
Canadian Journal of Chemistry p. 394 - 404 (2001)
Update date:2022-08-11
Topics:
 Gaisberger
Gaisberger
 Solar
Solar
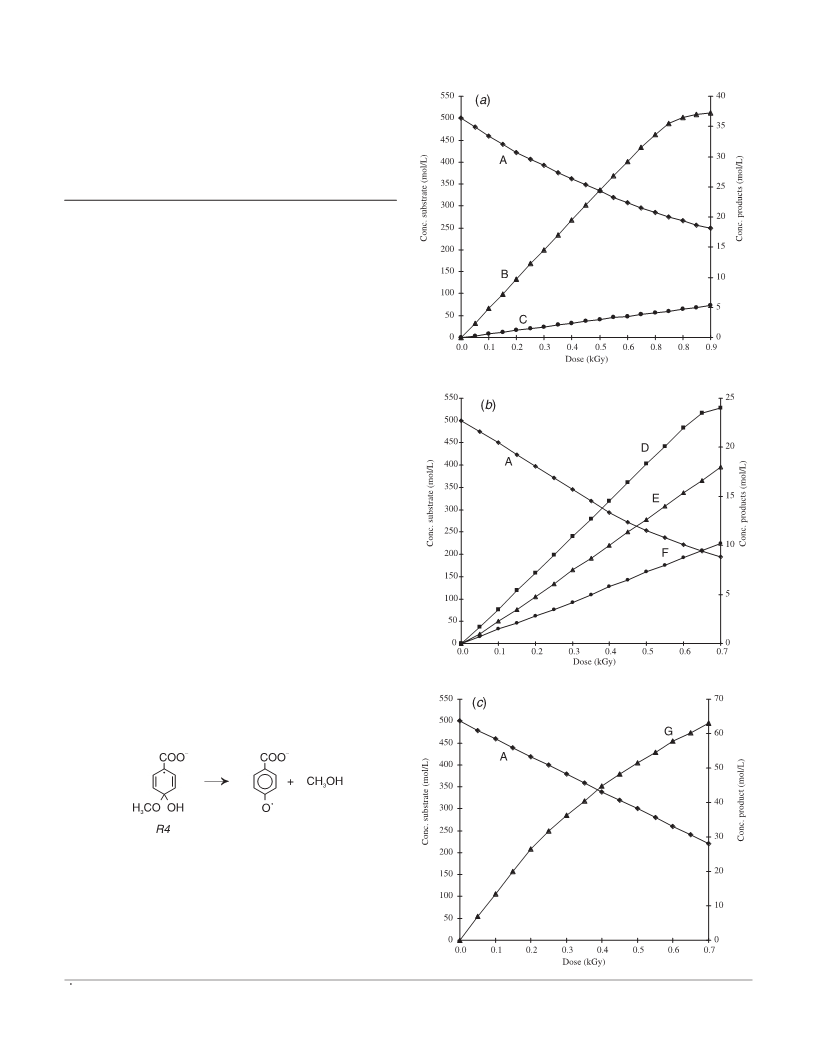
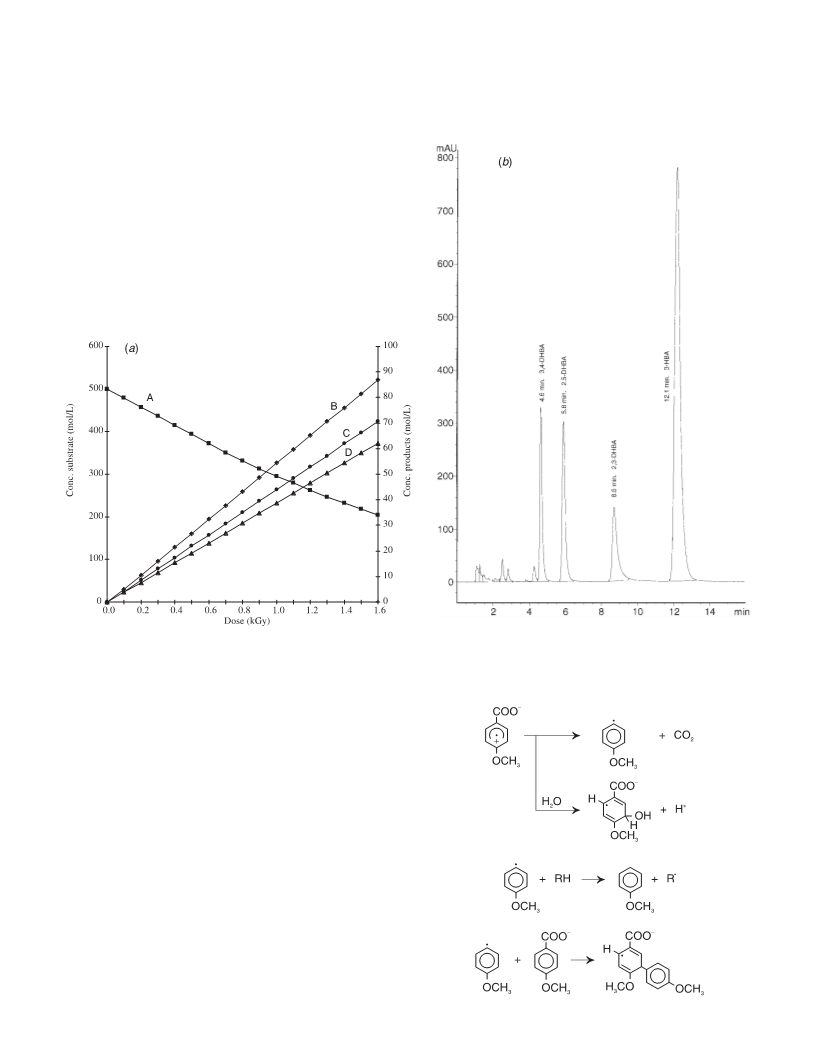
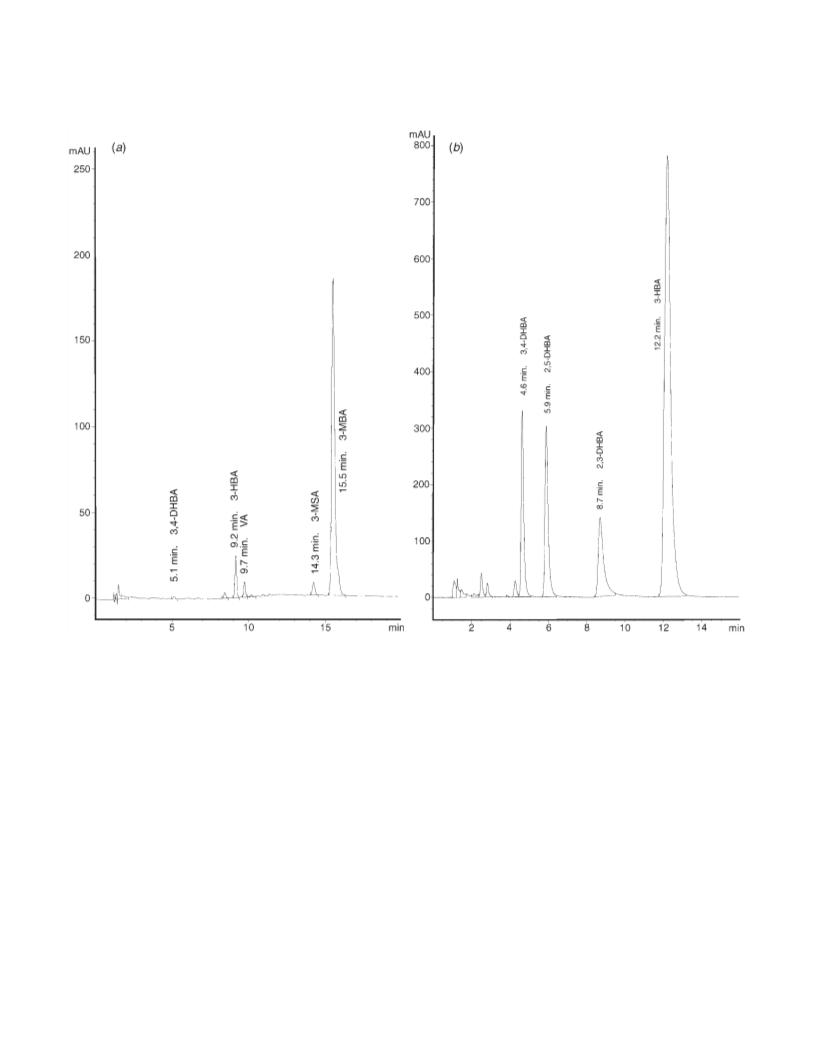
The hydroxylation process for methoxy- and hydroxy-benzoic acids (MBA, HBA) induced by γ-radiation is compared. 2-, 3-, and 4-methoxybenzoic acid as well as 3-hydroxybenzoic acid have been irradiated in N2O and aerated solutions up to 1.5 kGy. The products were analyzed by HPLC. The results for 2- and 4-HBA have been taken from literature data. The OH·-adduct distribution is generally the same for the hydroxy- as well as for the methoxy-benzoic acid isomers. With both 4-HBA and 4-MBA more than 65% C3-adducts and about 15% C4-adducts are formed, which could be proved by their reactions with K3 Fe(CN)6. Oxidation of the nonipso-adducts of 3-HBA and 3-MBA results in 84 and 87% of the corresponding