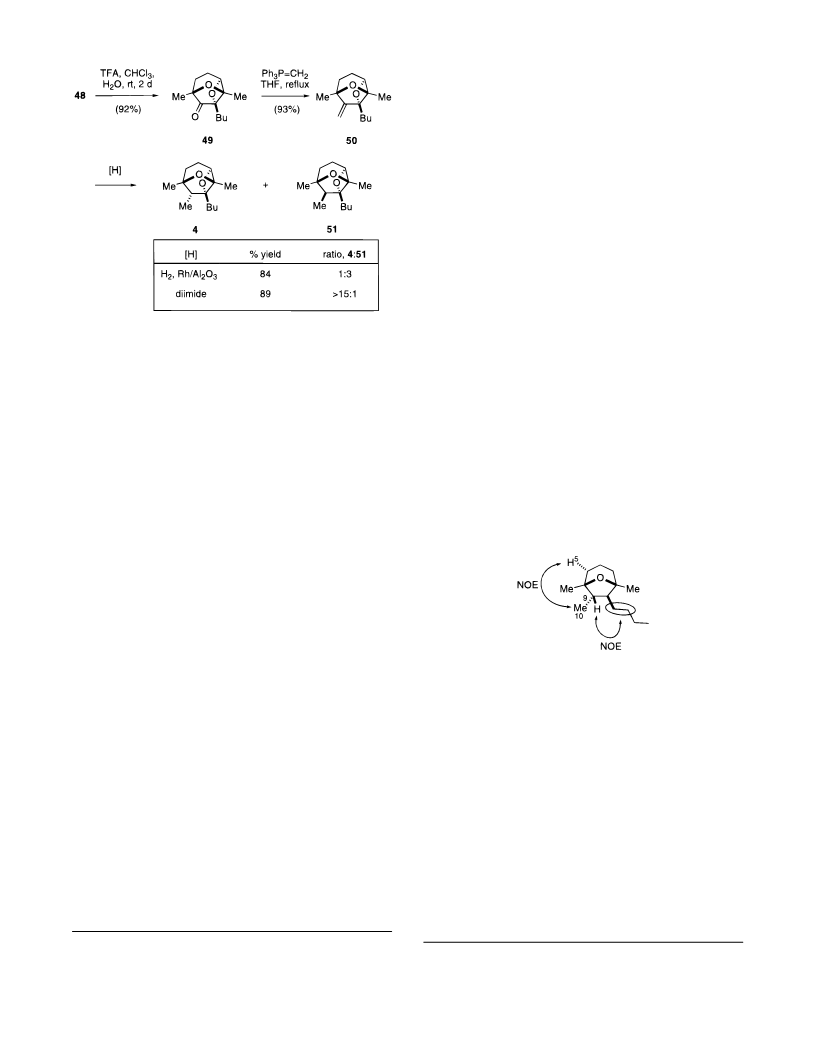
Synthesis of a 2,7-Dioxatricyclo[4.2.1.03,8]nonane
J . Org. Chem., Vol. 61, No. 26, 1996 9143
added via cannula. Immediately following the addition, dry
oxygen gas was bubbled into the solution for 30 min. The
reaction was quenched with 0.5 mL of 1 M stannous chloride
in 2 M HCL solution and stirred for 30 min at 0 °C. Then the
reaction mixture was diluted with ethyl acetate and washed
with water, 1 M NaOH, and brine. The organics were dried
and concentrated in vacuo. Flash chromatography of the
residue (50% ethyl acetate in hexanes) provided diol 28 (15
Cl
2
, and the combined organics were dried and concentrated
in vacuo. The residue was then dissolved in methanol (2.5
mL), and the resulting solution was cooled to 0 °C with
stirring. Sodium borohydride (27 mg, 0.72 mmol) was added,
and the reaction mixture was stirred at 0 °C for 10 min. The
2
reaction was quenched with H O and diluted with ethyl
acetate. The resulting biphasic mixture was stirred at room
temperature for 15 min, and then the layers were separated.
The aqueous layer was extracted twice with ethyl acetate, and
the combined organics were dried and concentrated in vacuo.
Flash chromatography of the residue provided the hydroxy
mesylate 32 (85 mg, 68%) as a white solid; mp 104-105 °C.
