E. Isarain-Chávez et al. / Electrochimica Acta 56 (2010) 215–221
221
face [26]. That means that organics could also react with generated
chlorine in very small extent in view of the low content of Cl−
[3] N. Oturan, M.A. Oturan, Agron. Sustain. Dev. 25 (2005) 267.
[4] M. Diagne, N. Oturan, M.A. Oturan, Chemosphere 66 (2007) 841.
[5] I. Sirés, J.A. Garrido, R.M. Rodríguez, E. Brillas, N. Oturan, M.A. Oturan, Appl.
Catal. B: Environ. 72 (2007) 382.
[6] M.A. Oturan, M. Pimentel, N. Oturan, I. Sirés, Electrochim. Acta 54 (2008) 173.
[7] A. Özcan, Y. S¸ ahin, K. Savas¸ , M.A. Oturan, J. Hazard. Mater. 153 (2008) 718.
[8] S. Hammami, N. Bellakhal, N. Oturan, M.A. Oturan, M. Dachraoui, Chemosphere
73 (2008) 678.
[9] N. Oturan, M. Panizza, M.A. Oturan, J. Phys. Chem. A 113 (2009) 10988.
[10] I. Sirés, N. Oturan, M.A. Oturan, Water Res. 44 (2010) 3109.
[11] E. Brillas, M.A. Ban˜os, J.A. Garrido, Electrochim. Acta 48 (2003) 1697.
[12] E. Brillas, B. Boye, M.A. Ban˜os, J.C. Calpe, J.A. Garrido, Chemosphere 51 (2003)
227.
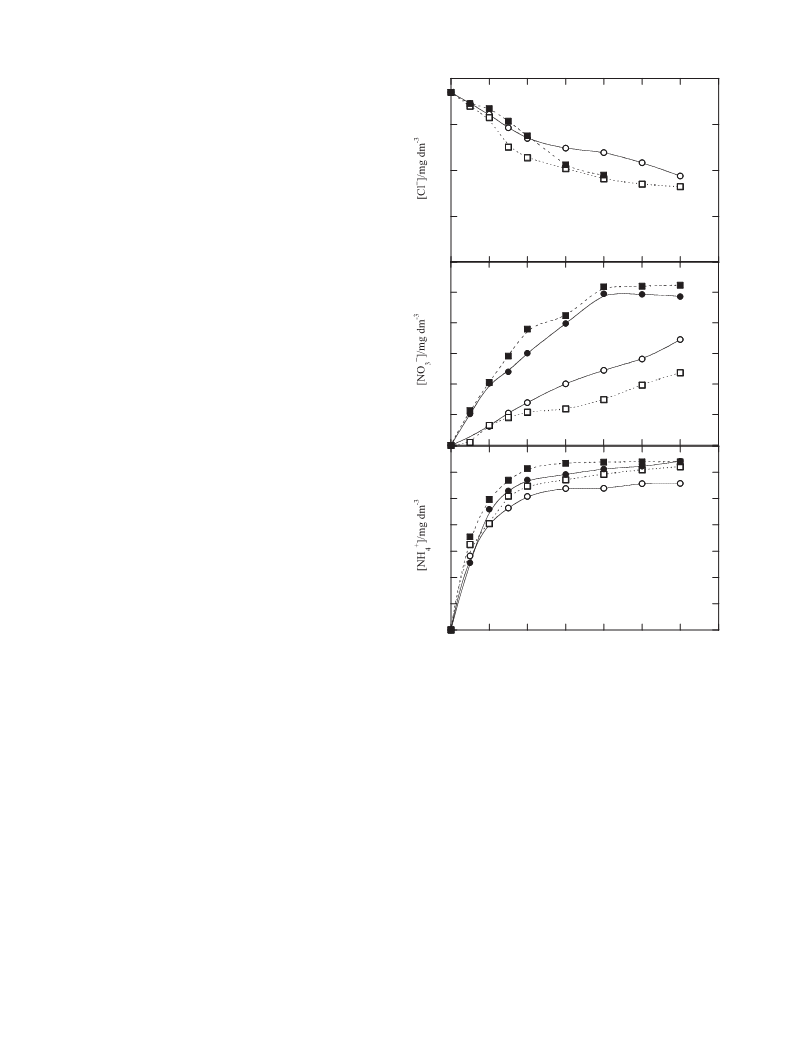
removed (10 mg dm−3 in 420 min). On the other hand, NO3 and
−
NH4 ions formed from the initial N of the drug (7.3 mg dm−3) are
+
gradually accumulated, as shown Fig. 6b and c, respectively. Steady
states of about 10 mg dm−3 of NO3− (31% of initial N) at 240 min and
6.4 mg dm−3 of NH4 (68% of initial N) at 120 min are obtained for
+
PEF in single and combined cells, indicating that all organic nitrogen
is mineralized with preponderance of NH4+. For the less potent EF
−
+
processes, NO3 concentration rises much more slowly and NH4
[13] I. Sirés, F. Centellas, J.A. Garrido, R.M. Rodríguez, C. Arias, P.L. Cabot, E. Brillas,
Appl. Catal. B: Environ. 72 (2007) 373.
