14 of 15
MEKKAOUI ET AL.
8
| CONCLUSION
[10] A. Louroubi, H. Ouahine, B. Boualy, M. Ait Ali, L. El
Firdoussi, J. Adv. Catal. Sci. Technol. 2016, 3, 63.
[
[
[
11] Ullmann's Encyclopedia of Industrial Chemistry, 5th ed. Vol.
A8, Willey–VCH, Weinheim 1985, 25.
12] D. Singh, S. M. Rao, A. K. Tripathi, Naturwissenschaften 1984,
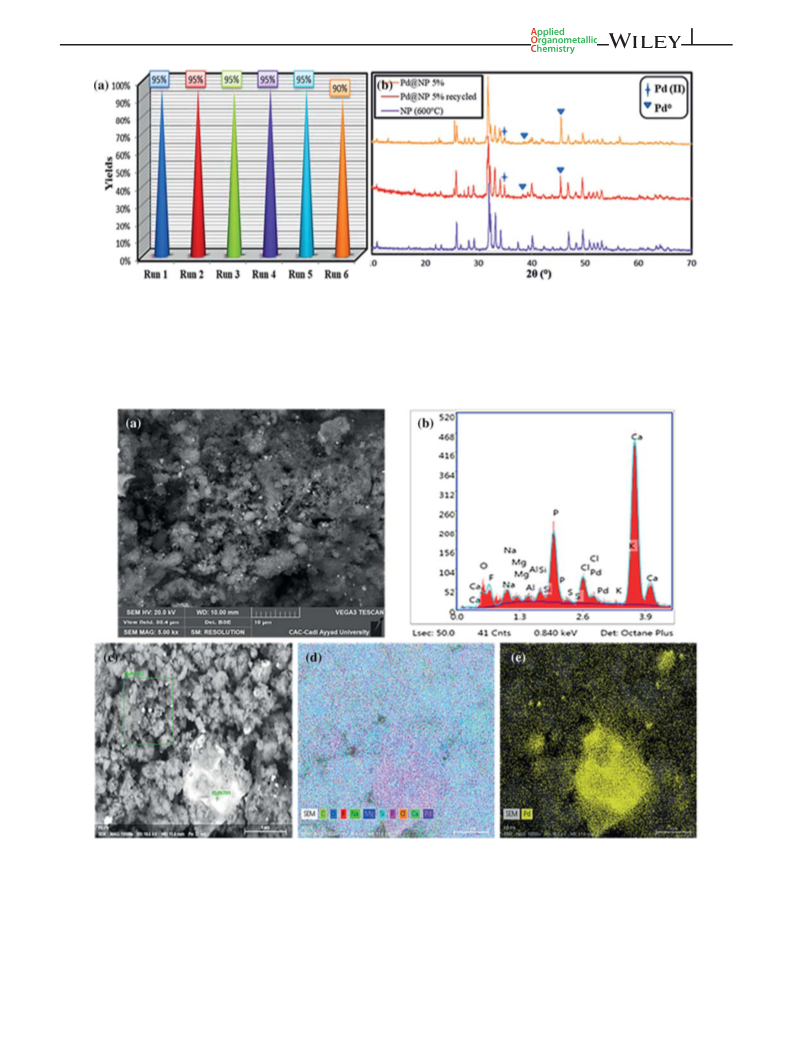
In summary, novel palladium nanoparticles Pd (0) with
a crystallite size of 10.88 nm were synthesized and
monitored by UV–Vis without using any reducing
agent. The palladium nanoparticles were supported on
mesoporous NP support leading to a different ratio
catalyst (Pd@NP 10%, 5% and 1%). The prepared
nanocatalysts were fully characterized using various
analysis techniques, and their performance was first
investigated in the solvent-free dehydroaromatization of
himachalene mixture (Cedrus atlantica oil) as a model
substrate before being extended to different natural
terpenic olefins. The Pd@NP exhibits marked selectiv-
ity, good reusability and high stability in solvent-free
dehydroaromatization of terpenes.
