PHASE DIAGRAMS OF SECTIONS
797
parameters of the hexagonal unit cell of
β
ꢀCu2S are
а
=
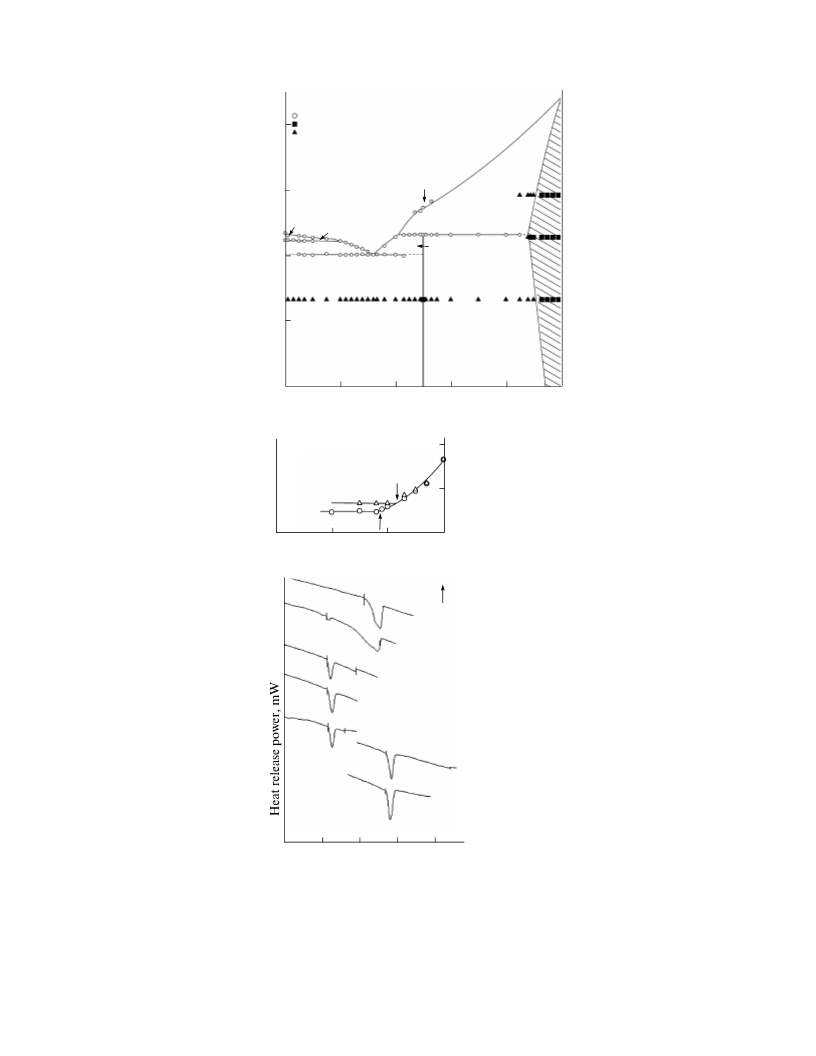
In the NdCuS2–EuNd2S4 system at the isothermalꢀ
section temperature, the NdCuS2 phase and the
γ
0.396 nm, = 0.678 nm [16]. Within the solidꢀsolution
с
phase are in equilibrium. Xꢀray powder diffraction
pattern showed no noticeable regions of homogeneity.
No changes were detected in the unit cell parameter of
the conjugate phases in the twoꢀphase region. On poliꢀ
shed sections, there are grains of the NdCuS2 phase
region, there is scatter of experimental values for the
Cu2S unit cell parameters as functions of composition.
For 5.9 mol % EuNdCuS3, the unit cell parameters are
а
= 0.395 nm,
с = 0.674 nm.
According to microstructural analysis, samples
(
H
= 3350 MPa), located inside the fields of phase
γ
containing up to 4.6 mol % EuNdCuS3 are homogeꢀ
neous. In samples containing 5.9 and 7.5 mol %
EuNdCuS3, the field of the Cu2S phase is pierced
throughout its volume by needleꢀshaped intrusions of
crystals (
Н
= 5520 MPa).
ACKNOWLEDGMENTS
10–100ꢀ
μ
mꢀlong, 2–5ꢀ mꢀwide grayꢀbrown crystals
μ
This work was supported by the Federal Target Proꢀ
gram “Research and ResearchꢀTeaching Staff in
Innovative Russia” for 2009–2013 (state contract
nos. 6K/143ꢀ09 (P646) and NKꢀ409/5 (P2263)).
of the conjugate phase EuNdCuS3; there is no eutectic.