[14] H. Wang, J. Wang, Appl. Catal. B: Environ. 77 (2007) 58.
[15] E. Guinea, C. Arias, P.L. Cabot, J.A. Garrido, R.M. Rodríguez, F. Centellas, E. Brillas,
Water Res. 42 (2008) 499.
[16] G.R. Agladze, G.S. Tsurtsumia, B.-I. Jung, J.-S. Kim, G. Gorelishvili, J. Appl. Elec-
trochem. 37 (2007) 985.
[17] M. Skoumal, C. Arias, P.L. Cabot, F. Centellas, J.A. Garrido, R.M. Rodríguez, E.
Brillas, Chemosphere 71 (2008) 1718.
ion follows the same tendency as for the PEF methods. After 360 min
of EF, 81% of the initial N is converted into inorganic ions in both
cells, suggesting that the remaining species in solution are com-
posed of a large proportion of N-derivatives, which mainly yield
NO3 when they are destroyed by UVA light in the homologous
PEF treatments.
−
[18] M. Panizza, G. Cerisola, Water Res. 43 (2009) 339.
[19] M. Skoumal, R.M. Rodríguez, P.L. Cabot, F. Centellas, J.A. Garrido, C. Arias, E.
Brillas, Electrochim. Acta 54 (2009) 2077.
4. Conclusions
It has been demonstrated that EF and PEF treatments with BDD
anode are able to mineralize naphthalene derivatives such as the
beta-blocker propranolol. The use of a combined BDD/ADE–Pt/CF
cell enhances the degradation processes compared with a sin-
gle BDD/ADE one because more oxidant •OH is formed from Fe2+
