M. Niemczak et al. / Tetrahedron xxx (2017) 1e11
9
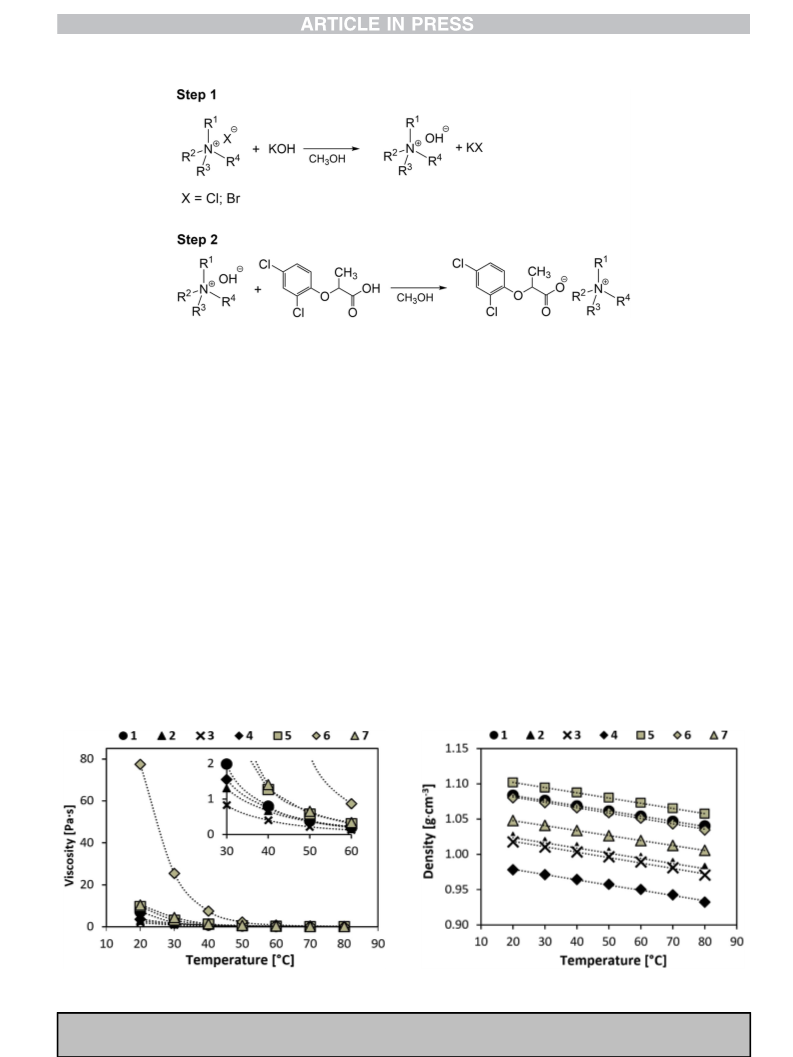
4.12. Preparation of HILs
29.0, 31.2, 31.3, 49.9, 62.7, 76.9,115.4,121.6,122.6,127.4,128.5,153.7,
171.9. IR ymax [cmꢂ1] ¼ 653, 680, 741, 808, 831, 867, 902, 941, 956,
1035, 1060, 1091, 1104, 1127, 1158, 1248, 1268, 1279, 1291, 1360,
1389, 1479, 1614, 2856, 2926. Elemental analysis calculated for
First, 0.015 mol of dried tetraalkylammonium halide was dis-
solved in 25 mL of anhydrous methanol. Then, a stoichiometric
amount of potassium hydroxide (0.84 g, 0.015 mol) dissolved in
25 mL of anhydrous methanol was added. The reaction was con-
ducted for 30 min, and the precipitated inorganic by-product (po-
tassium chloride or potassium bromide) was removed by filtration.
Then, the filtrate containing tetraalkylammonium hydroxide was
neutralized with 2-(2,4-dichlorophenoxy)propionic acid (3.53 g,
0.015 mol). All neutralization reactions were conducted in a Mettler
Toledo semi-automated reactor system EasyMax™ equipped with a
glass electrode at 25 ꢁC. Next, the solvent was removed under
reduced pressure and the residue was dissolved in 50 mL of
acetonitrile. The insoluble impurities were filtered off, and after the
evaporation of the acetonitrile under reduced pressure, the ob-
tained products were dried under vacuum for 24 h at 60 ꢁC.
