
Molecules p. 5319 - 5338 (2012)
Update date:2022-08-16
Topics:
 Ignat'ev, Nikolai V.
Ignat'ev, Nikolai V.
 Barthen, Peter
Barthen, Peter
 Kucheryna, Andryi
Kucheryna, Andryi
 Willner, Helge
Willner, Helge
 Sartori, Peter
Sartori, Peter
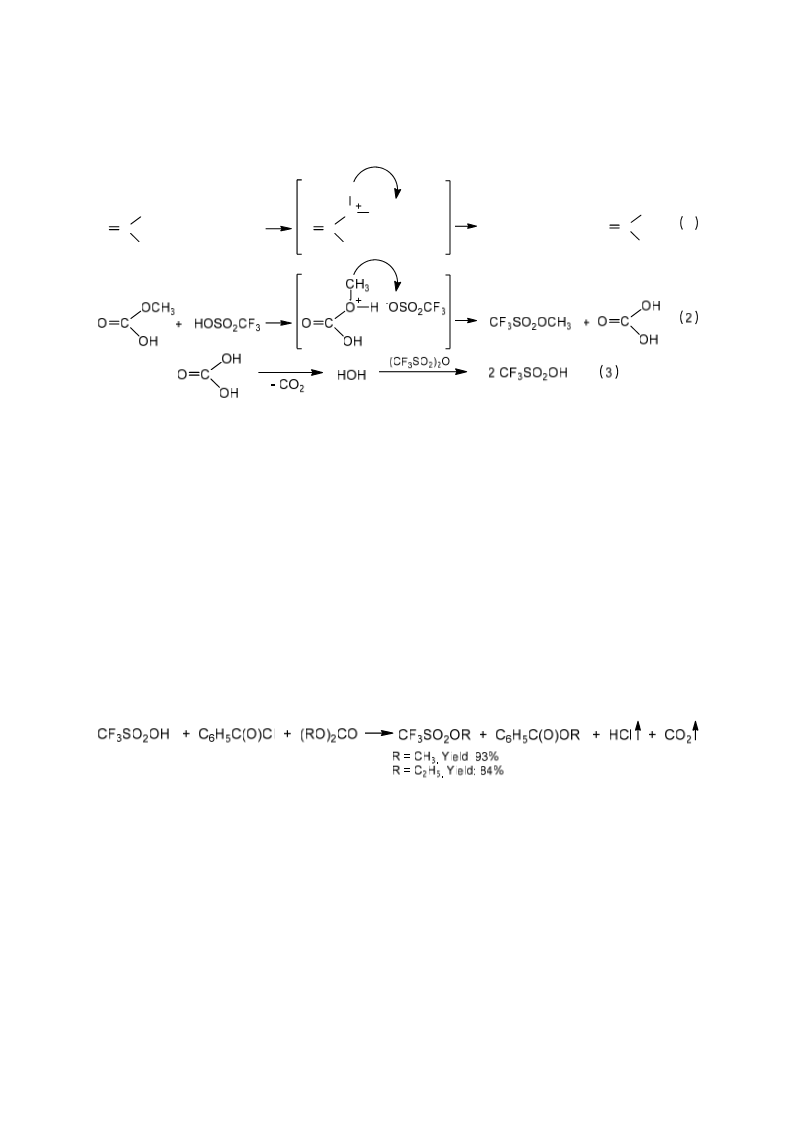
A solvent- and halogen-free synthesis of high purity triflate ionic liquids via direct alkylation of organic bases (amines, phosphines or heterocyclic compounds) with methyl and ethyl trifluoromethanesulfonate (methyl and ethyl triflate) has been developed. Cheap and non-toxic dimethyl and diethyl carbonate serve as source for the methyl and ethyl groups in the preparation of methyl and ethyl triflate by this invented process. The properties of ionic liquids containing the triflate anion are determined and discussed.
View More







Changzhou Welton Chemical Co., Ltd,
Contact:0086-519-85910828,85920537,85912897
Address:No.8 Jinlong Road, Binjiang Park, Changzhou, Jiangsu, China.
Contact:86-25-58619180
Address:Nanjing High-Tech Zone 10 Xinghuo Road Pukou District Nanjing, Jiangsu 210061 The People's Republic of China
website:http://www.guarson.com
Contact:+86-523-88059600,+86-13805268803
Address:Room B1006,Yafang Building,Jiangyan Avenue,Jiangyan District, Taizhou City,Jiangsu,China
Contact:86-28-61993785
Address:No.70-13-21, North Section, Erhuan
Ningxia Soochow Agrochemical Limited Company
Contact:(+86)0512 6320 8190
Address:wujiang
Doi:10.1016/S0009-2614(97)01096-8
(1997)Doi:10.1039/a909548i
(2000)Doi:10.1039/b201112n
(2002)Doi:10.1039/b514583j
(2006)Doi:10.1016/j.phytochem.2019.05.016
(2019)Doi:10.1016/j.ejmech.2007.05.012
(2008)