H. Matsunaga et al.
14. Abbott AP, Capper G, Davies DL, Rasheed RK, Tambyrajah V.
Novel solvent properties of choline chloride/urea mixtures. Chem
Commun. 2003;39:70–1.
15. Wilkes JS. A short history of ionic liquids—from molten salts to
neoteric solvents. Green Chem. 2002;4:73–80.
16. Matsunaga H, Katoh K, Habu H, Noda M, Miyake A. Preparation
and thermal decomposition behavior of high-energy ionic liquids
based on ammonium dinitramide and amine nitrates. Trans
JSASS Aerosp Technol Jpn. 2018;16:88–92.
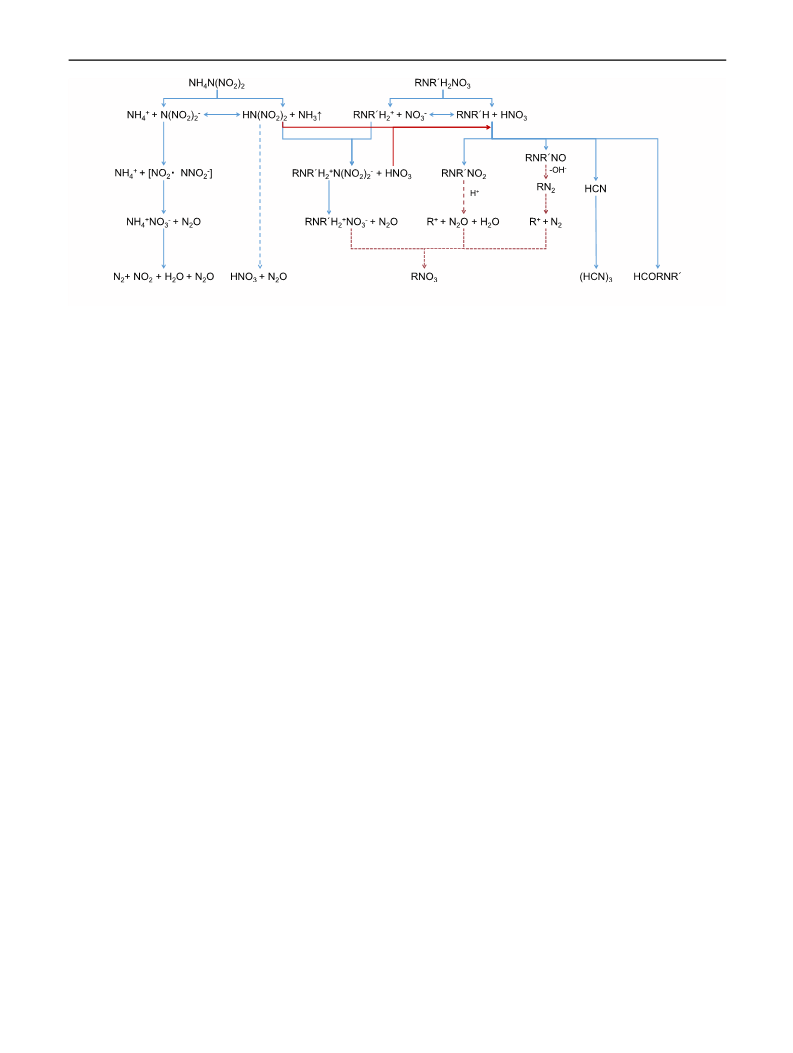
of highly acidic products, such as HN(NO2)2 and HNO3
and the promotion of nitration and nitrosation of amines
derived from the dissociation of amine nitrates, which is
thought to occur in mixtures of ADN and any aliphatic
amine nitrate.
Acknowledgements This work was supported by the JSPS
KAKENHI Grant Number 16H06134.
17. Shiota K, Izato Y, Matsunaga H, Habu H, Miyake A. Thermal
properties of ADN, monomethylamine nitrate and urea based
ionic liquid gel propellants. Trans JSASS Aerosp Technol Jpn.
2018;16:93–7.
18. Shiota K, Matsunaga H, Miyake A. Effects of amino acids on
solid-state phase transition of ammonium nitrate. J Therm Anal
Calorim. 2017;127:851–6.
19. Trache D, Khimeche K. Study on the influence of ageing on
thermal decomposition of double-base propellants and prediction
of their in-use time. Fire Matter. 2013;37:328–36.
References
1. Guerya JF, Chang IS, Shimada T, Glick M, Boury D, Robert E,
Napior J, Wardle R, Perut C, Calabro M, Glick R, Habu H,
Sekino N, Vigier G, Andrea BD. Solid propulsion for space
applications: an updated roadmap. Acta Astronaut.
2010;66:201–19.
20. Trache D, Khimeche K, Mezroua A, Benziane M. Physico-
chemical properties of microcrystalline nitrocellulose from Alfa
grass fibres and its thermal stability. J Therm Anal Calorim.
2016;124:1485–96.
21. Yamaki N, Shiota K, Izato Y, Miyake A. Analysis of the thermal
hazards of 1-butyl-3-methylimidazolium chloride mixtures with
¨
2. Trache D, Klapotke TM, Maiz L, Abd-Elghany M, DeLuca LT.
Recent advances in new oxidizers for solid rocket propulsion.
Green Chem. 2017;19:4711–36.
3. Kumar P. An overview on properties, thermal decomposition, and
combustion behavior of ADN and ADN based solid propellants.