-
1
1
mg, 56%) as a white solid. IR: 3406, 2212, 1633 cm
.
H
NMR (400 MHz): δ 1.30 (s, 3), 1.70-1.76 (m, 2), 1.91-1.99
(
m, 2), 2.19 (s, 3), 2.49 (d, 1, J ) 15.6), 2.57 (d, 1, J ) 15.6).
1
3
C NMR: δ 20.3, 30.2, 31.6, 39.3, 42.5, 69.3, 72.6, 104.6, 109.4,
-
1
1
1
61.9. Anal. Calcd for C10
Found: C, 66.08; H, 8.34, N, 7.58.
-Cya n o-1,5-d im et h yl-8-oxa b icyclo[3.2.1]oct a n -4-exo-
H
15NO
2
: C, 66.27; H, 8.34, N, 7.73.
IR: 3431, 2360 cm
.
H NMR (500 MHz): δ 1.30 (s, 3), 1.34
(s, 3), 1.43 (ddd, 1, J ) 13.5, 6.0, 1.2), 1.76 (dd, 1, J ) 13.4,
4.1), 1.85 (ddd, app td, 1, J ) 13.5, 13.5, 5.4), 2.08 (dddd, app
ddt, 1, J ) 15.7, 5.4, 1.6, 1.6), 2.17 (ddd, 1, J ) 13.4, 10.9,
1.1), 2.21 (br d, 1, J ) 5.0), 2.41 (dddd, 1, J ) 15.7, 13.6, 6.1,
3.8), 3.06 (s, 3), 4.23 (ddd, app dt, 1, J ) 10.9, 4.2, 4.2), 4.77
(dd, 1, J ) 3.6, 1.6). C NMR: δ 21.6, 25.2, 26.7, 31.9, 38.7,
43.9, 77.5, 78.7, 79.7, 82.1. Anal. Calcd for C10
47.99; H, 7.25. Found: C, 48.31; H, 7.34.
6
yl ter t-Bu tyld im eth ylsilyl Eth er (29). To a solution of
alcohol 27 (275 mg, 1.5 mmol) in DMF (3 mL) was added
imidazole (410 mg, 6.0 mmol) and tert-butyldimethylsilyl
chloride (450 mg, 3.0 mmol). The resulting solution was
stirred at room temperature for 48 h. The reaction was
1
3
18 5
H O S: C,
4
quenched with aqueous saturated NH Cl and diluted with
ethyl ether. The layers were separated, and the organics were
dried and concentrated in vacuo. Flash chromatography of
the residue (7% ethyl acetate in hexanes) provided silyl ether
6-Bu t yl-1,5-d im et h yl-8-oxa bicyclo[3.2.1]oct a n e-4-exo-
6-en d o-d iol (33). To a solution of butyllithium in hexanes
(0.6 mL, 1.8 mmol) in THF (4 mL) at -78 °C was added
dropwise a solution of 30 (100 mg, 0.35 mmol) in THF (0.5
mL). After stirring for 20 min, the dry ice bath was removed,
-
1
1
2
9 (400 mg, 90%) as a pale yellow oil. IR: 2239 cm
.
H
NMR (500 MHz): δ 0.06 (s, 3), 0.07 (s, 3), 0.91 (s, 9), 1.26 (ddd,
1
1
2
, J ) 13.0, 5.7, 1.4), 1.35 (s, 3), 1.47 (s, 3), 1.64-1.85 (m, 3),
.96 (ddd, 1, J ) 13.0, 5.8, 2.0), 2.31 (dd, 1, J ) 13.0, 9.7),
4
and the reaction was quenched with aqueous saturated NH -
Cl. The reaction mixture was extracted twice with ethyl
acetate, and the combined organics were dried and concen-
trated in vacuo. The residue was then dissolved in 3 mL of
1
3
.83 (dd, 1, J ) 9.7, 5.8), 3.30 (app t, 1, J ) 2.5). C NMR:
δ -4.9, -4.7, 18.0, 21.0, 25.7, 26.1, 26.4, 31.0, 36.7, 41.4, 69.5,
8
9
0.5, 83.9, 120.8. Anal. Calcd for C16
.89; N, 4.74. Found: C, 64.88; H, 10.14; N, 4.80.
