European Journal of Organic Chemistry
10.1002/ejoc.202000767
FULL PAPER
*
CDCl
3
) δ 7.33 (bs, 1H, NH), 6.19 (dt, J = 7.7, 2.5 Hz, 1H), 2.82 –
439.1933. N,N-diisopropyl amine was distilled over KOH pellets
272 (m, 2H), 2.55 – 245 (m, 1H) ppm. Subsequently, the
before use.
trifluoroacetate ester (922 mg) was hydrolysed, and purified by
means of column chromatography (EtOAc/pentane, 30:70 and
4
-((tert-butyldiphenylsilyl)oxy)-2-(methoxycarbonyl)
cyclopent ene-1-carboxylic acid (14a): Compound 14b was
dissolved in a mixture of THF/MeOH (83:17, 66 mL), and a
solution of KOH (603 mg, 10.7 mmol) in water (11 mL) was added.
The mixture was stirred at rt for 6 h, subsequently acidified with 1
M HCl until pH ~ 1, and extracted with EtOAc (4 × 100 mL). The
5
6
1
2
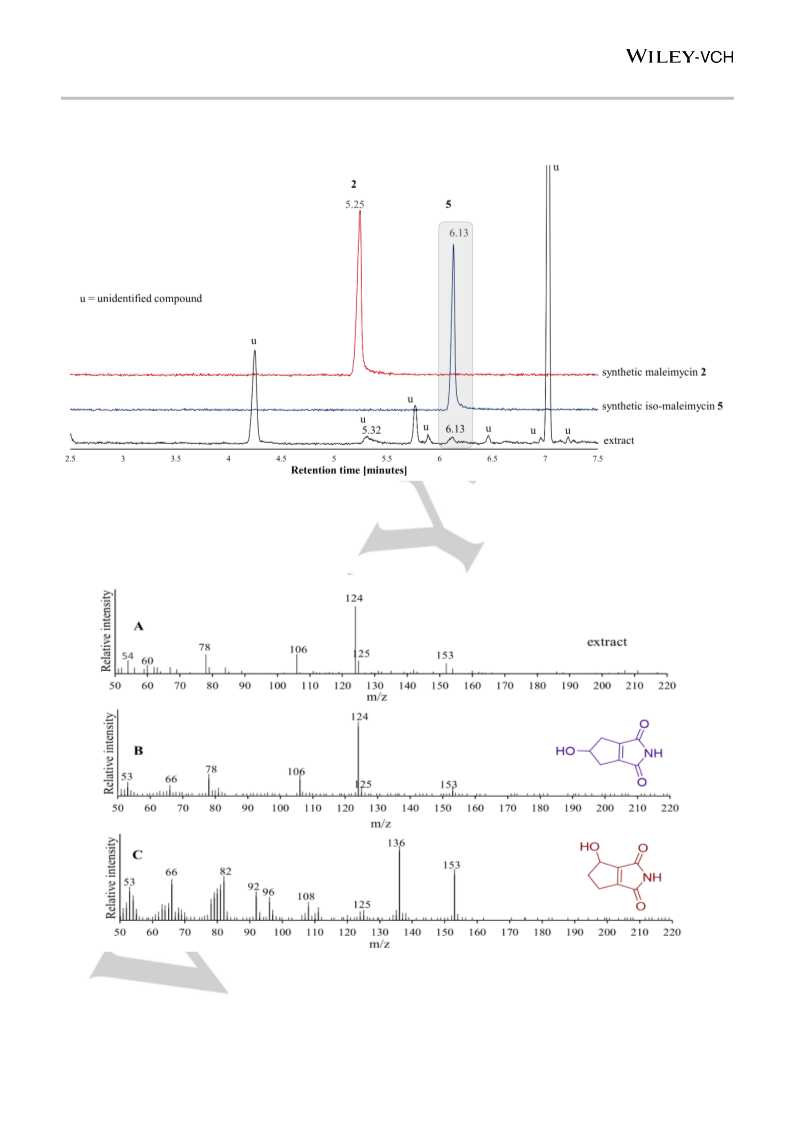
0:50), which afforded rac-maleimycin 2 (409 mg, 2.67 mmol,
3 % yield, based on the bromide), as a white crystalline solid. Mp
1
09 – 112 °C; H-NMR (400 MHz, MeOD) δ 5.09 – 5.01 (m, 1H),
.84 – 2.64 (m, 2H), 2.57 – 2.45 (m, 1H), 2.26 – 2.13 (m, 1H) ppm.
