Russian Chemical Bulletin, International Edition, Vol. 56, No. 6, pp. 1234—1238, June, 2007
1234
Comparative thermolysis of βꢀ and αꢀpinenes in supercritical ethanol:
the reaction characterization and enantiomeric ratios of products*
c
b
a
c
A. M. Chibiryaev,a,b A. Yermakova, I. V. Kozhevnikov, O. I. Sal´nikova, and V. I. Anikeev
ꢀ
aN. N. Vorozhtsov Novosibirsk Institute of Organic Chemistry, Siberian Branch of the Russian Academy of Sciences,
9 prosp. Akad. Lavrent´eva, 630090 Novosibirsk, Russian Federation.
Fax: +7 (383) 330 9752. Eꢀmail: chibirv@nioch.nsc.ru
bDepartment of Natural Sciences, Novosibirsk State University,
2 ul. Pirogova, 630090 Novosibirsk, Russian Federation
cG. K. Boreskov Institute of Catalysis, Siberian Branch of the Russian Academy of Sciences,
5 prosp. Akad. Lavrent'eva, 630090 Novosibirsk, Russian Federation
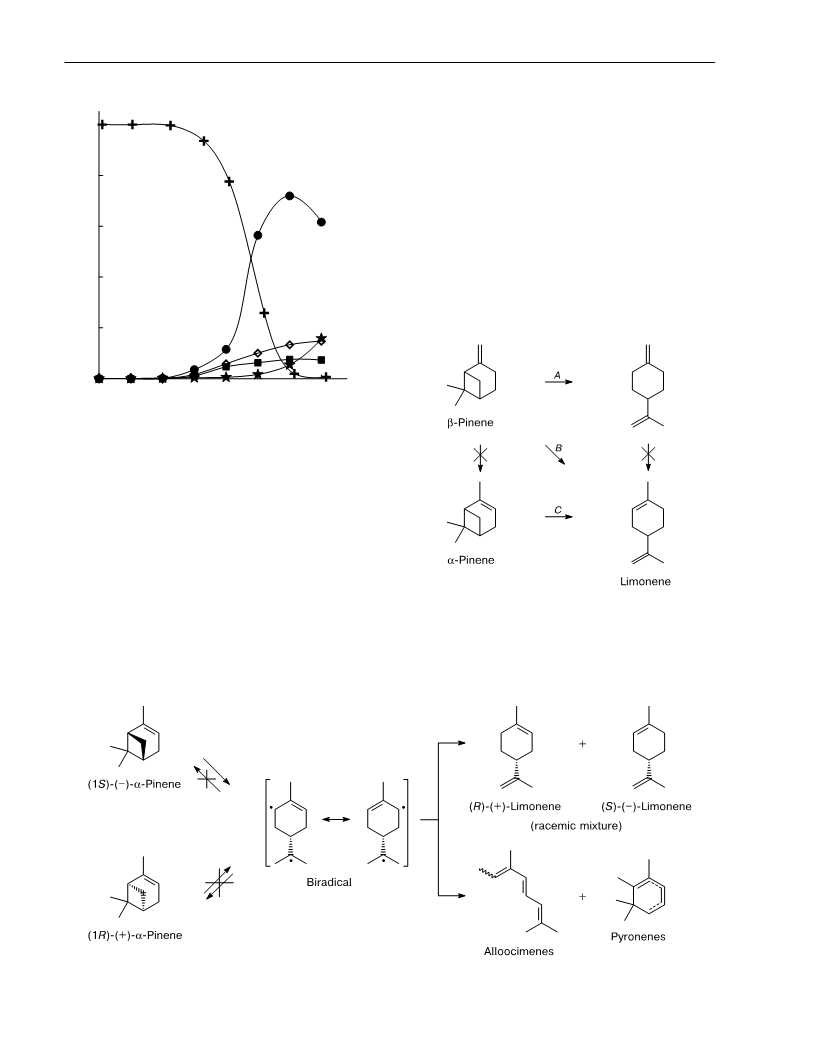
The thermolysis of βꢀpinene and the coꢀthermolysis of an equimolar mixture of βꢀ and
αꢀpinenes in supercritical ethanol were carried out. The reaction of βꢀpinene affords βꢀmyrcene
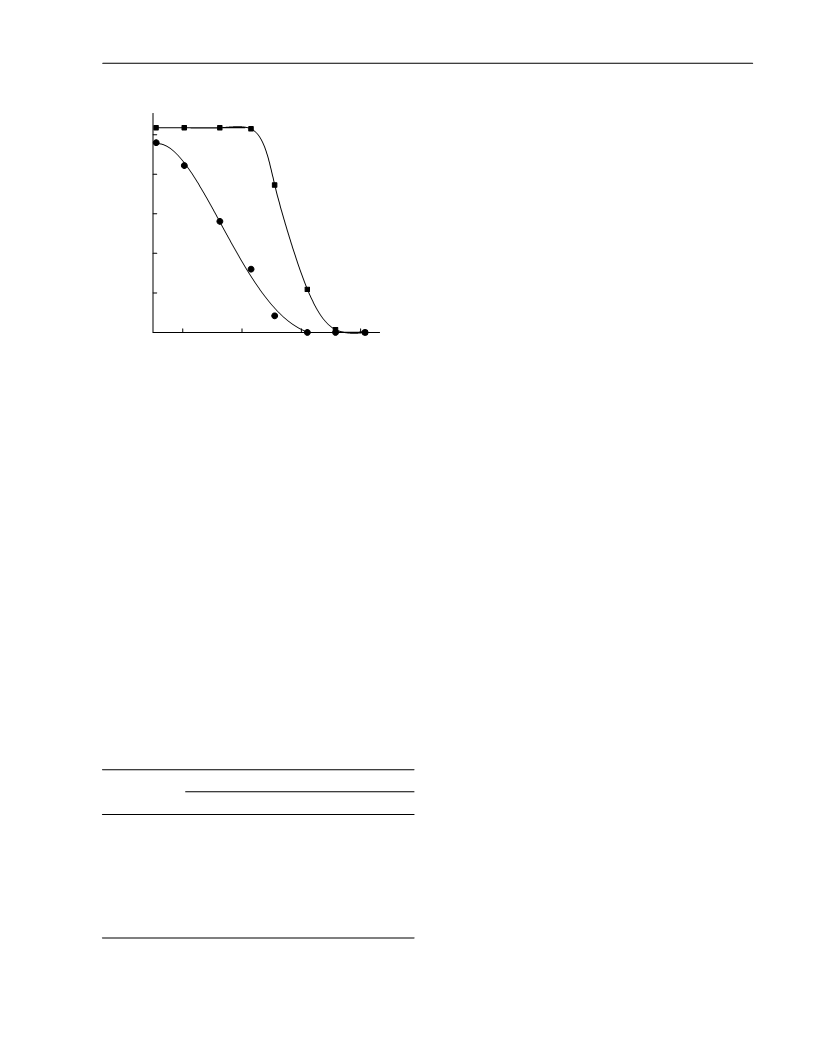
as the major product (>70%). Considerable differences in the temperature dependence of the
reaction rate were revealed for the transformations of βꢀpinene into βꢀmyrcene and of αꢀpinene
into limonene. The preꢀexponential factors and the activation energies were calculated. The
enantiomeric ratios of the thermolysis products of βꢀ and αꢀpinenes and limonene were estiꢀ
mated. The starting monoterpenes do not undergo racemization during thermolysis. The therꢀ
molysis of enantiomerically pure αꢀpinene affords racemic ( )ꢀlimonene, whereas (–)ꢀβꢀpinene
gives (–)ꢀlimonene. The enantiomeric ratio in the latter remains equal to the enantiomeric
purity of the starting βꢀpinene.
Key words: thermolysis, supercritical media, ethanol, monoterpenes, pinenes, βꢀmyrcene,
limonene.
Thermal transformations of terpene hydrocarbons proꢀ
vide the basis for the synthesis of polyunsaturated comꢀ
pounds (dienes, trienes, and aromatics), which are widely
used in organic synthesis and in the production of polyꢀ
mer adhesive materials. Limonene and βꢀmyrcene, which
are the major components of many essential oils and the
key substances of artificial perfumeryꢀcosmetic composꢀ
ites, can be synthesized by the thermolysis of the bicyclic
monoterpenes, such as αꢀ and βꢀpinenes.
