.
Angewandte
Communications
decomposition. The indophenol assay was also conducted on
metal distances coupled to the size of the ancillary donor
atoms. With respect to the disorder in the structure, we
observe two iron centers, each coordinated h to the internal
crude reduction products of 1 by 3, 4, 5, 6, or 10 equiv KC in
8
6
the presence of N (data not shown). Ammonia was detected
2
in all cases with the highest yield recorded for the reaction
with 6 equivalents.
face of benzene caps of the ligand with low occupancies
(~ 5%; Figure S3). These iron centers could arise from
a nacnac-to-arene shift, which is observed upon reduction of
monometallic nacnac complexes, and suggests that low-valent
We observed consistent morphologies and comparable
unit cell parameters for crystals isolated from several differ-
ent reduction reactions. We therefore selected one of the
crystals from a sample of 2 and determined the molecular
structure (2-1). Disorder is observed in the structure solution;
however, the occupancies of the bridging N atoms N7–N9 and
Fe1 are best modeled as 100% and of Fe2 and Fe3 as 95%. In
this higher occupancy solution, the Fe X core in 2-1, in which
[
15,16]
iron centers are generated upon reduction of 1.
Consistent with NH donors in 2 and 2-1, a weak IR
x
À1
absorption at 3397 cm is observed in spectra of 2 that shifts
À1
15
to 3390 cm
for samples reduced under N2 (2’). This
difference is in excellent agreement with a simple reduced-
mass calculation for an NH bond, and the absorption is at
comparable energy to other reported m-imide and m-amide
3
3
X denotes the donor atom in the bridging ligands, adopts
a planar hexagonal arrangement with each iron center in
