410
Chemistry Letters Vol.37, No.4 (2008)
Eu2þ-doped Thioaluminates: New Candidates for White LEDs
Ruijin Yu, Jing Wang,ꢀ Xinmin Zhang, Haibin Yuan, Jianhui Zhang, and Qiang Suꢀ
MOE Laboratory of Bioinorganic and Synthetic Chemistry, State Key Laboratory of Optoelectronic Materials and Technologies,
School of Chemistry and Chemical Engineering, Sun Yat-sen University, Guangzhou, Guangdong 510275, P. R. China
(Received November 30, 2007; CL-071323; E-mail: ceswj@mail.sysu.edu.cn)
Three Eu2þ-doped thioaluminate phosphors with different
light-emitting colors, green, bluish-green, and blue were synthe-
sized by the evacuated sealed quartz ampoule method. Under
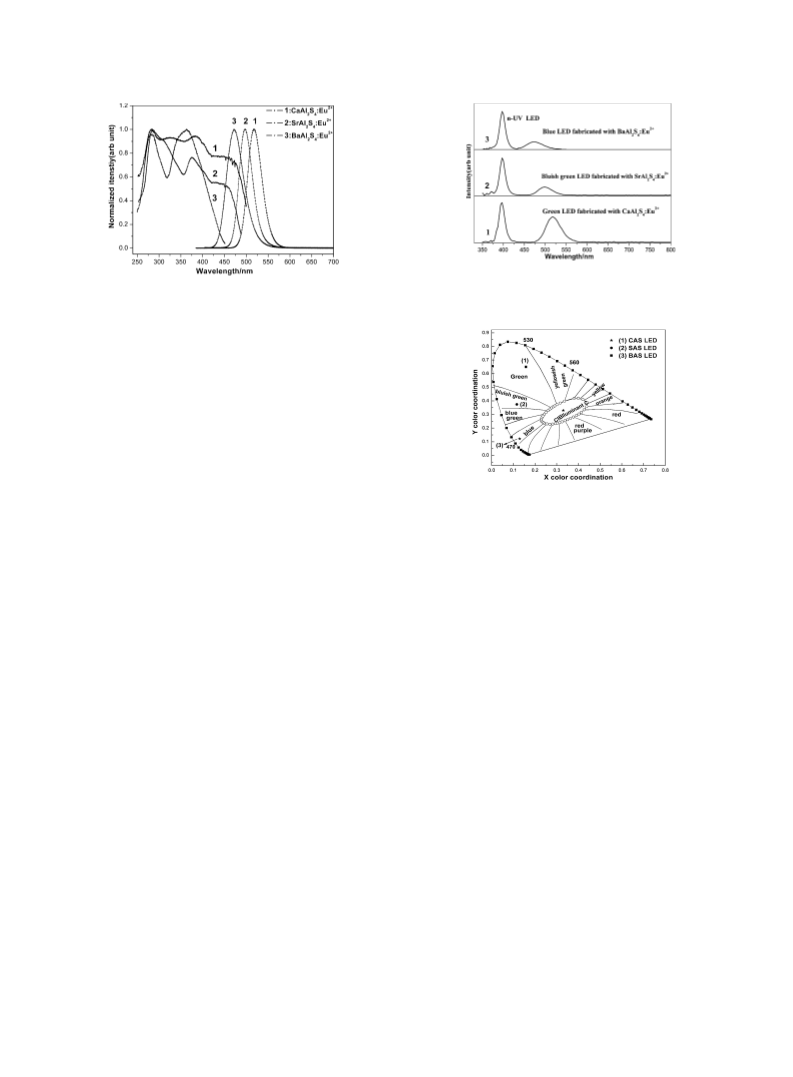
UV excitation, CaAl2S4:Eu2þ (CAS), SrAl2S4:Eu2þ (SAS),
and BaAl2S4:Eu2þ (BAS) exhibit strong emission bands peaked
at 516, 496, and 470 nm, respectively. The fabricated LEDs of
CAS, SAS, and BAS show intense green, bluish-green, and blue
lighting. They are all believed to be good phosphor candidates
for creating white light in phosphor-converted white LEDs.
phosphors.
The starting sulfide materials CaS, SrS, BaS, and EuS were
pre-prepared by a solid-state reaction method at high tempera-
ture in horizontal tube furnaces. Alkaline earth sulfides were
prepared under flowing H2S gas at 1000 ꢁC for 2 h. EuS was
prepared from Eu2O3 (99.99%) with CS2 reducing atmosphere
at 1200 ꢁC for 3 h.
