Evaluation Only. Created with Aspose.PDF. Copyright 2002-2021 Aspose Pty Ltd.
route from Na[Eu(S2CNEt2)4]?3.5H2O with white LED irradia-
tion. To the best of our knowledge, this is the first example of EuS
particles from SSP which have luminescent properties. The white
LED system also differs from UV irradiation systems and has
significant advantages including low cost and improved safety.
This new route may open the way to the creation of nanostructures
and avoid the use of high temperatures and toxic substances.
Studies are being carried out to fabricate EuS thin films by
decomposing the SSP in a suitable CVD process.
S. Y. would like to thank NEDO, Japan and P. O. B. thanks
EPRSC, UK for support.
Yasuchika Hasegawa,a Mohammad Afzaal,b Paul O’Brien,*b
Yuji Wadaa and Shozo Yanagida*a
aMaterial and Life Science, Graduate School of Engineering, Osaka
University, 2-1 Yamadaoka, Suita, Osaka, 565-0871, Japan.
E-mail: yanagida@mls.eng.osaka-u.ac.jp; Fax: (+81)6 6879 7875;
Tel: (+81)6 6879 7926
bThe Manchester Materials Science Centre and the Department of
Chemistry, University of Manchester, Oxford Road, Manchester, UK
M13 9PL. E-mail: paul.obrien@man.ac.uk; Fax: (+44)161 275 4598;
Tel: (+44)161 275 4653
Notes and references
{ Na[Eu(S2CNEt2)4]?3.5H2O was prepared according to the reported
method8 and characterized. All manipulations and reactions were carried
out in air. Analysis of C12H31N4O3.5NaS8Eu: calcd. (found) %: C 28.00
(27.64), H 5.87 (5.45), N 6.53 (6.42), Eu 17.71 (17.95).
§ White LED irradiation (3 V AC, 9000 cd m22, Marks & Spencer) was
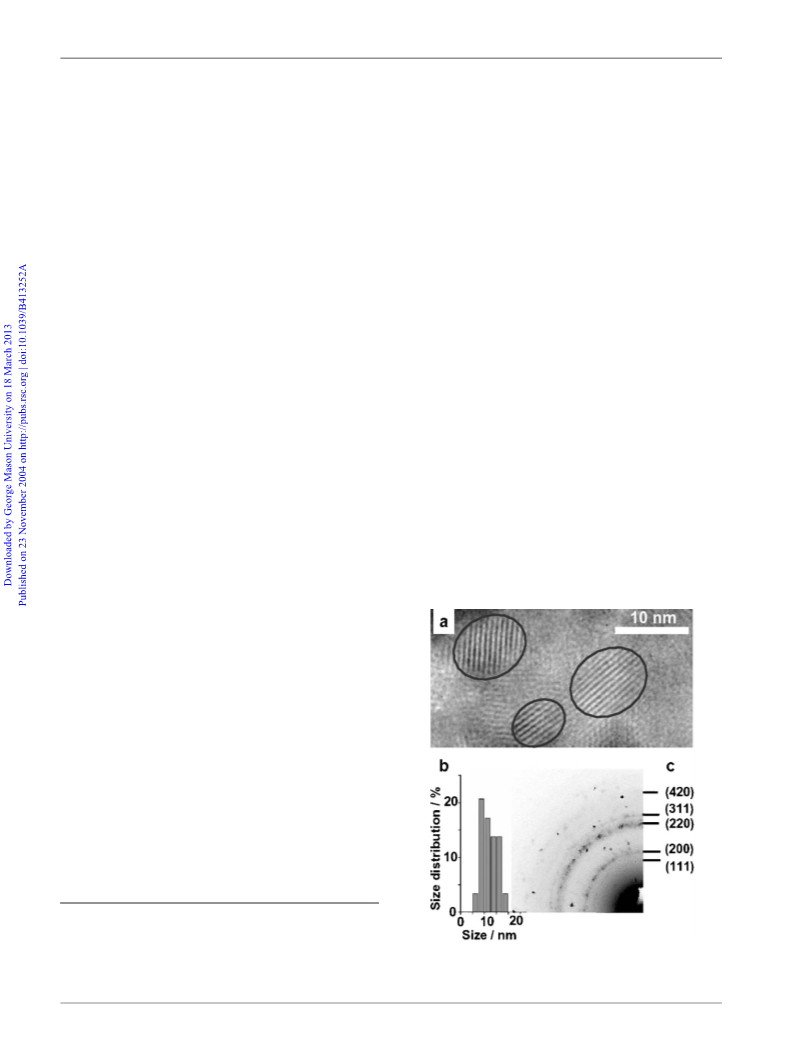
employed during the experiment. TEM and EDAX analyses of prepared
EuS nanocrystals were carried on Philips CM200, 200 kV DX4EDS. The
sample was briefly sonicated in acetonitrile, and then a drop of suspension
was placed on a TEM grid and allowed to dry. Absorption spectra were
measured by Thermo Spectronic Helios b. Solutions in quartz cells (optical
path length 5 mm) were excited at 345 nm using a spectrometer (JOBIN
YVON SPEX FL3-22). The spectra presented here have been corrected for
detector sensitivity and lamp intensity. The quantum yields were
determined by standard procedures using an integrating sphere and quartz
cells (optical path length 5 mm).10
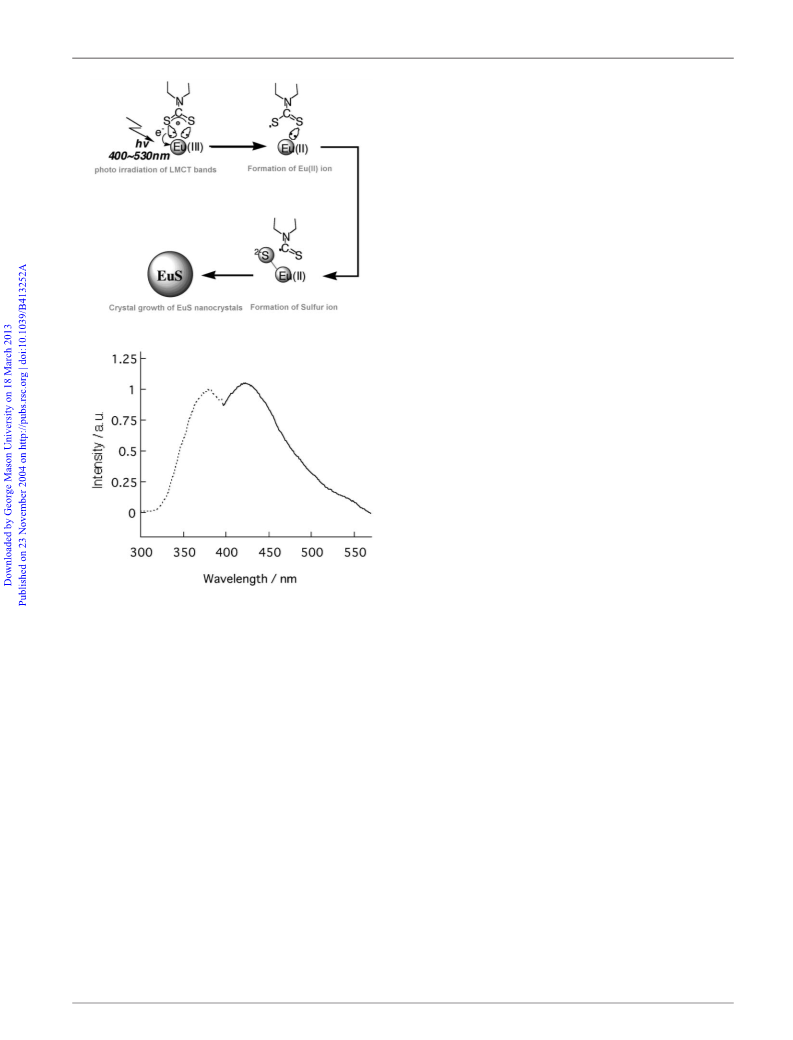
Fig. 2 The photochemical reaction mechanism, excitation (dotted line:
monitor at 470 nm) and emission spectra (bold line: excited at 370 nm) of
