X. Lin et al.
Molecular Catalysis 455 (2018) 143–149
to 10 h and 20 h to take full advantage of the high active catalyst
(Table 2, entries 10 and 11). Ethylbenzene conversion of 79% is
achieved as reaction time is prolonged to 10 h, and it is up to 82% for
Table 2
Catalytic performance for oxidation of ethylbenzene by S–CoNC.a
Entry
Catalyst
Conversion (%)
Selectivity (%)
2
0 h. S–CoNC is of higher catalytic performance under similar reaction
AcPO
PEA
BA
conditions than some reported catalysts (Table S1), as we know, it is so
far the highest conversion rate of ethylbenzene under solvent-free and
1
2
3
4
5
6
7
8
9
Blank
Co(Phen)
S–CoNC-700
S–CoNC
S–CoNC-900
CoNC
S–NC
S–Co
S–CoNCm
6
60
79
76
91
72
78
75
80
80
90
88
87
92
91
–
35
17
24
9
28
22
25
20
20
10
12
13
8
5
4
–
–
–
–
–
–
–
–
–
–
–
–
–
–
O
2
as oxidant. Unfortunately, the selectivity of AcPO is not further
improved.
In order to thoroughly investigate the active sites in the reaction
2
(SCN)
2
33
46
62
37
8
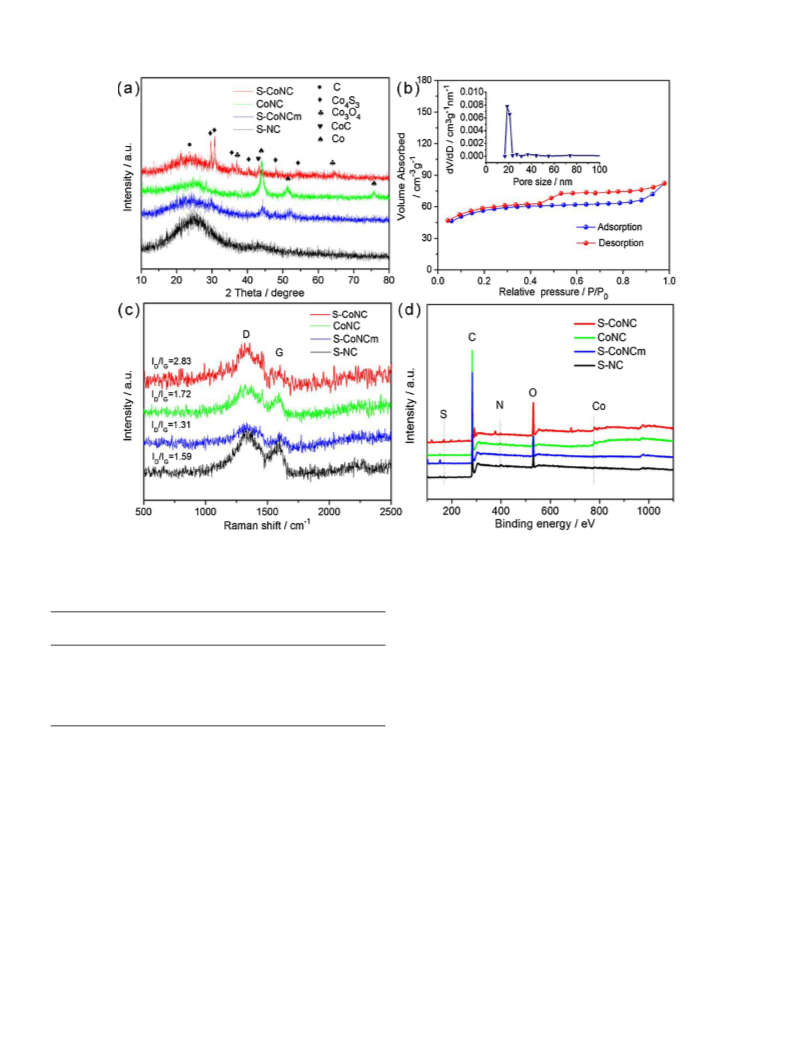
system, some detailed discussion was conducted. As shown in Fig. 1a,
there are only metallic Co peaks in CoNC and S–CoNCm besides gra-
phite-type carbon peak in XRD patterns, and the catalytic performance
of CoNC, S–CoNCm and S–NC is far lower than S–CoNC, suggesting that
7
39
12
79
82
8
65
61
–
b
10
11
12
13
14
15
16
S–CoNC
4 3
Co S may be the active site rather than metallic Co in the reaction
S–CoNCc
system. Remarkably, no cobalt sulfides are detected in S–CoNCm de-
rived from the pyrolysis of mercapto acetic acid, demonstrating that
KSCN with strong complexing ability has an important role in the
preparation of high performance samples. These samples have high
specific surface area and similar pore size besides S–CoNCm, demon-
strating that KSCN is used as a pore-forming agent in the process of
preparing sample (Table 1). According to the Raman spectrum (Fig. 1c),
d
S–CoNC-R
e
f
S–CoNC-R-C1
S–CoNC-R-C2
S–CoNCg
9
–
19
S–CoNCh
15
81
a
Reaction conditions: 120 °C, 5 h, 10 mL of ethylbenzene (82 mmol), 30 mg
of catalyst, and 0.8 MPa of O , acetophenone (AcPO), phenethyl alcohol (PEA),
2
D G
the I /I ratio of S–CoNC (2.83) is significantly larger than that of other
and benzaldehyde (BA).