phenols. Whereas in N2O-saturated solutions only part of the OH·-radicals leads to substrate decomposition, in the presence of air, the degradation of both kinds of compounds is equivalent to [OH·]. The nonipso OH·-adducts of the HBAs are converted into 68-77% hydroxylation products. With the MBAs, the hydroxylation process is ≤10%. This is attributed to different decay pathways of the peroxyl radicals, intermediates formed by O2 addition to the OH·-adducts. The hydroxyperoxycyclohexadienyl radicals of the HBAs decay mainly by HO2· elimination to the corresponding phenols, those of the MBAs decay predominantly by fragmentation of the benzene ring, yielding to nonidentified aliphatic products. The replacement of -OCH3 by -OH is practically not influenced by the presence of oxygen, it increases in the sequence 3-MBA < 4-MBA < 2-MBA. For 2-MBA, yields of more than 15% are obtained. Both processes, hydroxylation as well as demethoxylation, might be of importance for the recognition of radiolytical changes in foodstuff.
View More






Jiangsu Chunjiang AgroChemical Co.,Ltd.
Contact:+86-13817348163
Address:Panjiawan 84, Zhixi Town, Jintan City, China
Qingdao XinYongAn Chemicals Co., Ltd
Contact:+86-532-81107967
Address:Chengyang dual-port industrial park by the sea,Qingdao
Improve Medical Technology(Nanxiong) Co., Ltd
Contact:86-751-3836997
Address:No.33, Pingan First Road, Fine Chemical Industry Base, Nanxiong City, Shaoguan, Guangdong, China
Contact:86-512-69362780,69362785
Address:No.69 Weixin Road,Weiting Town,Suzhou Industrial Park
Laohekou Jinghong Chemical Co.,Ltd
Contact:+86-0710-3702747
Address:163.East,Huagong Road,Laohekou
Doi:10.1016/0040-4020(95)00921-T
(1995)Doi:10.1007/BF00853700
()Doi:10.1002/zaac.202100093
(2021)Doi:10.1016/j.molstruc.2020.127948
(2020)Doi:10.1134/S1070363215050163
(2015)Doi:10.1016/S0022-328X(00)89864-2
(1980)