4. Bottaro JC, Penwell PE, Schmitt RJ. 1,1,3,3-Tetraoxo-1,2,3-tri-
azapropene anion, a new oxy anion of nitrogen: the dinitramide
anion and its salts. J Am Chem Soc. 1997;119:9405–10.
5. Pak Z. Some ways to higher environmental safety of solid rocket
propellant application. In: Proceedings of the AIAA/SAE/ASME/
ASEE 29th joint propulsion conference and exhibition. 1993;
AIAA-93-1755.
cellulose and various metals.
2018;133:797–803.
J
Therm Anal Calorim.
22. Amrousse R, Katsumi T, Azuma N, Hori K. Hydroxylammonium
nitrate (HAN)-based green propellant as alternative energy
resource for potential hydrazine substitution: from lab scale to
pilot plant scale-up. Combust Flame. 2017;176:334–48.
23. Izato Y, Koshi M, Miyake A, Habu H. Kinetics analysis of
thermal decomposition of ammonium dinitramide (ADN).
J Therm Anal Calorim. 2017;127:255–64.
¨
6. Ostmark H, Bemm U, Langlet A, Sanden R, Wingborg N. The
properties of ammonium dinitramide (ADN): part 1, basic prop-
erties and spectroscopic data. J Energ Mater. 2000;18:123–8.
7. Venkatachalam S, Santhosh G, Nian KN. An overview on syn-
thetic routes and properties of ammonium dinitramide (ADN) and
other dinitramide salts. Propel Explos Pyrotech. 2004;29:178–87.
24. Karunakaran K. Theoretical prediction of eutectic temperature
and composition. J Sol Chem. 1981;10:431–5.
25. Oxley JC, Smith JL, Zheng W, Rogers E, Coburn MD. Thermal
decomposition studies on ammonium dinitramide (ADN) and 15N
2
and H isotopomers. J Phys Chem A. 1997;101:5642–52.
¨
8. Anflo K, Gronland TA, Wingborg N. Development and testing of
26. Vyazokin S, Wight CA. Ammonium dinitramide: kinetics and
mechanism of thermal decomposition.
1997;101:5653–8.
ADN-based monopropellants in small rocket engines. In: Pro-
ceedings of the 36th AIAA/ASME/SAE/ASEE joint propulsion
conference, 2000;AIAA-2000-3162.
J Phys Chem A.
¨
27. Lobbecke S, Krause H, Pfeil A. Thermal analysis of ammonium
Propel
9. Negri M, Wilhelm M, Hendrich C, Wingborg N, Gediminas L,
Adelow L, Maleix C, Chabernaud P, Brhmi R, Beauchet R,
Batonneau Y, Kappenstein C, Koopmans RJ, Schuh S, Bartok T,
Scharlemann C, Gotzig U, Schwentenwein M. New technologies
for ammonium dinitramide based monopropellant thrusters—the
project RHEFORM. Acta Astronaut. 2018;143:105–17.
10. Matsunaga H, Habu H, Miyake A. Preparation and thermal
decomposition behavior of ammonium dinitramide-based ener-
getic ionic liquid propellant. Sci Technol Energ Mater.
2017;78:69–74.
dinitramide
1997;22:184–8.
decomposition.
Explos
Pyrotech.
28. Kazakov AI, Rubtsov YI, Andrienko LP, Manelis GB. Kinetic of
the thermal decomposition of dinitramide 3. Kinetics of the heat
release during the thermal decomposition of dinitramide ammo-
nium salt in the liquid phase. Russ Chem Bull. 1998;47:379–85.
29. Kazakov AI, Rubtsov YI, Manelis GB. Kinetics and mechanism
of thermal decomposition of dinitramide. Propel Explos Pyrotech.
1999;24:37–42.
30. Pavlov AN, Grebennikov VN, Nazina LD, Nazin GM, Manelis
GB. Thermal decomposition of ammonium dinitramide and
mechanism of anomalous decay of dinitramide salts. Russ Chem
Bull. 1999;48:50–4.
11. Trache D, Khimeche K, Benelmir R, Dahmani A. DSC mea-
surement and prediction of phase diagrams for binary mixtures of
energetic
2013;565:8–16.
materials’
stabilizers.
Thermochim
Acta.
31. Tompa AS. Thermal analysis of ammonium dinitramide (ADN).
Thermochim Acta. 2000;357–8:177–93.
32. Mishra IB, Russell TP. Thermal stability of ammonium dini-
tramide. Thermochim Acta. 2002;384:47–56.
33. Shmakov AG, Korobenichev OP, Bol’shova TA. Thermal
decomposition of ammonium dinitramide vapor in a two-tem-
perature flow reactor. Combst Explos Shock Waves.
2002;38:284–94.
12. Trache D, Khimeche K, Benziane M, Dahmani A. Solid–liquid
phase equilibria for binary mixtures of propellant’s stabilizaers.
J Therm Anal Calorim. 2013;112:215–22.
13. Secordel X, Daigurande D, Beauchet R, Batonneau Y, Kappen-
stein C, Wingborg N. Calculated and experimental binary phase
diagrams for ADN and AN based solid propellant-H2020 GRAIL
project. In: Proceedings of the 7th European conference for
aeronautics and space sciences (EUCASS). 2017.
123

 Matsunaga, Hiroki
Matsunaga, Hiroki
 Katoh, Katsumi
Katoh, Katsumi
 Habu, Hiroto
Habu, Hiroto
 Noda, Masaru
Noda, Masaru
 Miyake, Atsumi
Miyake, Atsumi