b
samples (1.72, 1.59, and 1.31), indicating that S–CoNC possesses more
defects than other samples, which are beneficial to enhancing catalytic
performance of samples [30]. It is also proven that the defect of carbon
based materials is not only improved by doping heteroatoms, but also
linked with the connection between metal and heteroatom. Thus, we
propose that Co
sulfur is beneficial for inhibiting the growth of metal particles as de-
monstrated by Fig. 2. A large number of SO groups with chemical
inactivity and few eCeSeC and eC]Se structures are presented in the
S 2p of SeNC and SeCoNCm (Fig. S3), suggesting that there are hardly
any metal sulfides in these samples. The elements analysis of samples is
4 3
summarized in Table S2. To sum up, Co S should be the catalytic ac-
tive site for the aerobic oxidation of ethylbenzene.
Stability is an important parameter for judging the performance of
catalyst. Unexpectedly, an extremely low conversion was obtained after
the second reaction (Table 2, entry 12). There is no apparent agglom-
eration in the reacted catalyst (Fig. S5). It is amazing that S–CoNC re-
covers the conversion and selectivity after annealing the reacted cata-
lyst at the same temperature (Table 2, entries 13 and 14). The reacted
catalyst (S–CoNC-R) and reannealed catalyst (S–CoNC-R-C) were
characterized in order to investigate the differences. The XRD patterns
show that there are many undesired peaks in S–CoNC-R, but there are
Reaction time was prolonged to 10 h based on other same conditions.
Reaction time was prolonged to 20 h based on other same conditions.
The second reaction at the same conditions after the first reaction.
The catalyst reacted for 1 time was reannealed at the same temperature.
The catalyst was annealed for another 1 time based on the former condi-
c
d
e
f
4 3
S may cause more defects in samples. And the doped
tions of e.
g
Reaction conditions are consistent with this work beside adding butylated
hydroxytoluene (BHT) (1 mL).
x
h
1
mmol of fluobenzene was added based on the same conditions.
pyridinic N is more than that of pyrrolic N and graphitic N, and pyr-
idinic N which locates at the defect sites in the basal plane and is
bonded to two adjacent carbon atoms provides highly active sites due to
lower adsorption energy [34]. The Co 2p at 780.9 and 796.9 eV can be
divided into Co 2p3/2 and Co 2p1/2, respectively, which are fitted into
Co(III), Co(II), and satellites peaks (Fig. 3c). A high resolution of S 2p
spectrum mainly shows three different peaks (Fig. 3d). The peaks at
63.2 eV of S 2p3/2 and 164.6 eV of S 2p1/2 are consistent with reported
literature and assigned to eCeSeCe and eC]Se bonds [30,35]. The
other peaks belong to SO groups with chemically inactive and occur at
the edge of the carbon skeleton, meanwhile, the formation of SO
1
x
x
groups can be proved by the O 1s (Fig. S2). It is reported that the
components at 163.2 eV are corresponding to metal–sulfur bonds [29].
Sulfur and nitrogen-doped metal carbon catalysts have been ex-
tensively researched, but there are few reports in heterogeneous cata-
almost Co
be reformed through high temperature anneal, and proving the im-
portance of Co in the reaction system. Moreover, S–CoNC-R has no
4 3 4 3
S peaks in S–CoNC-R-C (Fig. 4), suggesting that Co S can
4 3
S
2
lysis. The oxidation of ethylbenzene with solvent-free and O as oxidant
was probed as the model reaction. It is known that Co/N-ligands
complexes (especially Co/phthalocyanines) have catalytic performance
for some catalytic reactions [36,37], so the catalytic activity of Co
(
2 2
Phen) (SCN) was tested (Table 2, entry 2). The influence of pyrolysis
temperature before studying the reaction system was investigated. The
S–CoNC pyrolyzed at 800 °C shows the highest conversion of ethyl-
benzene (62%) and selectivity of AcPO (91%) compared with that of
the catalysts pyrolyzed at 700 and 900 °C (Table 2, entries 3–5), de-
monstrating that 800 °C is the best temperature for preparing catalyst.
The blank testing (without catalyst) gives extremely low catalytic ac-
tivity in this system (Table 2, entry 1). In order to clearly study the
effect of catalyst compositions for catalytic performance, some control
experiments were tested (Table 2, entries 6–9). It is remarkable that
there is poor catalytic performance in CoNC, S–NC, and S–CoNCm. The
conversion of ethylbenzene is only around 10% and the selectivity of
AcPO is also lower than 80%, which is obviously far lower than S–CoNC
at the same reaction system. Moreover, the reaction time was prolonged
Fig. 4. XRD patterns of S–CoNC-R and S–CoNC-R-C.
147

 Lin, Xiu
Lin, Xiu
 Jie, Shanshan
Jie, Shanshan
 Liu, Zhigang
Liu, Zhigang