the EuS nanocrystals in acetonitrile, respectively.
1 P. Wachter, Handbook on the Physics and Chemistry of Rare Earths, 2nd
end., North-Holland Publishing Company, 1979, P. 189.
2 P. Fumagalli, A. Schirmeisen and R. J. Gambino, J. Appl. Phys.,
1996, 79, 5929; S. Thongchant, Y. Hasegawa, K. Tanaka, K. Fujita,
K. Hirao, Y. Wada and S. Yanagida, Jpn. J. Appl. Phys., 2003, 42,
L876.
solution including EuS nanoparticles is also due to an f–d
transition. The energy band gap in nanocrystals (of ca. 10 nm) was
estimated to be about 3.1 eV, a blue shift in comparison to bulk
EuS (1.7 eV)9 and is significantly larger than that observed for the
20 nm EuS particles (1.9 eV).3 The level of the conduction band
constructed from 5d orbitals should be affected by the crystal size
of EuS (a quantum size effect). The 10 nm semiconductor particles
confine the excited electron in the conduction band, resulting in an
emission.9 The quantum yield for EuS particles was found to be
only 0.05%. This observation represents the first example of the
luminescence of EuS at room temperature. Since the emission
depends on the surface condition of the EuS nanoparticles, work is
being carried out to modify the EuS surface in order to enhance
the emission.§
3 S. Thongchant, Y. Hasegawa, Y. Wada and S. Yanagida, J. Phys.
Chem. B., 2003, 107, 2193.
4 M. Green and P. O’Brien, Adv. Mater., 1998, 10, 527; P. O’Brien and
N. L. Pickett, Chem. Record, 2001, 467; N. Revaprasadu, M. A. Malik,
P. O’Brien and G. Wakefield, Chem. Commun., 1999, 1573.
5 G. G. Scholes, T. Mirkovic and M. A. Hines, 205th Meeting of the
Electrochemical Society, 2004, abstract 636.
6 Y. Hasegawa, S. Thongchant, Y. Wada, H. Tanaka, T. Kawai,
T. Sakata, H. Mori and S. Yanagida, Angew. Chem., Int. Ed., 2002, 41,
2073.
7 Y. Hasegawa, S. Thongchant, T. Kataoka, Y. Wada, T. Yatsuhashi,
N. Nakashika and S. Yanagida, Chem. Lett., 2003, 32, 708.
8 T. Kobayashi, H. Naruke and T. Yamase, Chem. Lett., 1997, 907.
9 D. E. Eastman, F. Holtzberg and S. Methfessel, Phys. Rev. Lett., 1969,
23, 226.
The above results demonstrate that reasonably monodispersed
EuS nanoparticles can be prepared by a simple and convenient
10 G. D. Stucky and J. E. MacDougall, Science, 1990, 247, 669.
This journal is ß The Royal Society of Chemistry 2005
Chem. Commun., 2005, 242–243 | 243

 Hasegawa, Yasuchika
Hasegawa, Yasuchika
 Afzaal, Mohammad
Afzaal, Mohammad
 O'Brien, Paul
O'Brien, Paul
 Wada, Yuji
Wada, Yuji
 Yanagida, Shozo
Yanagida, Shozo