C
31H55Cl2NO3 (Mmol ¼ 560.61 g molꢂ1) (%): C ¼ 66.41, H ¼ 9.89,
N ¼ 2.50; found: C ¼ 66.15, H ¼ 10.05, N ¼ 2.69. HRMS (ESI-Q-TOF),
(ESþ) for C22H48Nþ [m/z] ¼ 326.3781 (calculated), 326.3785
(found); (ES-) for C9H7Cl2Oꢂ3 [m/z]
232.9773 (found).
¼
232.9778 (calculated),
4.12.4. Di(hydrogenated tallow)dimethylammonium 2-(2,4-
dichlorophenoxy)propionate (4)
Colourless liquid (11.42 g, 92% yield). 1H NMR (300 MHz, DMSO-
d6)
d
[ppm] ¼ 0.74e0.98 (6H, m, CH2CH3), 1.08e1.36 (56H, m,
(CH2)nCH3), 1.37 (3H, d,
J
¼
6.7 Hz, CHCH3), 1.61 (4H, s,
þN(CH2CH2)2), 2.99 (6H, s, þN(CH3)2), 3.13e3.26 (4H, m,
þN(CH2CH2)2), 4.20 (1H, q, J ¼ 7.0 Hz, CHCH3), 6.81 (1H, d,
J ¼ 9.0 Hz, CClCH]CHC), 7.21 (1H, t, J ¼ 5.8 Hz, CClCH]CHC), 7.42
(1H, d, J ¼ 2.5 Hz, CClCH]CCl); 13C NMR (75 MHz, DMSO-d6)
4.12.1. (Hydrogenated tallow)benzyldimethylammonium 2-(2,4-
dichlorophenoxy)propionate (1)
d
[ppm] ¼ 13.9, 19.1, 21.6, 22.1, 25.7, 28.4, 28.7, 29.1, 32.3, 48.6, 76.9,
Colourless liquid (9.16 g, 94% yield). 1H NMR (400 MHz, DMSO-
115.4, 121.6, 122.5, 127.4, 128.5, 153.7, 171.8. IR ymax [cmꢂ1] ¼ 654,
683, 722, 740, 778, 809, 831, 866, 939, 1037, 1059, 1091, 1128, 1151,
1249, 1267, 1279, 1290, 1360, 1388, 1481, 1614, 2856, 2927, 2984.
HRMS (ESI-Q-TOF), (ESþ) for C38H80Nþ [m/z] ¼ 550.6285 (calcu-
lated), 550.6273 (found); (ES-) for C9H7Cl2Oꢂ3 [m/z] ¼ 232.9778
(calculated), 232.9773 (found).
d6)
d
[ppm] ¼ 0.86 (3H, t, J ¼ 5.4 Hz, CH2CH3); 1.16e1.35 (26H, m,
(CH2)nCH3); 1.43 (3H, d, J ¼ 5.7 Hz, CHCH3); 1.69e1.82 (2H, m,
þNCH2CH2); 2.96 (6H, s, þN(CH3)2); 3.22e3.30 (2H, m, þNCH2CH2);
4.27e4.37 (1H, m, CHCH3); 4.59 (2H, s, þNCH2C6H5); 6.88 (1H, d,
J ¼ 8.5 Hz, CClCH]CHC); 7.17e7.23 (1H, m, CClCH]CHC); 7.40 (1H,
d, J ¼ 3.8 Hz, CClCH]CCl); 7.48e7.57 (5H, m, þNCH2C6H5); 13C NMR
(100 MHz, DMSO-d6)
d
[ppm] ¼ 13.9, 19.1, 21.9, 22.1, 25.9, 28.4, 28.8,
4.12.5. Dodecyltrimethylammonium 2-(2,4-dichlorophenoxy)
propionate (5)
28.9, 29.1, 31.4, 48.9, 63.3, 66.0, 76.6, 115.3, 121.7, 122.8, 127.4, 128.3,