C-NMR (101 MHz, MeOD) δ 168.1, 167.5, 156.4, 153.1, 69.7,
1
3
combined organic layers were dried over MgSO
4
and
3
7 7 3
8.2, 23.6 ppm. HRMS (negative mode) calcd for C H NO {M-
-
concentrated by means of rotary evaporation. The crude product
was purified by column chromatography (pentane/EtOAc/AcOH,
gradient 90:9:1, 80:19:1 and 70:29:1) to give 14a (978 mg, 2.30
H} : 152.0353, found: 152.0356. Physical data of maleimycin
were in agreement with those reported in the literature.6a-d
dimethyl
cyclopentane-1,2-dicarboxylate (17): To a solution of racemic
trans)-dimethyl 4-oxocyclopentane-1,2-dicarboxylate 15 (2.00 g,
.99 mmol) in MeOH (40 mL) was added NaBH (189 mg, 5.00
mmol), while cooling on ice. The solution was left stirring at 0 C
for 20 min and quenched with 1 M HClaq (30 mL) followed by
dilution with water (40 mL). The solution was extracted with EtOAc
(1S,2S)-4-((tert-butyldiphenylsilyl)oxy)
mmol, 64% yield) as an off-white crystalline solid. Mp 109 ‒
1
117 °C, H-NMR (400 MHz, CDCl
3
) δ 7.68 – 7.58 (m, 4H), 7.84 –
(
9
733 (m, 6H), 4.44 (tt, J = 5.6, 4.0 Hz, 1H), 3.89 (s, 3H), 3.09 –
2.98 (m, 2H), 2.89 – 2.88 (m, 2H), 1.04 (s, 9H) ppm. 13C-NMR
4
o
(101 MHz, CDCl
3
) δ 168.4, 163.1, 146.5, 135.63, 135.62, 135.55,
133.7, 133.4, 129.9, 129.8, 127.8, 127.7, 69.0, 53.7, 46.6, 45.3,
+
26.8, 19.0 ppm. HRMS calcd for C24
found: 447.1591.
28 5
H O Si {M+Na} : 447.1598,
(
3 × 40 mL) and the combined organic layers were dried over
MgSO , concentration by rotary evaporation provided the crude
alcohol 16 as a colourless oil (1.70 g) in sufficient purity to be used
4
methyl 4-((tert-butyldiphenylsilyl)oxy)-2-carbamoylcyclopent
-1-ene-1-carboxylate (18):
1
in the next step. H-NMR (400 MHz, CDCl
3
) δ 4.41 (tt, J = 4.6, 2.3
Hz, 1H), 3.72 (s, 3H), 3.71 (s, 3H), 3.43 (dt, J = 10.0, 7.7 Hz, 1H),
Method 1, direct conversion of diester 14b. According to a
modified procedure , ammonium chloride (331 mg, 6.19 mmol)
3
5
2
5
.25 (ddd, J = 10.0, 7.0, 4.8 Hz, 1H), 2.25 (ddd, J = 14.6, 10.0,
14
.0 Hz, 1H), 2.15 (ddt, J = 13.3, 8.3, 2.2Hz, 1H), 2.05 – 1.92 (m,
was suspended in toluene (10 mL). While cooling the suspension
in an ice bath, 2 M trimethyl aluminium (solution in hexanes, 3.1
mL, 6.19 mmol) was added dropwise, after which evolution of
methane was observed. The ice bath was removed and the
reaction mixture was stirred at rt for 3 h. A solution of 14b (905
mg, 2.06 mmol) in toluene (10 mL) was added dropwise to the
1
3
H) ppm. C-NMR (101 MHz, CDCl
3
) δ 176.1, 175.2, 72.9, 52.4,
2.2, 45.3, 45.2, 39.9, 38.6 ppm. HRMS calcd for C
9
14 5
H O
+
{
M+Na} : 225.0734, found: 225.0734. Crude 16 (1.70 g, 8.41
mmol) and imidazole (1.15 g, 16.8 mmol) were dissolved in DMF
34 mL), the solution was cooled in an ice bath. TBDPSCl (2.4 mL,
.50 g, 9.01 mmol) was added dropwise via syringe, after which
(
2
°
reaction mixture, subsequently the mixture was heated to 50 C
it was allowed to reach rt and left stirring overnight. The reaction
mixture was diluted with water (50 mL) and extracted with EtOAc
and stirred for 18 h. The reaction mixture was cooled in an ice
bath and quenched with 1 M HClaq (15 mL) and stirred for 15 min.
The resulting clear biphasic mixture was extracted with EtOAc (5
(
(
3 × 50 mL). The combined organic layers were washed with brine
4 × 50 mL), dried over MgSO and concentrated by means of
4
×
15 mL), the combined organic layers were dried over MgSO
4
rotary evaporation. The crude material was purified by flash
column chromatography (Et O/pentane, gradient 10:90 and
0:80), to afford 17 (3.38 g, 7.67 mmol, 77% yield, over two steps)
and concentrated in vacuo. The crude product was purified by
column chromatography, (pentane/EtOAc, 60:40) affording 18
2
2
(
467 mg, 1.10 mmol, 53% yield) as a pale brown crystalline solid.