The shape, size, and distribution of grains of the
EuNdCuS3 phase suggest solidꢀphase decomposition
of the primary
βꢀCu2S solid solution. In an initially
homogeneous sample (5.9 mol % EuNdCuS3
)
annealed at 970 K, needleꢀshaped crystals of the
EuNdCuS3 phase appear, which is indicative of the
temperature dependence of solubility in solid βꢀCu2S.
REFERENCES
1. O. V. Andreev, Extended Abstract of Doctoral Dissertaꢀ
tion in Chemistry (Tyumen, 1999).
Within the homogeneity region, the Cu2S microꢀ
hardness decreases from 1470 to 990 MPa (Fig. 3b),
which is indicative of incorporation of ions having a
2. M. Sato, G. Y. Adachi, and J. Shiokawa, J. Solid State
Chem. 31, 337 (1980).
3. A. A. Eliseev, O. A. Sadovskaya, and G. M. Kuzmicheva,
larger radius (
r
Eu2+(VII) = 0.12 nm [12]).
J. Inorg. Mater. (Engl. Transl.) 18, 1435 (1982).
A eutectic is formed between
β
ꢀCu2S ss and
4. A. V. Ruseikina and O. V. Andreev, Proceedings of the
AllꢀRussia Scientific Conference “SolidꢀState Chemistry
and Functional Materials” (Yekaterinburg, 2008), p.
304 [in Russian].
EuNdCuS3 phases. The peak of melting of the eutectic
has a pronounced linear portion and is detected at an
average temperature of 1142 K. As the eutectic composiꢀ
tion is approached, the sizes of Cu2S primary yellow
grains and EuNdCuS3 grayꢀbrown grains systematically
5. A. V. Rusejkina, O. V. Andreev, and N. A. Khritokhin,
Proceedings of XVII International Conference on Chemiꢀ
cal Thermodynamics in Russia (Kazan, 2009), Vol. 2,
p. 306 [in Russian].
decrease from 200
from 900 50 to 60
of the phases in the eutectic are 10
×
30 to 60
×
20
Н
μ
m (
= 2320 MPa). Crystals
20 m in size.
Н= 990 MPa) and
×
×
20 m (
μ
×
μ
6. V. G. Bamburov and O. V. Andreev, Zh. Neorg. Khim.
47 (4), 676 (2002) [Russ. J. Inorg. Chem. 47 (4), 598
(2002)].
According to the Tammann triangle construction and
microstructural analysis, the eutectic composition was
taken to be 20.5 mol % EuNdCuS3, with the heat of
melting being 12 J/g. A phase transformation equation
was composed (table).
7. A. V. Ruseikina, A. S. Zalineeva, and O. V. Andreev, in
Physicochemical Analysis of Natural and Engineering
Systems: Collected Works (Tyumen, 2008), p. 159 [in
Russian].
Between the EuS + L and EuNdCuS3 + L fields in
the system under investigation, there should be a EuS +
EuNdCuS3 + L field (outlined by dashed line). DSC
detected a weak decrease in the EuNdCuS3 melting
point from 1470 to 1464 K. The coordinates of the
point of intersection of the liquidus line and the line of
phase decomposition of the compound EuNdCuS3 are
8. J. W. Visser, J. Appl. Crystallogr. 2, 89 (1996).
9. L. A. Solovyov, J. Appl. Crystallogr. 37, C. 743 (2004).
10. A. E. Christuk, P. Wu, and J. A. Ibers, J. Solid State
Chem. 110, 330 (1994).
11. P. Wu, A. E. Christuk, and J. A. Ibers, J. Solid State
Chem. 110, 337 (1994).
43.0 mol % EuNdCuS3 and
Т = 1464 K.
12. R. D. Shannon, Acta Crystallogr. 32, 751 (1976).
In the EuNdCuS3–EuNd2S4 system, EuNdCuS3
and EuNd2S4 phases are in equilibrium at the isotherꢀ
malꢀsection temperature of 970 K. No change was
detected in the unit cell parameter within the twoꢀ
phase region relative to the individual compounds,
which is indicative of the absence of reciprocal solid
solutions. A eutectic is formed between the phases,
and the melting peak was detected at 1467 K and a
13. Database “The Thermal Constants of materials”
14. L. G. Gorbunova, Extended Abstract of Candidate’s
Dissertation in Chemistry (Novosibirsk, 1990).
15. Phase Diagrams for Binary Metallic Systems: A Handꢀ
book, Ed. by N. P. Lyakishev (Mashinostroenie, Mosꢀ
cow, 1996), Vol. 1, p. 475 [in Russian].
composition of ~(30.0 3.0) mol % EuNd2S4
.
16. O. Andreev, Russ. J. Inorg. Chem. 34, 909 (1989).
RUSSIAN JOURNAL OF INORGANIC CHEMISTRY Vol. 56 No. 5 2011

 Andreev
Andreev
 Ruseikina
Ruseikina
 Solov'Ev
Solov'Ev