regeneration at the CF cathode. The PEF treatments lead to almost
overall mineralization due to the photolysis of Fe(III)–carboxylate
complexes by UVA light, which are hardly oxidized with hydroxyl
radicals in the less potent EF processes. The oxidation power of all
methods increases with increasing current density. The PEF process
is optimal with 0.5 mmol dm−3 Fe2+ at pH 3.0 and is more viable at
lower current density and higher drug content. Propranolol decay
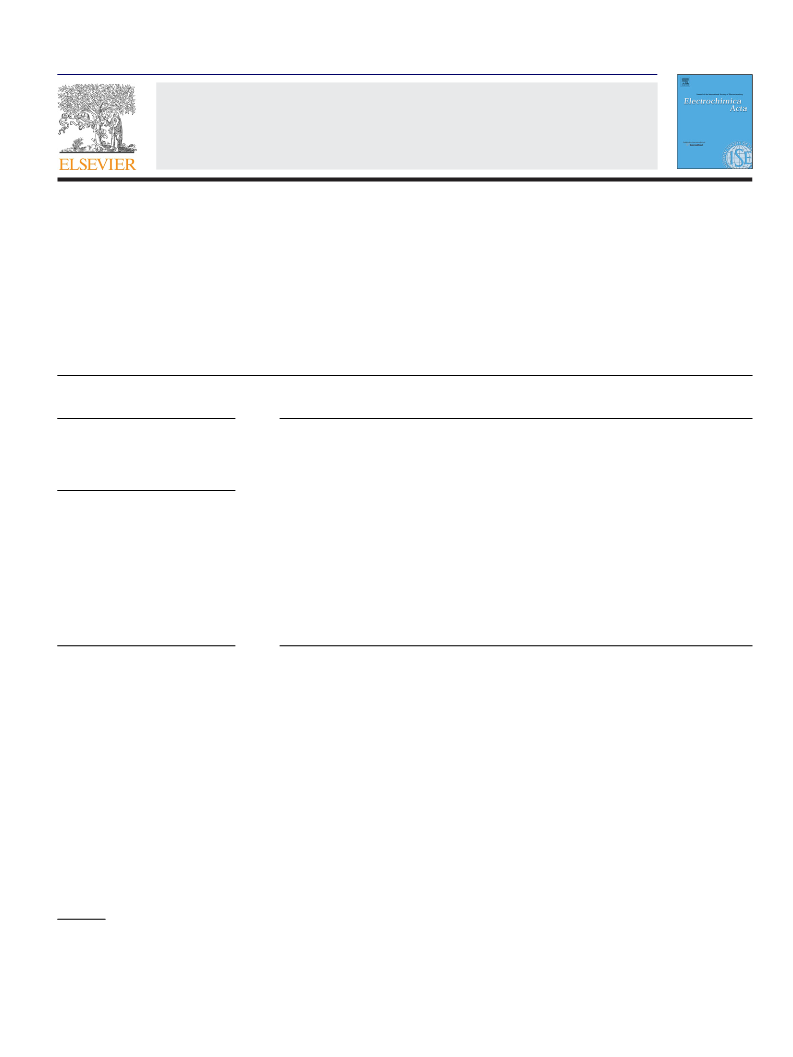
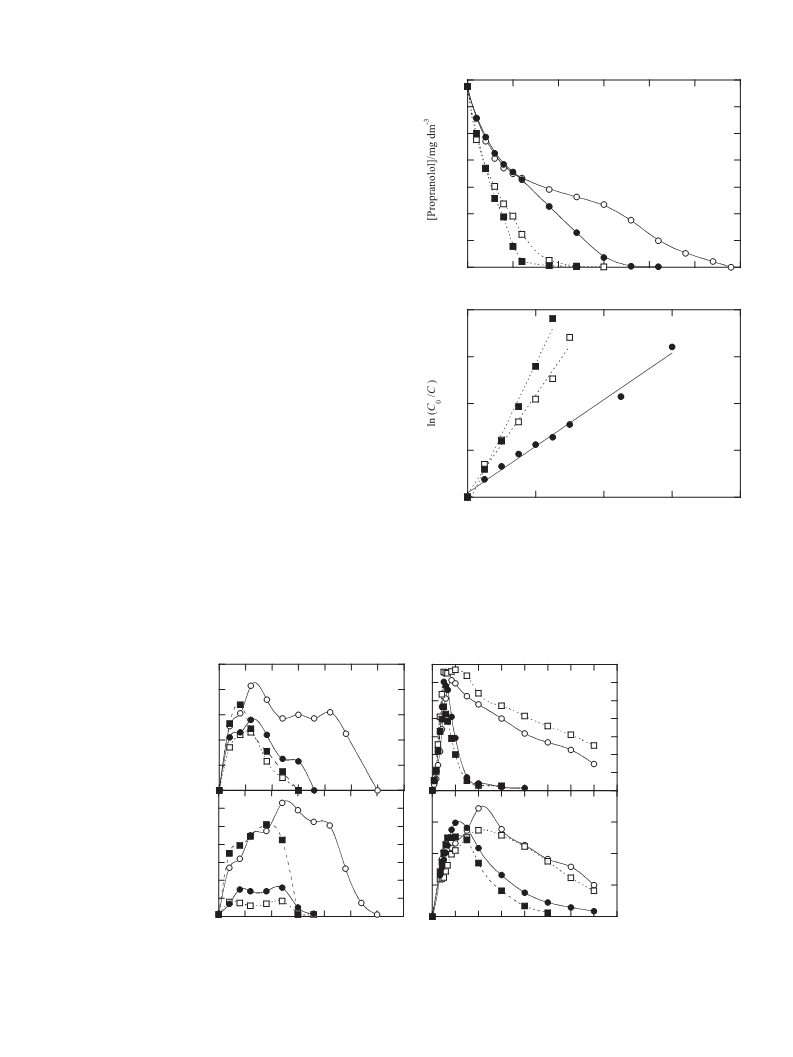
follows a pseudo first-order kinetics in most cases, mainly being
attacked by •OH. 1-Naphthol and phthalic acid are detected as
aromatic products while the drug persists and are also mainly
destroyed with •OH. Final Fe(III)–oxalate and Fe(III)–oxamate com-
plexes are slowly oxidized with BDD(•OH) in EF and more rapidly
photodecarboxylated by UVA light in PEF. The initial Cl− is sl−owly
[20] E. Isarain-Chávez, C. Arias, P.L. Cabot, F. Centellas, R.M. Rodríguez, J.A. Garrido,
E. Brillas, Appl. Catal. B: Environ. 96 (2010) 361.
[21] A. Da Pozzo, L. Di Palma, C. Merli, E. Petrucci, J. Appl. Electrochem. 35 (2005)
413.
[22] S. Yuan, M. Tian, Y. Cui, L. Lin, X. Lu, J. Hazard. Mater. B137 (2006) 573.
[23] C. Badellino, C.A. Rodrigues, R. Bertazzoli, J. Hazard. Mater. B137 (2006) 856.
