ACS Catalysis
Page 4 of 6
group (entries 2 and 3), methoxy (entry 4), bromo (entry 5),
X-ray crystallographic data for 8 (CIF)
1
2
3
4
5
6
7
8
9
1
1
1
1
1
1
1
1
1
1
2
2
2
2
2
2
2
2
2
2
3
3
3
3
3
3
3
3
3
3
4
4
4
4
4
4
4
4
4
4
5
5
5
5
5
5
5
5
5
5
6
and chloro (entry 6) substituents were tolerated in this catalyt-
ic system. The reaction of 2-naphthalenemethanol gave 2-
naphthoic acid in high yield (entry 7). In the case that the sol-
ubility of the substrates in water was extremely low, addition
of 1,4-dioxane as a co-solvent improved the yields of carbox-
ylic acids (entries 5-7).
AUTHOR INFORMATION
Corresponding Author
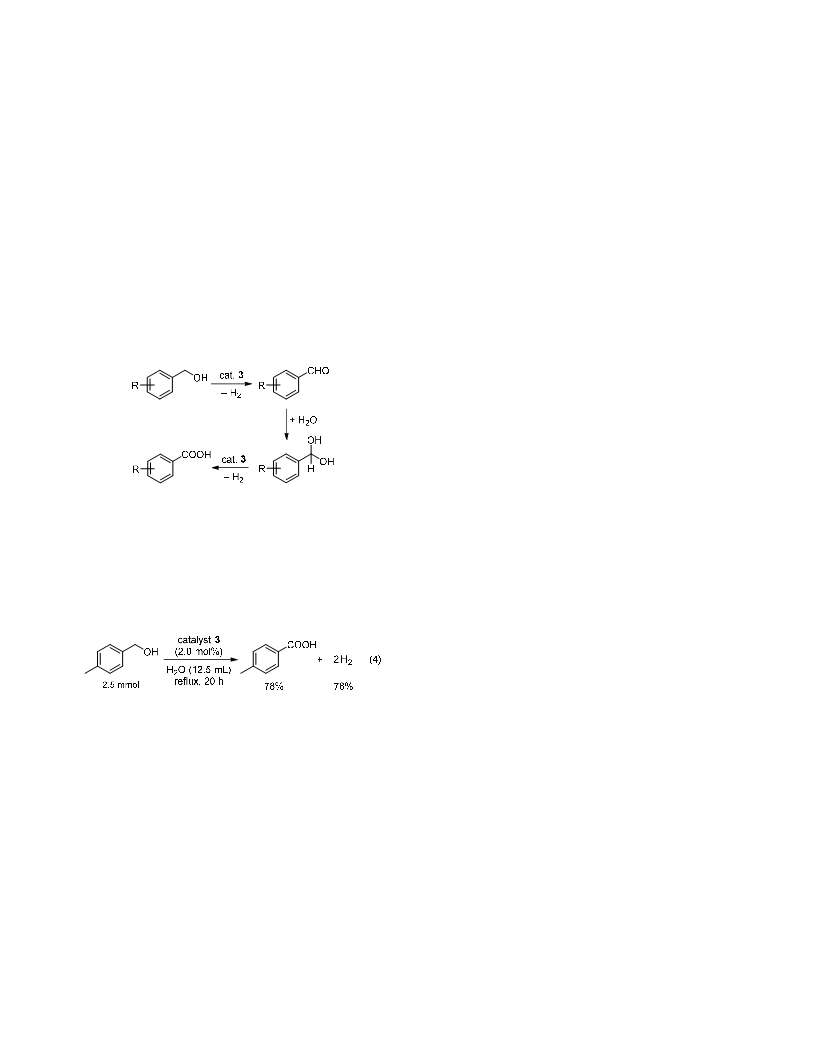
A possible reaction pathway for the dehydrogenative oxida-
tion of primary alcohols to carboxylic acids in aqueous media
is illustrated in Scheme 5. The first step of the reaction would
be the dehydrogenation catalyzed by 3 to afford the aldehyde.
This step would proceed by a similar mechanism to that shown
in Scheme 3. Then, addition of water to the aldehyde would
occur to give a gem-diol. Finally, dehydrogenation of the gem-
diol catalyzed by 3 would proceed to give the carboxylic acid
product.
ACKNOWLEDGMENT
This work was supported by JSPS KAKENHI Grant Number
JP16H01018 in Precisely Designed Catalysts with Customized
Scaffolding and Grant-in-Aid for Scientific Research (B) Grant
Number 26288047.
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
REFERENCES
(1) (a) Tsuji, J. Transition Metal Reagents and Catalysts: Innova-
tions in Organic Synthesis, John Wiley & Sons, Ltd., 2000. (b) Min-
gos, D. M. P.; Crabtree, R. H. (Eds), Comprehensive Organometallic
Chemistry III: From Fundamentals to Applications, vol. 10 (volume
editor: Ojima, I.), Elsevier, 2007. (c) Mingos, D. M. P.; Crabtree, R.
H. (Eds), Comprehensive Organometallic Chemistry III: From Fun-
damentals to Applications, vol. 11 (volume editor: Hiyama, T.), Else-
vier, 2007. (d) Hartwig, J. F. Organotransition Metal Chemistry:
From Bonding to Catalysis, University Science Books, 2010. (e)
Yamaguchi, R.; Fujita, K. Ligand Platforms in Homogenous Catalytic
Reaction with Metals, John Wiley & Sons, Ltd., 2015.
