DOI: 10.1002/cssc.201701841
Communications
Methylation of Polyols with Trimethylphosphate in the
Presence of a Lewis or Brønsted Acid Catalyst
Marie-Christine Duclos, Aurꢀlien Herbinski, Anne-Sophie Mora, Estelle Mꢀtay,* and
[a]
Marc Lemaire*
The alkylation of alcohols and polyols has been investigated
with alkylphosphates in the presence of a Lewis or Brønsted
acid catalyst. The permethylation of polyols was developed
under solvent-free conditions at 1008C with either iron triflate
or Aquivion PW98, affording the isolated products in yields be-
tween 52 and 95%. The methodology was also adjusted to
carry out peralkylation with longer alkyl chains.
Among possible methylating agents, trimethylphosphate
(TMP) has mainly been reported as an alkylating agent for
[15]
amines, whereas its use with alcohols is unusual. Methylation
and alkylation (using trialkylphosphate) of phenolic alcohols
[16–18]
were realized by using a Lewis acid or a base, respectively.
To our knowledge, only two reports have described the meth-
ylation of aliphatic alcohols with trimethylphosphate. In 1944,
Toy described the methylation of alcohols in moderate yield by
[19]
heating both reagents at reflux. Unfortunately, as mentioned
by the author, this procedure is not compatible with alcohols
with a boiling point lower than 1608C. Later, similar conditions
were developed by Van Dyke Tiers, with polyphosphoric acid
Interest in oxygenated bio-based molecules has increased with
[1,2]
the research into alternatives to petroleum-based products.
[
3]
[4]
Even in light of research into new solvents, hydrotropes,
[
5]
[6]
[20]
surfactants, or additives for gasolines, the transformation of
glycerol and other polyols remains in strong demand. Ether de-
rivatives are generally preferred to esters because of their sta-
bility, notably in the presence of water. In addition, a recent
study dealing with the toxicity of glycerol ethers has further in-
creased interest in the selective synthesis of such com-
as catalyst at a temperature of almost 1808C. In this rstudy,
the author noted that a distillation of the crude at atmospheric
pressure can be dangerous and an explosion can occur. In ad-
dition, experiments on rats have demonstrated the low toxicity
[21]
of trimethylphosphate. Herein we propose a simple proce-
dure to carry out the methylation of alcohols and polyols at
1008C in the presence of a Lewis acid or a Brønsted acid cata-
[
7,8]
pounds.
Although the alkylation of glycerol has been stud-
[
6]
[22]
ied under a wide range of reported conditions, the peralkyla-
tion of polyols has rarely been described and, in particular,
their methylation remains problematic.
lyst.
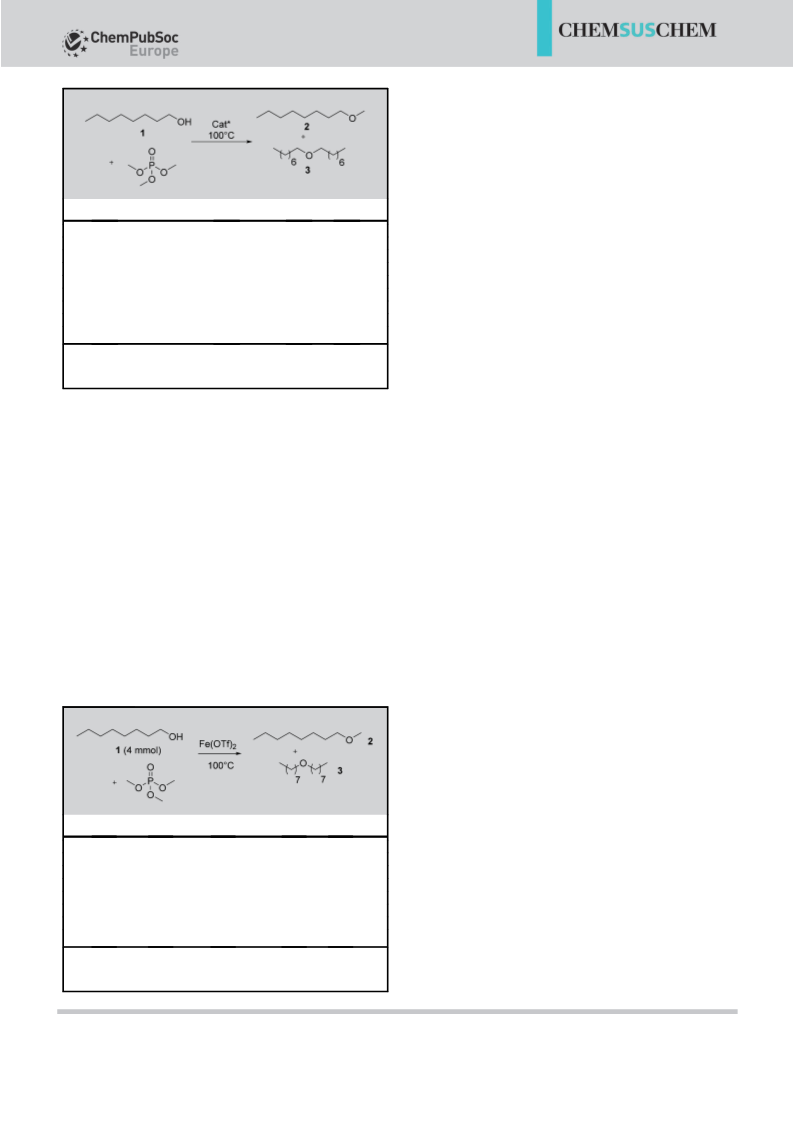
To determine the best conditions, octanol was retained as
the starting material. In agreement with previous reports, of
trimethylphosphate exhibited a lack of reactivity was also no-
ticed without catalyst at temperatures lower than 1608C. To in-
crease the reactivity of the phosphate, the influence of differ-
ent catalysts on the formation of methyl octyl ether was evalu-