-exo-[(ter t-Bu t yld im et h ylsilyl)oxy]-1,5-d im et h yl-8-
H
29NO
2
Si: C, 65.03; H,
2% HF/CH
3
CN, and the resulting solution was stirred for 1 h.
The reaction was quenched with aqueous saturated NaHCO
3
4
and diluted with ethyl acetate. The layers were separated,
and the aqueous layer was extracted twice with ethyl acetate.
The combined organics were dried and concentrated in vacuo.
Flash chromatography of the residue (40% ethyl acetate in
hexanes) provided the diol 33 (67 mg, 84%) as a white solid;
oxa bicyclo[3.2.1]octa n -6-on e (30). To a solution of diiso-
propylethylamine (0.16 mL, 1.15 mmol) in THF (5 mL) at -78
°
C was added butyllithium in hexanes (0.63 mL, 1.15 mmol).
The solution was warmed to 0 °C and stirred for 30 min. Then
the solution was recooled to -78 °C, and a solution of 29 (295
mg, 1.00 mmol) in THF (0.5 mL) was added via cannula.
Immediately following the addition, dry oxygen gas was
bubbled into the solution for 30 min. The reaction was
quenched with 2 mL of 1 M stannous chloride in 2 M HCL
solution and stirred for 30 min at 0 °C. Then the reaction
mixture was diluted with ethyl acetate and washed with water,
-
1
1
mp 85-86 °C. IR: 3403 cm
. H NMR (400 MHz): δ 0.94 (t,
3, J ) 6.9), 1.19 (s, 3), 1.24 (s, 3), 1.33-1.40 (m, 5), 1.48-1.62
(m, 2), 1.67-1.79 (m, 3), 1.85 (d, 1, J ) 13.5), 1.89 (d, 1, J )
13.5), 2.22 (d, 1, J ) 11.0), 2.39 (dddd, 1, J ) 15.7, 11.4, 8.0,
1
3
3.4), 3.56 (br d, 1, J ) 10.1). C NMR: δ 14.1, 16.9, 23.2,
25.2, 26.8, 27.2, 31.9, 40.1, 48.0, 68.0, 79.1, 82.3, 86.0. Anal.
Calcd for C13
10.58.
24 3
H O : C, 68.39; H, 10.60. Found: C, 68.42; H,
1
M NaOH, and brine. The organics were dried and concen-
trated in vacuo. Flash chromatography of the residue (10%
ethyl acetate in hexanes) provided ketone 30 (175 mg, 62%)
6-Bu tyl-4-exo-[(m eth a n esu lfon yl)oxy]-1,5-d im eth yl-8-
oxa bicyclo[3.2.1]octa n -6-en d o-ol (34). To a solution of diol
33 (14 mg, 0.06 mmol) in CH Cl (0.3 mL) at 0 °C was added
2 2
triethylamine (17 µL, 0.12 mmol), followed by methanesulfonyl
chloride (7 mg, 0.06 mmol). After stirring for 30 min, the
-1
1
as a colorless oil. IR: 1754 cm
(
(
2
-
8
.
H NMR (300 MHz): δ 0.06
s, 3), 0.07 (s, 3), 0.92 (s, 9), 1.20 (s, 3), 1.34-1.41 (m, 1), 1.47
s, 3), 1.66-1.72 (m, 2), 1.95-2.05 (m, 1), 2.30 (d, 1, J ) 17.9),
1
3
.41 (d, 1, J ) 17.9), 3.45 (app t, 1, J ) 2.7). C NMR: δ
reaction was quenched with H
rated. The aqueous layer was extracted once with CH
2
O, and the layers were sepa-
Cl , and
4.7, -4.6, 17.3, 18.2, 25.8, 26.7, 27.8, 30.4, 47.0, 67.1, 77.3,
2
2
5.9, 217.8. Anal. Calcd for C15 Si: C, 63.33; H, 9.92.
H
28
O
3
the combined organics were dried and concentrated in vacuo.
Flash chromatography of the residue (30% ethyl acetate in
hexanes) provided hydroxy mesylate 34 (17 mg, 93%) as a
Found: C, 62.96; H, 9.99.