Scheme 5. Possible Reaction Pathway for the Dehydro-
genative Oxidation of Primary Benzylic Alcohols under
Reflux in Water Catalyzed by 3
(
2) (a) Lindström, U. M. Chem. Rev. 2002, 102, 2751-2772. (b)
Koba-yashi, S.; Manabe, K. Acc. Chem. Res. 2002, 35, 209-217. (c)
Manabe, K.; Kobayashi, S. Chem. Eur. J. 2002, 8, 4094-4101. (d) Li,
C.-J. Chem. Rev. 2005, 105, 3095-3165. (e) Chanda, A.; Fokin, V. V.
Chem. Rev. 2009, 109, 725-748. (f) Butler, R. N.; Coyne, A. G.
Chem. Rev. 2010, 110, 6302-6337. (g) Simon, M.-O.; Li, C.-J. Chem.
Soc. Rev. 2012, 41, 1415-1427. (h) Gawande, M. B.; Bonifácio, V. D.
B.; Luque, R.; Branco, P. S.; Varma, R. S. Chem. Soc. Rev. 2013, 42,
5522-5551.
Quantitative analysis of the evolved hydrogen gas in the de-
hydrogenative formation of the carboxylic acids from primary
alcohols was also conducted. The reaction of 4-methylbenzyl
alcohol on 2.5 mmol scale was conducted, as shown in eq 4.
Hydrogen (2 equiv.) evolved in 78% yield, which was the
corresponding amount expected because the carboxylic acid
product was also obtained in 78% yield.
(
3) (a) Fujita, K.; Tanino, N.; Yamaguchi, R. Org. Lett. 2007, 9,
109-111. (b) Fujita, K.; Yoshida, T.; Imori, Y.; Yamaguchi, R. Org.
Lett. 2011, 13, 2278-2281. (c) Kawahara, R.; Fujita, K.; Yamaguchi,
R. Angew. Chem. Int. Ed. 2012, 51, 12790-12794. (d) Yamaguchi, R.;
Kobayashi, D.; Shimizu, M.; Fujita, K. J. Organomet. Chem. 2017,
843, 14-19.
(
4) (a) Yamaguchi, R.; Ikeda, C.; Takahashi, Y.; Fujita, K. J. Am.
Chem. Soc. 2009, 131, 8410-8412. (b) Fujita, K.; Tanaka, Y.; Koba-
yashi, M.; Yamaguchi, R. J. Am. Chem. Soc. 2014, 136, 4829-4832.
(
5) (a) Kawahara, R.; Fujita, K.; Yamaguchi, R. J. Am. Chem. Soc.
012, 134, 3643-3646. (b) Fujita, K.; Ito, W.; Yamaguchi, R. Chem-
CatChem 2014, 6, 109-112. (c) Toyomura, K.; Fujita, K. Chem. Lett.
017, 46, 808-810.
6) Dehydrogenative oxidation of alcohols in aqueous media re-
2
2
(
ported by other research groups: (a) Feng, B.; Chen, C.; Yang, H.;
Zhao, X.; Hua, L.; Yu, Y.; Cao, T.; Shi, Y.; Hou, Z. Adv. Synth.
Catal. 2012, 354, 1559-1565. (b) Tang, L.; Sun, H.; Li, Y.; Zha, Z.;
Wang, Z. Green Chem. 2012, 14, 3423-3428. (c) Sawama, Y.; Morita,
K.; Yamada, T.; Nagata, S.; Yabe, Y.; Monguchi, Y.; Sajiki, H. Green
Chem. 2014, 16, 3439-3443. (d) Ngo, A. H.; Adams, M. J.; Do, L. H.
Organometallics 2014, 33, 6742-6745.
In summary, we have designed and prepared the new dica-
tionic iridium catalyst 3 bearing a chelating bidentate ligand
that comprises NHC and α-hydroxypyridine moieties. Catalyst
3 exhibited high activity in aqueous media for the dehydro-
genative oxidation of secondary alcohols to ketones accompa-
nying the evolution of hydrogen. Furthermore, the dehydro-
genative transformation of primary alcohols in aqueous media
to give carboxylic acids was also accomplished by employing
catalyst 3 without using base.
(
7) There are a few publications describing the dehydrogenative
oxidation of alcohols catalyzed by NHC-iridium based complexes: (a)
Prades, A.; Corberán, R.; Poyatos, M.; Peris, E. Chem. Eur. J. 2008,
14, 11474-11479; (b) Gülcemal, S.; Gülcemal, D.; Whitehead, G. F.
S.; Xiao, J. Chem. Eur. J. 2016, 22, 10513-10522.
(
8) During the present investigations and preparation of the manu-
script, Grotjahn, Webster, and Papish have very recently reported the
synthesis of the ligand comprises NHC and α-hydroxypyridine moie-
ties by a route different from our present study. They have also pre-
pared the monocationic iridium complex bearing this ligand and in-
vestigated its catalytic activity in hydrogenation of carbon dioxide and
dehydrogenation of formic acid. Siek, S.; Burks, D. B.; Gerlach, D.
L.; Liang, G.; Tesh, J. M.; Thompson, C. R.; Qu, F.; Shankwitz, J. E.;
ASSOCIATED CONTENT
Supporting Information
The Supporting Information is available free of charge on the
ACS Publications website at DOI: 10.1021/acscatal. *****.
Experimental details and characterization data (PDF)
X-ray crystallographic data for 3 (CIF)
ACS Paragon Plus Environment

 Fujita, Ken-Ichi
Fujita, Ken-Ichi
 Tamura, Ryuichi
Tamura, Ryuichi
 Tanaka, Yuhi
Tanaka, Yuhi
 Yoshida, Masato
Yoshida, Masato
 Onoda, Mitsuki
Onoda, Mitsuki
 Yamaguchi, Ryohei
Yamaguchi, Ryohei