ated. We first explored the influence of the addition of a
Brønsted acid to the reaction mixture. At 1008C in the pres-
ence of sulfuric acid (20 mol%), the octanol was almost quanti-
tatively converted (94%) and methyl octyl ether was obtained
in 70% yield (Table 1, entry 1). The other product was identi-
fied as dioctyl ether (24%). The use of a stronger acid—triflic
acid (20 mol%)—was also evaluated. In this case, the conver-
sion was lower but the selectivity towards the methylated
product was higher (Table 1, entry 2). Next, a range of Lewis
acids (all triflate) were also tested under similar conditions. For
all triflates tested, good conversions were obtained but the
best results were observed with bismuth and iron triflates
(Table 1, entries 4, 7, and 8).
Methylation is usually performed under Williamson condi-
[
9]
tions, utilizing toxic reagents such as iodomethane or dime-
[
6]
thylsulfate in nonprotic dipolar solvents. The use of less toxic
[
10]
[11]
reagents, such as dimethylcarbonate
or dimethylsulfite,
has also been explored. In these cases, the required tempera-
tures can limit the application of the conditions to specific sub-
strates. More precisely, at high temperatures (above 1608C) the
alkylation of glycerol is in competition with the formation of
[
12]
acrolein. An alternative was recently proposed as 1,2-dime-
thoxyethane was synthesized in a continuous flow reactor,
[
13]
with methanol and microporous zeolite as catalyst. Among
the targeted compounds, 1,2,3-trimethoxyproprane was pre-
pared from glycerol with dimethylsulfate under solvent-free
conditions. This development allowed the preparation of this
product to evaluate its toxicity and its applicability as a sol-
[
14]
vent. The results of these studies prompted us to look for an
alternative pathway for the methylation of alcohols and poly-
ols.
With lanthum, cerium, and yttrium triflates, the conversions
of octanol were lower (Table 1, entries 3, 5, 6), and the only
tested sulfate (FeSO ) gave a very low conversion (Table 1,
[a] M.-C. Duclos, Dr. A. Herbinski, A.-S. Mora, Dr. E. Mꢀtay, Prof. M. Lemaire
4
University Claude Bernard Lyon 1, CNRS, INSA-Lyon, CPE-Lyon
Institut de Chimie et Biochimie Molꢀculaires et Supramolꢀculaires (ICBMS)
UMR 5246, Equipe CAtalyse, Synthꢁse et ENvironnement (CASYEN)
entry 9). Reactions were performed in a pressure tube and
overpressure was never detected, indicating no formation of
volatile compounds such as dimethyl ether. To select the best
Lewis acid catalyst, bismuth and iron triflates were tested in
parallel with loadings of 2 mol%. The best result was obtained
4
3, bd du 11 Novembre 1918, 69622 Villeurbanne cedex (France)
E-mail: estelle.metay@univ-lyon1.fr
ChemSusChem 2018, 11, 1 – 6
1
ꢁ 2018 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
&
These are not the final page numbers! ÞÞ

 Duclos, Marie-Christine
Duclos, Marie-Christine
 Herbinski, Aurélien
Herbinski, Aurélien
 Mora, Anne-Sophie
Mora, Anne-Sophie
 Métay, Estelle
Métay, Estelle
 Lemaire, Marc
Lemaire, Marc