4
-exo-Hyd r oxy-1,5-d im eth yl-8-oxa bicyclo[3.2.1]octa n -
-on e (31). A solution of 30 (65 mg, 0.23 mmol) in 2% HF/
CH CN (2.5 mL) was stirred at room temperature for 1 h. Then
the reaction was quenched with aqueous saturated NaHCO
-
1
1
6
white solid; mp 118 °C. IR: 3517 cm
.
H NMR (400 MHz):
3
δ 0.95 (t, 3, J ) 6.9), 1.21 (s, 3), 1.27 (s, 3), 1.38-1.43 (m, 5),
1.49-1.58 (m, 3), 1.81-1.93 (m, 3), 2.09 (dddd, app ddt, 1, J
) 15.7, 5.4, 2.1, 2.1), 2.53 (dddd, 1, J ) 15.7, 13.7, 6.2, 3.7),
3
and diluted with ethyl acetate. The layers were separated,
and the aqueous layer was extracted twice with ethyl acetate.
The combined organics were dried and concentrated in vacuo.
Flash chromatography of the residue (50% ethyl acetate in
hexanes) provided the hydroxy ketone 31 (40 mg, 100%) as a
1
3
3.06 (s, 3), 4.81 (dd, 1, J ) 3.4, 2.1). C NMR: δ 14.1, 17.2,
23.2, 25.1, 25.6, 27.1, 31.7, 38.8, 39.8, 47.9, 78.6, 78.7, 82.6,
84.7. Anal. Calcd for C14
C, 54.88; H, 8.89.
26 5
H O S: C, 54.88; H, 8.55. Found:
-
1
1
1-Bu t yl-6,8-d im e t h yl-2,7-d ioxa t r icyclo[4.2.1.03,8]n o-
n a n e (3). To a solution of hydroxy mesylate 34 (35 mg, 0.11
mmol) in THF (1.1 mL) was added 60% NaH in oil (13.7 mg,
0.34 mmol), and the resulting suspension was heated to reflux.
After stirring for 2 h, the reaction was cooled to 0 °C and was
quenched by the dropwise addition of aqueous saturated
clear oil. IR: 3462, 1754 cm
(
1
1
2
.
H NMR (400 MHz): δ 1.26
s, 3), 1.44-1.49 (m, 4), 1.71 (dddd, 1, J ) 15.1, 13.4, 5.7, 3.4),
.79-1.94 (m, 2), 2.33, (dd, 1, J ) 17.9, 1.1), 2.43 (d, 1, J )
13
7.9), 2.50 (br d, 1, J ) 9.1), 3.43 (br s, 1). C NMR: δ 16.8,
6.5, 26.8, 30.4, 46.7, 66.9, 78.2, 85.5, 216.2. Anal. Calcd for
C
9
H
4
14
O
3
: C, 63.51; H, 8.29. Found: C, 63.75; H, 8.52.
-exo-[(Meth an esu lfon yl)oxy]-1,5-dim eth yl-8-oxabicyclo-
3.2.1]octa n -6-en d o-ol (32). To a solution of alcohol 31 (85
mg, 0.50 mmol) in CH Cl (2.5 mL) at 0 °C was added
3
NaHCO . The mixture was then extracted with ethyl ether,
and the organics were dried and concentrated in vacuo. Flash
[
2
2
chromatography of the residue (5% ethyl acetate in hexanes)
1
triethylamine (0.11 mL, 0.77 mmol), followed by methane-
provided the oxetane 3 (18 mg, 77%) as a colorless liquid. H
sulfonyl chloride (59 µL, 0.77 mmol). After stirring for 30 min,
NMR (400 MHz): δ 0.91 (t, 3, J ) 7.2), 1.19-1.27 (m, 1), 1.31-
1.37 (m, 5), 1.39-1.46 (m, 4), 1.47-1.54 (m, 2), 1.64-1.72 (m,
1), 1.79-1.93 (m, 4), 2.00 (d, 1, J ) 13.0), 4.39 (app t, 1, J )
2
the reaction was quenched with H O, and the layers were
2
separated. The aqueous layer was extracted once with CH -

 Marshall
Marshall
 Mapp
Mapp
 Heathcock
Heathcock