128.6, 128.8, 130.1, 133.0, 153.6, 172.3. IR
y
max [cmꢂ1] ¼ 654, 679,
Colourless liquid (6.94 g, 95% yield). 1H NMR (400 MHz, DMSO-
702, 736, 779, 807, 832, 864, 941, 1037, 1056, 1088, 1126, 1152, 1246,
1266, 1280, 1360, 1391, 1478, 1611, 2854, 2955. HRMS (ESI-Q-TOF),
(ESþ) for C21H38Nþ [m/z] ¼ 304.2999 (calculated), 304.3001
d6)
d
[ppm] ¼ 0.86 (3H, t, J ¼ 7.0 Hz, CH2CH3); 1.15e1.36 (18H, m,
(CH2)9CH3); 1.41 (3H, d, J ¼ 7.0 Hz, CHCH3); 1.58e1.68 (2H, m,
þNCH2CH2); 3.06 (9H, s, þN(CH3)3); 3.25e3.32 (2H, m, þNCH2CH2);
4.23 (1H, q, J ¼ 7.0 Hz, CHCH3); 6.86 (1H, d, J ¼ 6.9 Hz, CClCH]CHC);
7.24 (1H, q, J ¼ 4.9 Hz, CClCH]CHC); 7.41 (1H, d, J ¼ 4.0 Hz, CClCH]
(found); (ES-) for C9H7Cl2Oꢂ3 [m/z]
¼
232.9778 (calculated),
232.9770 (found).
CCl); 13C NMR (100 MHz, DMSO-d6)
d
[ppm] ¼ 13.9, 19.1, 22.1, 25.8,
4.12.2. Bis(2-hydroxyethyl)methyloleylammonium 2-(2,4-
dichlorophenoxy)propionate (2)
28.6, 28.7, 28.8, 28.9, 29.0, 31.3, 51.9, 65.1, 76.9, 115.4, 121.6, 122.6,
127.4, 128.5, 153.7, 172.1. IR ymax [cmꢂ1] ¼ 655, 680, 740, 809, 831,
867, 911, 942, 969, 1036, 1058, 1091, 1105, 1125, 1247, 1267, 1280,
1360,1391,1478,1609, 2856, 2927. Elemental analysis calculated for
Colourless liquid (8.97 g, 89% yield). 1H NMR (400 MHz, DMSO-
d6)
d
[ppm]
¼
0.80 (3H, s, eCH2CH3); 1.08e1.32 (20H, m,
(CH2)nCH3); 1.38 (3H, d, J ¼ 5.0 Hz, CHCH3); 1.52e1.65 (2H, m,
þNCH2CH2); 1.85e2.00 (3H, m, CH2CHCHCH2); 3.03 (3H, s, þNCH3);
3.30 (2H, t, J ¼ 9.0 Hz, þNCH2CH2); 3.39 (4H, s, þN(CH2CH2OH)2);
3.76 (4H, t, J ¼ 5.0 Hz, þN(CH2CH2OH)2); 4.20e4.28 (1H, m, CHCH3);
5.21e5.33 (1H, m, CH2CHCHCH2); 6.16 (2H, s, þN(CH2CH2OH)2);
6.77 (1H, t, J ¼ 9.0 Hz, CClCH]CHC); 7.17e7.22 (1H, m, CClCH]
CHC); 7.37 (1H, t, J ¼ 3.0 Hz, CClCH]CCl); 13C NMR (100 MHz,
C
24H41NCl2NO3 (Mmol ¼ 462.50 g molꢂ1) (%): C ¼ 62.33, H ¼ 8.94,
N ¼ 3.03; found: C ¼ 62.58, H ¼ 8.70, N ¼ 2.80. HRMS (ESI-Q-TOF),
(ESþ) for C15H34Nþ [m/z] ¼ 228.2686 (calculated), 228.2685
(found); (ES-) for C9H7Cl2Oꢂ3 [m/z]
¼
232.9778 (calculated),
232.9765 (found).