[
17–19]
donors.
These IR data and the observed H-atoms in the
[13]
a distorted tetrahedral geometry (t = 0.92–0.94; Figure 1).
difference map provide strong evidence for protonated N2-
derived ligands in the isolated compound. Expectedly, an
absorption feature at coincident energy is also present in IR
spectra of Fe (NH ) L (3) that has similar intensity relative to
4
3
2 3
other ligand vibrations. We also observed a feature at
À1
3
657 cm in the IR spectrum of 2, which is comparable to
[
20]
reported OH vibrations in related compounds.
This
absorption is absent from the crude reaction product, is
present only after crystallization, and is not observed in
spectra of 3 and 4 (Figure S12). Significant incorporation of
OH ligands in 2 is unlikely based on our elemental analysis
data for which C, H, N, and Fe contents account for 99.87% of
the sample mass.
To gain further insight into the electronic structure and
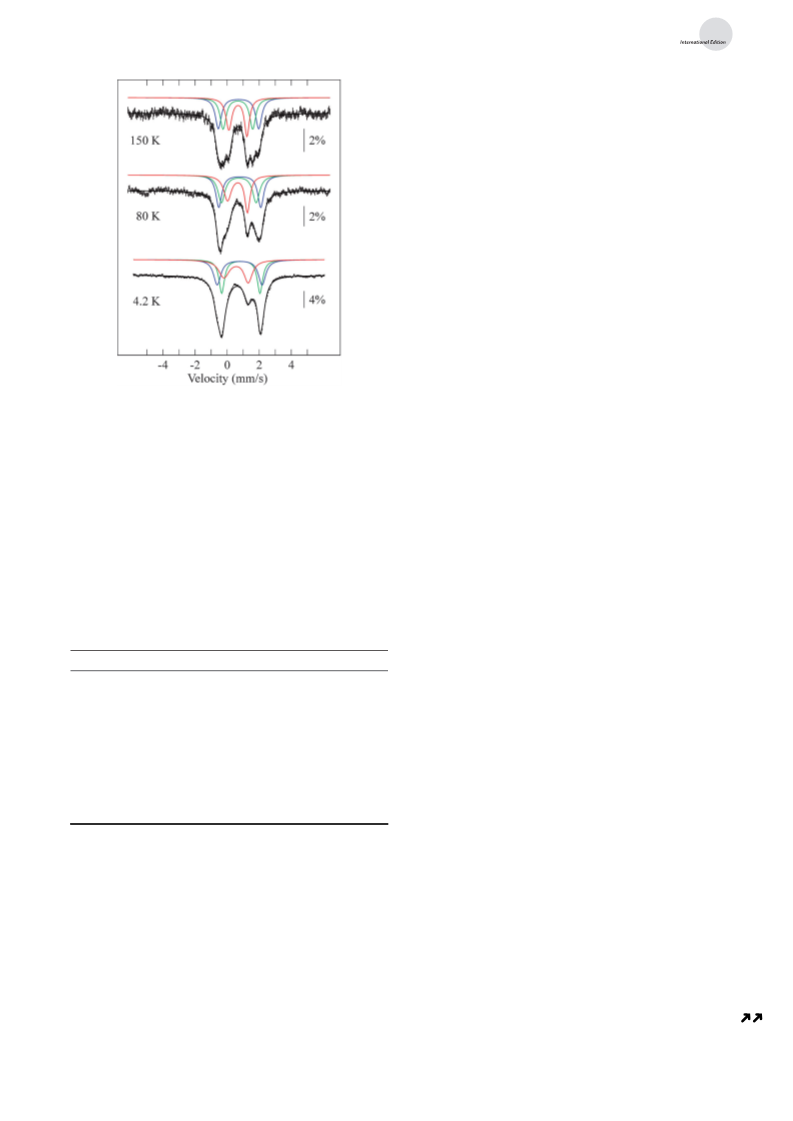
oxidation state of the iron sites, we studied 2 by Mçssbauer
spectroscopy. The zero-field Mçssbauer spectra of 2 are
composed of four quadrupole doublets, each of which
accounts for ca. 25% of total iron in the sample (Figure 2).
The doublet formed by the two outermost absorptions is also
detected in samples of Fe (NH ) L (3) and Fe (OMe) L (4),
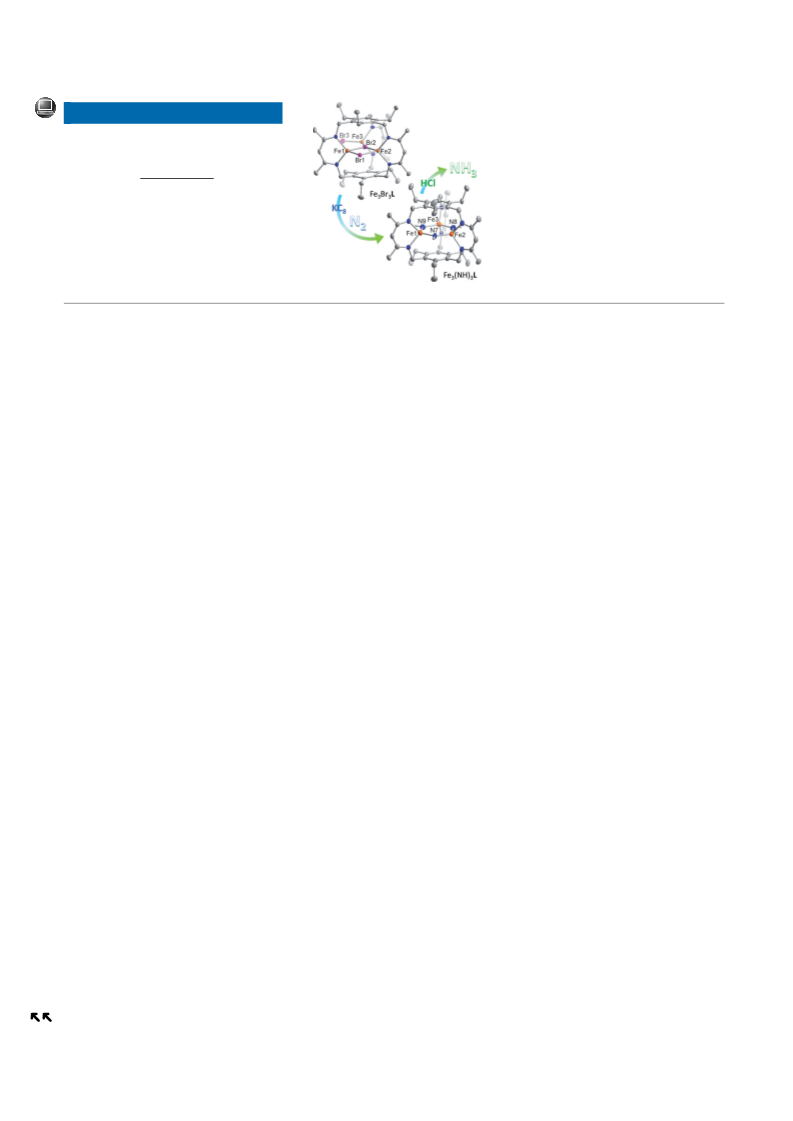
Figure 1. Solid-state structures of 2-1 (left) and 3 (right) at 65%
thermal ellipsoid. The pentane solvent molecule cocrystallized in 2-
1
and hydrogen atoms have been omitted for clarity, except for the
bridging nitrogen-bonded ones. C, H, N, and Fe atoms are depicted as
gray, white, blue, and orange ellipsoids or spheres.
