Organic & Biomolecular Chemistry
Communication
species, whereas II could be observed only in a tiny amount. In
a low dielectric constant solvent like chloroform, only I is
present.18,20 Meanwhile, Susz discovered that II was stable and
was the major species in solids at low temperature when AlCl3
reacted with acyl chloride.19 The existence of the two species
was well confirmed, while their reactivities towards ArH were
not clear. Hence, it remains problematic to establish which
one is the reactive electrophile in the AlCl3 promoted Friedel–
Crafts acylation reaction.
As for the true reactive species in Friedel–Crafts acylation,
they were mainly identified relying on a special Lewis acid, acy-
lation reagents and solvents.16,23,24 For example, from the flu-
oride method or the silver salt method, an acylium ion crystal
has been prepared and was established as the reactive electro-
phile in SO2 and HF solutions by Olah.25 Cook proposed that
(PhCO)2O→(BF3)3 was the reactive electrophile in BF3 induced
Friedel–Crafts acylation with benzoic anhydride.26,27 In AlCl3-
catalyzed Friedel–Crafts benzoylation of benzene, Brown pro-
posed that both the acylium ion and the donor–acceptor
complex I could be the reactive electrophile with benzoyl chlor-
ide as the solvent.28 F. Effenberger supported an acylium ion
as the reactive electrophile between aromatic compounds and
aroyl triflates without the Friedel–Crafts catalyst.29 However,
for AlCl3 promoted Friedel–Crafts acylation reactions, it
remains a challenge to confirm the true active species,
especially under live reaction conditions. Herein, we commu-
nicate our results in “capturing” the reactive electrophile
through a kinetic study from a live AlCl3 promoted acylation.
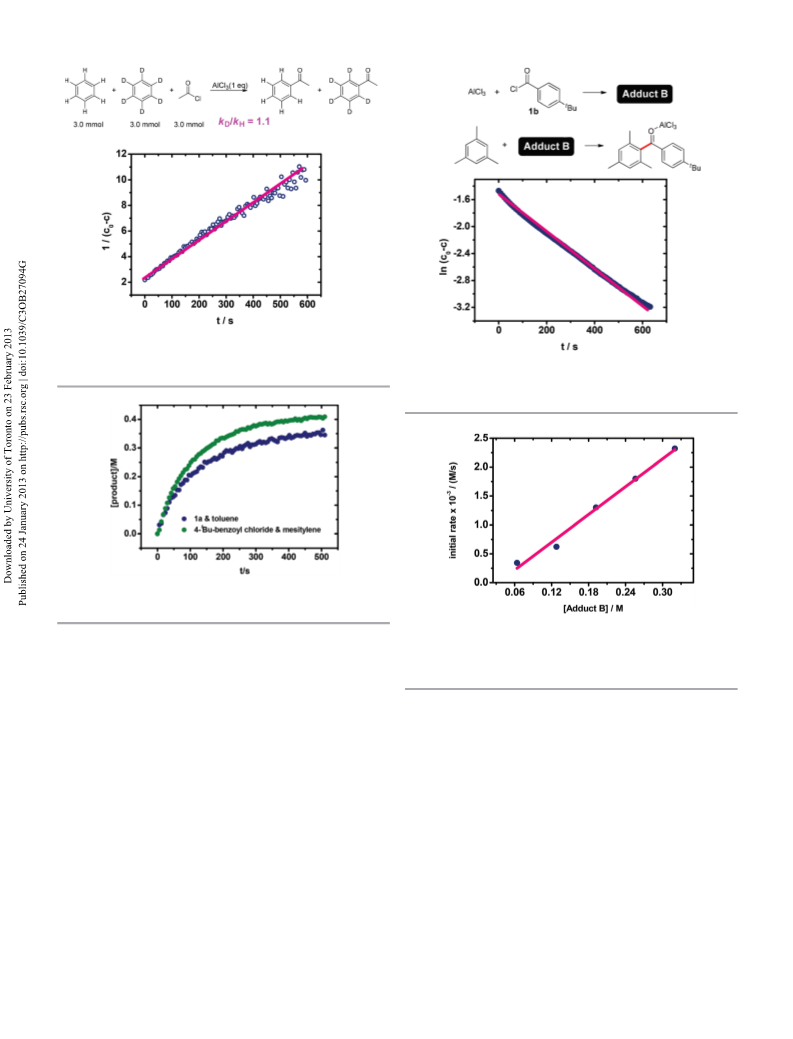
Initially, AlCl3 promoted Friedel–Crafts acylation between
1a and toluene was monitored by in situ IR shown in Fig. 1.