DMSO-d6)
d
[ppm] ¼ 13.9, 19.1, 21.6, 22.1, 25.5, 25.9, 26.6, 28.6, 29.1,
4.12.6. Trimethyl(tetradecyl)ammonium 2-(2,4-dichlorophenoxy)
propionate (6)
32.3, 32.0, 49.0, 54.7, 62.0, 62.4, 63.3, 76.5, 115.2, 121.8, 122.9, 127.5,
128.7, 129.6, 130.0, 153.6, 173.0. IR ymax [cmꢂ1] ¼ 655, 684, 740, 809,
833, 869, 955, 1041, 1059, 1092, 1130, 1159, 1248, 1268, 1283, 1365,
1394, 1479, 1609, 2856, 2927. HRMS (ESI-Q-TOF), (ESþ) for
Colourless liquid (7.36 g, 93% yield). 1H NMR (300 MHz, DMSO-
d6)
d
[ppm] ¼ 0.85 (3H, t, J ¼ 5.2 Hz, CH2CH3); 1.13e1.37 (22H, m,
(CH2)11CH3); 1.43 (3H, d, J ¼ 5.8 Hz, CHCH3); 1.54e1.72 (2H, m,
þNCH2CH2); 3.05 (9H, s, þN(CH3)3); 3.22e3.34 (2H, m, þNCH2CH2);
4.36 (1H, q, J ¼ 7.0 Hz, CHCH3); 6.88 (1H, d, J ¼ 9.4 Hz, CClCH]CHC);
7.19e7.27 (1H, m, CClCH]CHC); 7.43 (1H, d, J ¼ 2.9 Hz, CClCH]
C
23H48NOþ2 [m/z] ¼ 370.3680 (calculated), 370.3688 (found); (ES-)
for C9H7Cl2Oꢂ3 [m/z] ¼ 232.9778 (calculated), 232.9767 (found).
4.12.3. Didecyldimethylammonium 2-(2,4-dichlorophenoxy)
propionate (3)
CCl); 13C NMR (75 MHz, DMSO-d6)
d
[ppm] ¼ 13.9, 18.9, 22.1, 28.6,
28.7, 28.9, 29.0, 29.01, 31.3, 52.0, 65.1, 76.1, 115.3, 121.8, 123.0, 127.4,
128.7, 153.4, 172.4. IR ymax [cmꢂ1] ¼ 655, 680, 740, 782, 809, 831,
866, 912, 941, 968, 1037, 1059, 1090, 1127, 1156, 1249, 1267, 1285,
1360, 1391, 1480, 1611, 2856, 2929, 2984. Elemental analysis
calculated for C26H45Cl2NO3 (Mmol ¼ 490.55 g molꢂ1) (%): C ¼ 63.66,
H ¼ 9.25, N ¼ 2.86; found: C ¼ 63.92, H ¼ 9.43, N ¼ 2.97. HRMS (ESI-
Colourless liquid (8.41 g, 90% yield). 1H NMR (400 MHz, DMSO-
d6)
d
[ppm] ¼ 0.86 (6H, t, J ¼ 7.6 Hz, CH2CH3); 1.16e1.34 (28H, m,
(CH2)7CH3); 1.40 (3H, d, J ¼ 6.8 Hz, CHCH3); 1.54e1.67 (4H, m,
þN(CH2CH2)2); 2.92e3.09 (6H, m, þN(CH3)2); 3.16e3.32 (4H, m,
þN(CH2CH2)2); 4.16e4.27 (1H, m, CHCH3); 6.85 (1H, d, J ¼ 8.9 Hz,
CClCH]CHC); 7.19e7.24 (1H, m, CClCH]CHC); 7.41 (1H, d,
Q-TOF), (ESþ) for
C
17H38Nþ [m/z]
¼
256.2999 (calculated),
J
d
¼
3.4 Hz, CClCH]CCl); 13C NMR (100 MHz, DMSO-d6)
256.2998 (found); (ES-) for C9H7Cl2Oꢂ3 [m/z] ¼ 232.9778 (calcu-
[ppm] ¼ 13.9, 19.4, 21.7, 22.0, 22.1, 25.8, 28.4, 28.5, 28.7, 28.9, 28.9,
lated), 232.9776 (found).
Please cite this article in press as: Niemczak M, et al., Preparation and characterization of new ionic liquid forms of 2,4-DP herbicide,

 Niemczak, Micha?
Niemczak, Micha?
 Biedziak, Agnieszka
Biedziak, Agnieszka
 Czerniak, Kamil
Czerniak, Kamil
 Marcinkowska, Katarzyna
Marcinkowska, Katarzyna