E.; Song, W. J.; Ryu, Y. O.; Lim, M. H.; Song, R.; Kim, K. M.;
Nam, W. J. Inorg. Biochem. 2005, 99, 424-431.
(5) (a) Sharefkin, J. G.; Saltzman, H. Org. Synth. 1963, 5, 660. (b) Sharefkin,
J. G.; Saltzman, H. Org. Synth. 1963, 5, 658.
(6) (a) Collman, J. P.; Chien, A. S.; Eberspacher, T. A.; Brauman, J. I. J. Am.
Chem. Soc. 2000, 122, 11098-11100. (b) Varma, R. S.; Dahiya, R.; Saini,
R. K. Tetrahedron Lett. 1997, 38, 7029-7032.
(2) (a) Palombi, L.; Bonadies, F.; Scettri, A. Tetrahedron 1997, 53, 15867-
15876. (b) Varma, R. S.; Dahiya, R. Tetrahedron Lett. 1997, 38, 2043-
2044. (c) Varma, R. S.; Dahiya, R. Tetrahedron Lett. 1998, 39, 1307-
1308. (d) Bogdal, D.; L-ukasiewicz, M. Synlett 2000, 1, 143-145. (e)
Alvarez, H. M.; Plut´ın, A. M; Rodr´ıguez, Y.; Perez, E.; Loupy, A.
Synth.Commun. 2000, 30, 1067-1069. (f) Alvarez, H. M.; Perez, E.; Plut´ın,
A. M; Morales, M.; Loupy, A. Tetrahedron Lett. 2000, 41, 1753-1755.
(g) Varma, R. S. Pure Appl. Chem. 2001, 73, 193-198. (h) Shaabani, A.;
Bazgir, A.; Teimouri, F.; Lee, D. G. Tetrahedron Lett. 2002, 43, 5165-
5167. (i) Loupy, A. C. R. Chim. 2004, 7, 103-112. (j) Graebin, C. S.;
Lima, V. L. E. Quim. NoVa 2005, 28, 73-76. (k) Loupy, A. Top. Curr.
Chem. 1999, 206, 153. (l) Loupy, A., Ed. MicrowaVes in Organic Synthesis;
Wiley-VCH: New York, 2003. (m) Lidstro¨m, P., Tierney, J.P., Eds.
MicrowaVe Assisted Organic Synthesis; Blackwell: Oxford, 2004. (n)
Stadler, A.; Yousefi, B. H.; Dallinger, D.; Walla, P.; Van der Eycken, E.;
Kaval, N.; Kappe, C. O. Org. Process Res. DeV. 2003, 7, 707. (o) Kappe,
C. O. Angew. Chem., Int. Ed. 2004, 43, 6250-6284.
(7) In, J.-H.; Park, S.-E.; Song, R.; Nam, W. Inorg. Chim. Acta 2003, 343,
373-376.
10.1021/op060117t CCC: $33.50 © 2006 American Chemical Society
Published on Web 08/16/2006
Vol. 10, No. 5, 2006 / Organic Process Research & Development
•
941

 Alvarez, Heiddy Marquez
Alvarez, Heiddy Marquez
 Barbosa, Dayse P.
Barbosa, Dayse P.
 Fricks, Alini Tinoco
Fricks, Alini Tinoco
 Aranda, Donato A. G.
Aranda, Donato A. G.
 Valdes, Ricardo H.
Valdes, Ricardo H.
 Antunes
Antunes