3
2
3
3
3
This structure type is unprecedented in iron cluster chemistry,
but not unexpected given the steric constraints imposed by
our ligand. Similar Fe X arrangements are observed in two
although the intensities of this doublet vary between the
spectra (Figure S8). This common feature is assigned to an
exogenous complex and subtracted from the spectra. This
exogenous species likely arises from complex decomposition
and is not present in spectra collected on the crude reaction
filtrate (i.e., prior to crystallization. Figure S9). The three iron
centers in 2-1 are crystallographically inequivalent, in agree-
ment with the three distinct quadrupole doublets observed in
spectra of 2. Two of these doublets have parameters that are
comparable to those determined for 3, and the third doublet is
3
3
other complexes, 3 and 4, in which the N-atom of the m-amide
and O-atom of the m-methoxide donors are coplanar with the
three iron centers (Figures 1 and S2). In 2-1, the H-atom
bonded to each bridging N-atom ligand was readily located in
the difference map and freely refined, implying a formulation
III
of 2-1 as Fe (NH) L. As anticipated with a higher oxidation
3
3
state of the metal centers in 2-1 as compared to 3, the Fe–mN
bonds (1.960(3)–1.978(3) ꢀ) are shorter than observed for the
III
À1
consistent with an Fe center (d = 0.56 mms , DE =
Q
À1
Fe–NH bonds in 3 (Table S2). Interestingly, the shortening of
1.49 mms at 4.2 K). A careful examination of the temper-
2
Fe–mN bond results in slightly more acute diketiminate
ature-dependence of each doubletꢁs isomer shift in spectra of
(
nacnac) bite angles (~ 958) at each iron center (e.g., N1-
2 (Table 1) indicates that the ferric site contains an admixture
II
2+
Fe1-N2) in 2-1 relative to those in 3 (~ 968), shorter distances
between the two N-atom donors of each nacnac arm
of Fe and vice versa (i.e., one of the Fe sites contains
3
+
2+
3+
admixture of Fe ). This is only possible if the Fe and Fe
(
3
3
2.993(3)–3.009(3) ꢀ for 2-1 versus 3.013(3)–3.034(3) ꢀ for
), and compression of the cyclophane cavity in 2-1 relative to
. The bonds and angles around each iron center in 2-1 and 3
sites are both present in the same complex. This feature is
absent in spectra of the all-ferrous complex 3 (Figure S10).
From our Mçssbauer analysis, we conclude that 2 contains
mainly mixed-valent triiron(II/II/III) complexes, although
other combinations of oxidation states are likely present as
minor products, their doublets largely overlapping with those
of the former. The doublets observed in 2 are present in
Mçssbauer spectra of the crude reaction filtrate but in
are significantly larger than those reported for mononuclear
or self-assembled polynuclear iron b-diketiminate com-
pounds.
at the unique steric effects imposed by the macrobicyclic
ligand, which arise from the limited range of allowable metal–
[
9a,14]
These correlated structural parameters all hint
2
ꢀ 2014 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
Angew. Chem. Int. Ed. 2014, 53, 1 – 6
These are not the final page numbers!

 Lee, Yousoon
Lee, Yousoon
 Sloane, Forrest T.
Sloane, Forrest T.
 Blondin, Genevive
Blondin, Genevive
 Abboud, Khalil A.
Abboud, Khalil A.
 Garca-Serres, Ricardo
Garca-Serres, Ricardo
 Murray, Leslie J.
Murray, Leslie J.