When 1a (band A at 1806 cm−1) reacted with AlCl3, adduct A
was afforded. The band B at 1653 cm−1 accumulated and was
assigned as the donor–acceptor complex I-A according to litera-
ture reported methods.13,17–21,27,30–32 When toluene was
added, as shown in Fig. 1, the bands at 1583 cm−1 (C),
1548 cm−1 (D), which were assigned as the AlCl3 adduct of
p-methylacetophenone, appeared proportionally (Fig. S1B and
S2†). In the meantime, I-A decreased quickly. It seemed that
I-A was the true reactive species. However, when the region at
2250–2350 cm−1 was focused, the band E at 2308 cm−1, which
was a fairly tiny absorption and almost invisible compared to
I-A, also disappeared immediately (Fig. 1(B)). If this band
could be assigned to the acylium ion II-A, it also might be the
active species.13–15,25,31,33 Therefore, how to distinguish these
two high reactive species and “capture” the true reactive elec-
trophile deserved to be further discussed.
Fig. 1 3D-profile of the reaction between AlCl3 (0.5 M), 1a (0.5 M) and
toluene (0.5 M) in 1,2-dichloroethane (4 mL) at −10 °C through in situ IR.
Approximate Model, the rate law could be more complicated. If
the electrophilic reaction with ArH is the rate-limiting step
(Scheme 1, path 2 eqn (4)), which means k′−1 ≫ k′2[ArH], the
rate law could be simplified as shown in Scheme 1 (1) and the
reaction rate is dependent on [ArH] and [I]. If the transfor-
mation from I to II is the rate-limiting step (Scheme 1, path 2
eqn (3)), which means k′−1 ≪ k′2[ArH], the reaction rate will be
independent of [ArH] and first-order kinetic on [I]. In other
words, kinetic differences exist in the two pathways. By
choosing proper substrates, the reaction rate law of eqn (3)
and (4) could be switched from one kinetic behavior to the
other if the reaction follows pathway II.
On the basis of the above understanding, further experi-
ments were carried out in this AlCl3 promoted acylation
between 1a and toluene. As shown in Fig. 2, plotting 1/(c0 − c)
vs. t resulted in a line, suggesting that this reaction exhibits a
second-order kinetic behavior. The rate law was: rate = k[A]-
[toluene] (Fig. S3–S7†). As shown in Fig. 1, the major species in
A was I-A, while II-A was almost invisible. Namely, [A] was
approximately equal to [I-A]. Therefore, rate ≈ k[I-A][toluene].
The kinetic result could be fitted well with that of the two
pathways. In this case, the two pathways could not be differen-
tiated from each other in this AlCl3 promoted acylation
between acetyl chloride and toluene.
In both pathways, deprotonation is the last step (eqn (2)
and (5)). KIE experiment (eqn (6)) was performed and kD/kH
=
1.1, revealing that the deprotonation was not the rate-limiting
step. Namely, the rate-limiting step should be assigned to the
process prior to deprotonation. Regarding the two pathways, if
the reaction follows path I and I is the reactive species to react
with ArH, the rate law would be: rate = k[I][ArH] (shown in
Scheme 1), and the overall reaction will always exhibit a first-
order kinetic behavior on [ArH] and first-order on [I]. While if
the II is the reactive intermediate, according to the Steady-State
According to the mechanistic discussion, the key to the
kinetic differences in the two paths is that the rate-limiting
step could be varied when employing different substrates.
This journal is © The Royal Society of Chemistry 2013
Org. Biomol. Chem., 2013, 11, 1810–1814 | 1811

 Huang, Zhiliang
Huang, Zhiliang
 Jin, Liqun
Jin, Liqun
 Han, Heyou
Han, Heyou
 Lei, Aiwen
Lei, Aiwen