1
as a colourless oil. H-NMR (400 MHz, CDCl
3
) δ 7.66 – 7.59 (m,
Method 2, from acid-ester 14a. Following a modified literature
4
3
2
9
1
4
H), 7.47 – 7.33 (m, 6H), 4.31 (pent., J = 4.3 Hz, 1H), 3.71 (s, 3H),
1
1
procedure , to a solution of 14a (785 mg, 1.85 mmol) in CH
2 2
Cl
.67 (s, 3H), 3.61 (dt, J = 9.8, 8.0 Hz, 1H), 3.18 – 305 (m, 1H),
.17 – 1.96 (m, 3H), 1.75 (ddd, J = 13.2, 8.9, 5.1 Hz, 1H), 1.03 (s,
(
7.4 mL) were added Et N (1.0 mL, 748 mg, 7.40 mmol) and DMF
3
1
3
(0.1 mL). The mixture was cooled in an ice bath, and thionyl
chloride (0.5 mL, 885 mg, 7.45 mmol) was added dropwise,
followed by stirring for 15 min. The reaction mixture was added
H) ppm. C-NMR (101 MHz, CDCl
35.69, 133.9, 133.8, 129.7, 127.7, 127.6, 74.0, 52.1, 52.0, 45.0,
4.3, 39.7, 39.1, 26.7, 19.4 ppm. HRMS calcd for C25 Si
3
) δ 175.6, 174.7, 135.73,
32 5
H O
+
4 aq
dropwise to an ice-cold solution of concentrated 14 M NH OH
{
M+Na} : 463.1911, found: 463.1911.
(
100 mL) under vigorous stirring, and the resulting turbid mixture
was extracted with EtOAc (3 × 50 mL), the combined organic
layers were washed with brine (50 mL), dried over MgSO and
concentrated in vacuo. Purification by column chromatography
pentane/EtOAc, 60:40) afforded 18 (420 mg, 0.99 mmol, 54%
dimethyl 4-((tert-butyldiphenylsilyl)oxy)cyclopent-1-ene-1,2-
dicarboxylate (14b): According to modified literature
a
4
1
3
procedure , LDA was generated by adding n-BuLi (1.6 M solution
in hexanes, 7.6 mL, 12.1 mmol) to a solution of N,N-diisopropyl
amine* (1.8 mL, 12.8 mmol) in dry THF (10 mL), at -78 C.
Compound 17 (2.35 g, 5.34 mmol) was dissolved in dry THF (25
mL) and added to the reaction mixture dropwise. To the resulting
bright yellow solution was added a solution of iodine (1.42 g, 5.61
mmol) in THF (31 mL). Subsequently, the purple-brown solution
was allowed to reach rt and carefully quenched by adding 1 M
HClaq (50 mL). The mixture was extracted with EtOAc (3 × 50 mL),
the combined organic layers were washed with brine (50 mL),
(
°
1
yield) as a pale brown crystalline solid. Mp 119 ‒ 128 °C; H-NMR
400 MHz, CDCl ) δ 8.62 (bs, 1H), 7.73 – 7.59 (m, 4H), 7.51 –
.30 (m, 6H), 5.91 (bs, 1H), 4.43 (pent., J = 5.1 Hz, 1H), 3.77 (s,
H), 3.03 – 2.93 (m, 2H), 2.93 – 2.82 (m, 2H), 1.05 (s, 9H) ppm.
(
3
7
3
13
C-NMR (101 MHz, CDCl
3
) δ 166.4, 165.7, 145.9, 135.65, 135.64,
1
4
4
34.0, 133.7, 132.2, 129.8, 129.7, 127.72, 127.67, 69.4, 52.4,
+
6.4, 45.6, 26.8, 19.1 ppm. HRMS calcd for C24
24.1939, found: 424.1920.
4
H29NO Si {M+H} :
dried over MgSO
4
and concentrated by means of rotary
evaporation. The concentrate was purified by column
chromatography (EtOAc/pentane, gradient 10:90 and 20:80), to
afford 14b as a viscous pale yellow syrup (1.73 g, 3.94 mmol,
5
-((tert-butyldiphenylsilyl)oxy)-5,6-dihydrocyclopenta
[c]
pyrrole-1,3(2H,4H)-dione (19): A round bottomed flask was
equipped with a stir bar and NaH 60% suspension in mineral oil
1
7
–
2
1
1
4% yield). H-NMR (400 MHz, CDCl
3
) δ 7.67 – 7.61 (m, 4H), 7.46
(
55 mg, 1.65 mmol) which was rinsed with pentane three times. A
7.34 (m, 6H), 4.55 (tt, J = 6.4, 4.4 Hz, 1H), 3.76 (s, 6H), 2.87 –
.69 (m, 4H), 1.05 (s, 9H) ppm. C-NMR (101 MHz, CDCl
65.6, 137.4, 135.7, 133.7, 129.8, 127.7, 71.5, 52.1, 44.1, 26.8,
30 5
9.0 ppm. HRMS calcd for C25H O Si {M+Na} : 439.1935, found:
*
solution of 18 (467 mg, 0.551 mmol) in THF (5.5 mL) was added
dropwise via syringe, while cooling the reaction mixture on ice.
After 15 min, when H evolution had ceased, the reaction mixture
2
1
3
3
) δ
+
6
This article is protected by copyright. All rights reserved.

 Minnaard, Adriaan J.
Minnaard, Adriaan J.
 Santiago, Isabel Nu?ez
Santiago, Isabel Nu?ez
 Uiterweerd, Michiel T.
Uiterweerd, Michiel T.
 van Wezel, Gilles P.
van Wezel, Gilles P.
 van der Heul, Helga
van der Heul, Helga