71, 265.
13] R. P. Adams, H. F. Linskens, J. F. Jackson (Eds), Essential Oils
and Waxes. Modern Methods of Plant Analysis, Vol. 5, Springer
1991 159.
[14] M. Dı ꢀg rak, A. I_ lçim, M. Hakkı Alma, Phyther. Res. 1999,
13, 584.
[
[
15] K. Mori, Pure Appl. Chem. 2001, 73, 601.
16] A. Chaudhary, P. Sharma, G. Nadda, D. K. Tewary, B. Singh,
J. Insect Sci. 2011, 11, 1.
[
[
[
17] J.-C. Chalchat, R.-P. Garry, A. Miehet, B. Benjilali, J. Essent.
Oil Res. 1994, 6, 323.
18] R. J. Bartelt, A. A. Cossé, B. W. Zilkowski, D. Weisleder,
F. A. Momany, J. Chem. Ecol. 2001, 27, 2397.
19] K. Mori, Tetrahedron: Asymmetry 2005, 16, 685.
ORCID
[20] A. Chaudhary, S. Sood, P. Das, P. Kaur, I. Mahajan, A. Gulati,
B. Singh, EXCLI j. 2014, 13, 1216.
[21] K. Spielmann, R. M. De Figueiredo, J. M. Campagne, J. Org.
Chem. 2017, 82, 4737.
[22] I. Hossini, M. A. Harrad, M. A. Ali, L. El Firdoussi, A. Karim,
P. Valerga, M. C. Puerta, Molecules 2011, 16, 5886.
[
[
[
23] R. C. Pandey, S. Dev, Tetrahedron 1968, 24, 3829.
REFERENCES
24] G. Mehta, S. K. Kapoor, J. Org. Chem. 1974, 39, 2618.
25] R. J. Bartelt, D. Weisleder, F. A. Momany, Synthesis 2003,
[
1] a) B. Khodadadi, M. Bordbar, M. Nasrollahzadeh, J. Colloid
Interface Sci. 2017, 490, 1; b) C. Xu, M. Nasrollahzadeh,
M. Selva, Z. Issaabadi, R. Luque, Chem. Soc. Rev. 2019, 48,
1
, 117.
26] S. E. Muto, M. Bando, K. Mori, Eur. J. Org. Chem. 2004, 2004,
946.
27] S. P. Chavan, H. S. Khatod, Tetrahedron: Asymmetry 2012, 23,
410.
[
[
[
[
4
791; c) C. Xu, M. Nasrollahzadeh, M. Sajjadi, M. Maham,
R. Luque, A. R. Puente-Santiago, Renew. Sustain. Energy Rev.
019, 112, 195.
1
2
1
[
2] a) Z. Issaabadi, M. Nasrollahzadeh, S. M. Sajadi, J. Cleaner
Prod. 2017, 142, 3584; b) M. Nasrollahzadeh, S. M. Sajadi,
J. Colloid Interface Sci. 2016, 462, 243; c) A. Hatamifard,
M. Nasrollahzadeha, J. Lipkowski, RSC Adv. 2015, 5, 91 372.
3] a) S. S. Momeni, M. Nasrollahzadeh, A. Rustaiyan, J. Colloid
Interface Sci. 2017, 499, 93; b) T. Baran, M. Nasrollahzadeh,
Carbohydr. Polym. 2019, 222, 115 029.
4] a) M. Nasrollahzadeh, E. Mehdipour, M. Maryami, J. Mater.