the temperature dependence of the reaction rate, to reꢀ
veal the differences in thermal transformations of βꢀ and
αꢀpinenes, and to determine the enantiomeric ratios of
the thermolysis products of pinenes and limonene.
Experimental
(–)ꢀβꢀPinene (>99%, sum of enantiomers, Fluka),
(+)ꢀαꢀpinene (>98 %, sum of enantiomers, Aldrich), and
(+)ꢀlimonene (>99 %, sum of enantiomers, Aldrich) were used
in experiments.
Earlier, we have demonstrated1 that the thermolysis of
terpenes can be performed in supercritical media. For
example, it was found that the thermolysis of αꢀpinene in
supercritical alcohols (methanol, ethanol, or propanꢀ1ꢀol)
gives acyclic and monocyclic unsaturated nonaromatic
compounds, whereas limonene does not undergo thermal
transformations under these conditions. In continuing
studies of transformations of terpenes in supercritical
media, it was of interest to compare the reactivity of strucꢀ
turally similar αꢀ and βꢀpinenes in the same supercritical
solvent.
The thermolysis of terpenes in supercritical ethanol was carꢀ
ried out in a laboratory apparatus using a tubular flowꢀtype
reactor with a length of 3 m and a diameter of 1.75 mm; the
volume of the reaction zone was 7 mL. A solution of pinene (or a
mixture of pinenes) in 96% ethanol was fed to the reactor using a
piston pump. Then the reaction mixture was successively passed
to a heat exchanger for cooling and to a separator, where samples
were taken for analysis. In all cases, the compounds were kept in
the active zone of the reactor (the residence time τ) for 70 s. This
time was calculated as the ratio of the reaction zone volume (mL)
to the volumetric rate, at which the reaction mixture was fed to
the reactor (mL min–1).
The aim of the present study was to investigate the
thermolysis of βꢀpinene in supercritical ethanol, to find
The compositions of the reaction products were analyzed by
gas chromatography mass spectrometry on a Hewlett—Packard
5890/II gas chromatograph equipped with an HP MSD 5971
quadrupole mass spectrometer as the detector. A 30ꢀm HPꢀ5
* Dedicated to the memory of Academician N. N. Vorozhtsov
on the 100th anniversary of his birth.
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 6, pp. 1188—1192, June, 2007.
1066ꢀ5285/07/5606ꢀ1234 © 2007 Springer Science+Business Media, Inc.

 Chibiryaev
Chibiryaev
 Yermakova
Yermakova
 Kozhevnikov
Kozhevnikov
 Sal'nikova
Sal'nikova
 Anikeev
Anikeev