[24] A. Wang, J. Qu, H. Liu, J. Ru, Appl. Catal. B: Environ. 84 (2008) 393.
[25] K. Cruz-González, O. Torres-López, A. García-León, J.L. Guzmán-Mar, L.H. Reyes,
A. Hernández-Ramírez, J.M. Peralta-Hernández, Chem. Eng. J. 160 (2010) 199.
[26] M. Panizza, G. Cerisola, Chem. Rev. 109 (2009) 6541.
[27] Ch. Comninellis, G. Chen (Eds.), Electrochemistry for the Environment, Springer,
New York, 2009.
[28] B. Boye, P.A. Michaud, B. Marselli, M.M. Dieng, E. Brillas, Ch. Comninellis, New
Diamond Front. Carbon Technol. 12 (2002) 63.
[29] C. Flox, J.A. Garrido, R.M. Rodríguez, F. Centellas, P.L. Cabot, C. Arias, E. Brillas,
Electrochim. Acta 50 (2005) 3685.
[30] R.G. Zepp, B.C. Faust, J. Hoigné, Environ. Sci. Technol. 26 (1992) 313.
[31] Y. Zuo, J. Hoigné, Environ. Sci. Technol. 26 (1992) 1014.
[32] R. Andreozzi, R. Marotta, P. Nicklas, Chemosphere 50 (2003) 1319.
[33] A.I. Balciog˘lu, M. Ötker, Chemosphere 50 (2003) 85.
[34] L.J. Fono, D.L. Sedlak, Environ. Sci. Technol. 39 (2005) 9244.
[35] J.P. Bound, N. Voulvoulis, Water Res. 40 (2006) 2885.
[36] M. Maurer, B.I. Escher, P. Richle, C. Schaffner, A.C. Alder, Water Res. 41 (2007)
1614.
oxidized by BDD(•OH). NH4 and in smaller proportion NO3 are
+
[37] K. Kümmerer, Chemosphere 75 (2009) 417.
lost during all degradations.
[38] M. Ramil, T. El Aref, G. Fink, M. Scheurer, T. Ternes, Environ. Sci. Technol. 44
(2010) 962.
[39] M. Cleuvers, Chemosphere 59 (2005) 199.
Acknowledgements
[40] G. Nałecz-Jawecki, G. Persoone, Environ. Sci. Pollut. Res. 13 (2006) 22.
[41] S.F. Owen, E. Giltrow, D.B. Huggett, T.H. Hutchinson, J. Saye, M.J. Winter, J.P.
Sumpter, Aquat. Toxicol. 82 (2007) 145.
[42] J. Benner, E. Salhi, T. Ternes, U. Gunten, Water Res. 42 (2008) 3003.
[43] R. Rosal, A. Rodríguez, J.A. Perdigón-Melón, M. Mezcua, M.D. Hernando, P. Letón,
E. García-Calvo, A. Agüera, A.R. Fernández-Alba, Water Res. 42 (2008) 3719.
[44] W. Song, W.J. Cooper, S.P. Mezyk, J. Greaves, B.M. Peake, Environ. Sci. Technol.
42 (2008) 1256.
[45] I. Kim, N. Yamashita, H. Tanaka, Chemosphere 77 (2009) 518.
[46] E. Marco-Urrea, J. Radjenovic´, G. Caminal, M. Petrovic´, T. Vicent, D. Barceló,
Water Res. 44 (2010) 521.
[47] P.M. Lorz, F.K. Towae, W. Enke, R. Jäckh, N. Bhargava, W. Hillesheim, Ullmann’s
Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2007.
Financial support from MEC (Ministerio de Educación y Ciencia,
Spain) under project CTQ 2007-60708/BQU, cofinanced with Feder
funds, is acknowledged. The authors are also indebted to CONACYT
(Consejo Nacional de Ciencia y Tecnología, Mexico) by the grant
given to E. Isarain-Chávez.
References
[1] Y. Sun, J.J. Pignatello, Environ. Sci. Technol. 27 (1993) 304.
[2] E. Brillas, I. Sirés, M.A. Oturan, Chem. Rev. 109 (2009) 6570.

 Isarain-Chávez, Eloy
Isarain-Chávez, Eloy
 Rodríguez, Rosa María
Rodríguez, Rosa María
 Garrido, José Antonio
Garrido, José Antonio
 Arias, Conchita
Arias, Conchita
 Centellas, Francesc
Centellas, Francesc
 Cabot, Pere Lluís
Cabot, Pere Lluís
 Brillas, Enric
Brillas, Enric