Sci.: Mater. Electron. 2018, 29, 17 054; b) M. Nasrollahzadeh,
M. Sajjadi, J. Dadashi, H. Ghafuri, Adv. Colloid Interface Sci.
28] J. B. Bredenberg, H. Erdtman, K.-I. Persson, H. Dam,
B. Sjöberg, J. Toft, Acta Chem. Scand. 1961, 15, 685.
29] B. N. Joshi, R. Seshadri, K. K. Chakravarti,
S. C. Bhattacharyya, Tetrahedron 1964, 20, 2911.
[
[
[
[
[
30] T. C. Joseph, S. Dev, Tetrahedron 1968, 24, 3809.
31] B. Abouhamza, S. Allaoud, A. Karim, Molecules 2001, 6, M236.
32] G. H. Jimenez-Aleman, T. Schöner, A. L. Montero-Alejo,
W. Brandt, W. Boland, ARKIVOC 2012, 2012, 371.
[
[
33] G. W. Mcgraw, R. W. Hemingway, L. L. Ingram, C. S. Canady,
W. B. Mcgraw, Environ. Sci. Technol. 1999, 33, 4029.
2
020, 276, 102 103.
34] D. Makarouni, S. Lycourghiotis, E. Kordouli, K. Bourikas,
C. Kordulis, V. Dourtoglou, Appl. Catal. Environ. 2018,
[
5] F. El Aroui, S. Lahrich, A. Farahi, M. Achak, L. El Gaini,
M. Bakasse, A. Bouzidi, M. A. El Mhammedi, Electroanalysis
224, 740.
2
014, 26, 1751.
[
35] E. Yılmazo ꢀg lu, M. Akgün, J. Supercrit. Fluids 2018, 131, 37.
36] A. Wróblewska, P. Miądlicki, J. Tołpa, J. Sre nꢁ scek-Nazzal,
Z. C. Koren, B. Michalkiewicz, Catalysts 2019, 9, 396.
[
6] A. Hassine, S. Sebti, A. Solhy, M. Zahouily, C. Len,
M. N. Hedhili, A. Fihri, Appl. Catal. Gen. 2013, 450, 13.
7] A. A. Mekkaoui, S. Jennane, A. Aberkouks, B. Boualy,
A. Mehdi, M. Ait Ali, L. El Firdoussi, S. El Houssame, Appl.
Organomet. Chem. 2019, 33, e5117.
[
[
[37] C. Perego, P. Ingallina, Catal. Today 2002, 73, 3.
[38] J. Du, H. Xu, J. Shen, J. Huang, W. Shen, D. Zhao, Appl. Catal.
Gen. 2005, 296, 186.
[
[
8] S. Sebti, M. Zahouily, H. Lazrek, J. Mayoral, D. Macquarrie,
Curr. Org. Chem. 2008, 12, 203.
[39] M. A. Martín-Luengo, M. Yates, M. J. Martínez Domingo,
B. Casal, M. Iglesias, M. Esteban, E. Ruiz-Hitzky, Appl. Catal.
Environ. 2008, 81, 218.
9] a) Y. E. L. Bouabi, A. Farahi, M. Achak, M. Zeroual, K. Hnini,
S. El Houssame, M. Bakasse, A. Bouzidi, M. A. El Mhammedi,
J. Taiwan Inst, Chem. Eng. 2016, 66, 33; b) M. Shokouhimehr,
S. M. G. Yek, M. Nasrollahzadeh, A. Kim, R. S. Varma, Appl.
Sci. 2019, 9, 4183.
[40] M. A. Martin-Luengo, M. Yates, E. S. Rojo, D. Huerta
Arribas, D. Aguilar, E. Ruiz Hitzky, Appl. Catal. Gen. 2010,
387, 141.

 Mekkaoui, Ayoub Abdelkader
Mekkaoui, Ayoub Abdelkader
 Aberkouks, Abderrazak
Aberkouks, Abderrazak
 Fkhar, Lahcen
Fkhar, Lahcen
 Ait Ali, Mustapha
Ait Ali, Mustapha
 El Firdoussi, Larbi
El Firdoussi, Larbi
 El Houssame, Soufiane
El Houssame, Soufiane