The stoichiometric amounts of the starting sulfide MS
(M = Ca, Sr, and Ba), Al (AR), EuS, and 25 mass % excess S
(AR) were thoroughly mixed and put in quartz ampoules and
then evacuated to 1 ꢂ 10ꢃ6 Torr and sealed, finally fired at
1050 ꢁC for 5 h.
Compared with reported preparation methods,8,9 the cheap
and stable aluminum powder was first used as the starting
material instead of expensive and weakly hygroscopic Al2S3
to prepare MAl2S4:Eu2þ under vacuum in sealed silica tubes.
The structure of the final products was examined by X-ray
powder diffraction using a Rigaku D/max 2200 vpc X-ray
diffractometer with Cu Kꢀ radiation at 40 kV and 30 mA. The
photoluminescence (PL) and photoluminescence excitation
(PLE) spectra of phosphors were measured by a Fluorolog-3
spectrofluorometer (Jobin Yvon Inc/specx) equipped with a
450-W Xe lamp and double excitation monochromators. The
emission spectra of the LEDs were recorded on an LED-1100
spectral/goniometric analyzer (Labsphere Inc.) under a direct
current of 20 mA. The above measurements were carried out
at room temperature.
The XRD patterns of CaAl2S4:0.10Eu2þ (CAS), SrAl2S4:
0.10Eu2þ (SAS), BaAl2S4:0.10Eu2þ (BAS) phosphors are
shown in Figure 1. The XRD patterns of CAS, SAS, and BAS
match well with 77–1186, 77–1188, and 76–1054 by JCPDS
fingerprints. No second phases are observed indicating that
all samples show a single phase. It is concluded that the
cheap and stable aluminum powder can be used as a good
substitute for aluminum sulfide (Al2S3) to prepare pure
thioaluminates.
Recently, the Eu2þ-doped thioaluminates have attracted
more and more attention owing to their excellent electrolumines-
cent (EL) performance. BaAl2S4:Eu2þ was introduced as a
bright and saturated blue-emitting phosphor for full-color EL
devices.1 Thereafter, the iFire Company adopted it as a new
blue source and announced a full-color 34-in. thick dielectric
electroluminescent (TDEL) screen using the Color-By-Blue
(CBB) technique.2 In 2003, CaAl2S4:Eu2þ was also reported
as a green-emitting thin film for EL applications with consider-
able high luminance 3041 cd/m2 at a driving frequency of
1 kHz.3 Since the absorptions of the 4f–5d transitions of the
Eu2þ-doped thiogallates and thioaluminates extend to the
visible region, they are also appropriate phosphors for excitation
by near-UV or blue-emitting diodes for solid-state lighting
(SSL) applications.4 CaGa2S4:Eu2þ and Sr2Ga2S5:Eu2þ green-
ish-yellow phosphors showed a higher luminescent efficiency
(120 and 110%, respectively) than the commercial YAG:Ce3þ
phosphor, and they also can be fabricated with blue-chips to
produce white light.5,6
This paper describes the photoluminescence properties and
application of the thioaluminates MAl2S4:Eu2þ (M = Ca, Sr,
and Ba) in white LEDs. This family of materials was chosen
for the following reasons:7,8 (i) Eu2þ-doped thioaluminates show
a broad absorption band extending from the near ultraviolet to
the blue region which was a satisfaction to the requirements
for LED fabrication, (ii) Eu2þ-doped thioaluminates have small-
er Stokes shifts and narrower emission bands than Eu2þ-doped
thiogallates, (iii) luminescence quenching temperatures are
markedly higher for thioaluminates than for thiogallates. It is
favorable for reducing the temperature effect brought by the
LED’s p–n junctions. All the characteristics indicate that
Eu2þ-doped thioaluminates exhibit possibly a high luminous
output. Nevertheless, Eu2þ-doped thioaluminates as high
efficient phosphors have not been applied in the field of
phosphor-converted white-light-emitting diodes (pc-WLED)
up to now.
In this work, MAl2S4:Eu2þ (M = Ca, Sr, and Ba) phosphors
were synthesized and the optical properties of the phosphors
were investigated. Finally, three intense light-emitting LEDs
were fabricated through combining InGaN chips with these
Figure 1. XRD patterns of MAl2S4:Eu2þ (M = Ca, Sr,
and Ba).
Copyright Ó 2008 The Chemical Society of Japan

 Yu, Ruijin
Yu, Ruijin
 Wang, Jing
Wang, Jing
 Zhang, Xinmin
Zhang, Xinmin
 Yuan, Haibin
Yuan, Haibin
 Zhang, Jianhui
Zhang, Jianhui
 Su, Qiang
Su, Qiang